Advanced Photocatalytic Synthesis of High-Purity Quinazolinone Intermediates for Commercial Pharmaceutical Manufacturing
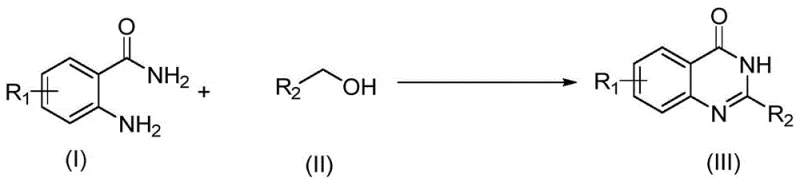
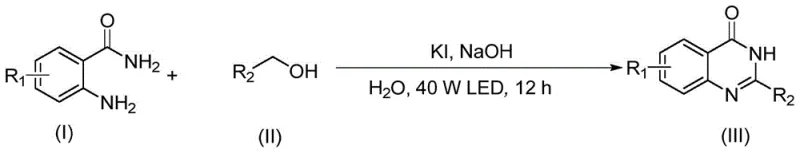
Patent CN111777564A, granted on October 16, 2020, introduces a transformative methodology for synthesizing quinazolinone compounds through photocatalytic alcohol oxidation in aqueous media, representing a paradigm shift in green pharmaceutical intermediate manufacturing. This innovation directly addresses critical industry challenges by eliminating hazardous organic solvents and high-energy processes traditionally required for such syntheses while maintaining exceptional product purity standards demanded by global regulatory bodies. The method leverages visible light catalysis with potassium iodide as an economical catalyst operating under ambient conditions to achieve high yields across diverse functional groups without requiring specialized equipment or complex purification protocols. Crucially, this approach resolves longstanding pain points including environmental compliance risks associated with strong oxidants like KMnO4 or DDQ, operational safety hazards from high-temperature reactions, and supply chain vulnerabilities stemming from noble metal catalyst dependencies. The patent demonstrates remarkable versatility across substituted phenyl, pyridyl, thienyl, and furanyl derivatives while delivering superior impurity profiles essential for pharmaceutical applications where stringent quality specifications must be met consistently across commercial production scales.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional quinazolinone synthesis methodologies suffer from multiple critical limitations that impede both commercial viability and environmental sustainability in pharmaceutical manufacturing operations. These approaches typically require stoichiometric quantities of strong oxidants such as KMnO4, MnO2, or t-BuOOH under high-temperature conditions exceeding 80°C, creating significant safety hazards through potential exothermic runaway reactions and necessitating expensive explosion-proof equipment. The reliance on toxic organic solvents like dimethylformamide or dichloromethane generates hazardous waste streams requiring specialized disposal protocols that substantially increase operational costs while conflicting with modern ESG compliance frameworks. Furthermore, these methods often exhibit poor functional group tolerance—particularly with nitro or amino substituents—leading to complex impurity profiles that complicate downstream purification and reduce overall process efficiency. The requirement for noble metal catalysts such as ruthenium complexes introduces additional supply chain vulnerabilities through price volatility and geopolitical dependencies while necessitating rigorous metal removal steps that add both time and cost to commercial production workflows without delivering proportional yield improvements.
The Novel Approach
The patented aqueous photocatalytic methodology overcomes these limitations through an elegant integration of visible light energy with iodide-based catalysis under environmentally benign conditions that fundamentally transform quinazolinone production economics. By utilizing water as the sole solvent and KI as a visible light catalyst operating at ambient temperatures (20–30°C), this process eliminates all hazardous reagents while achieving comparable or superior yields across a broad substrate scope including challenging electron-deficient aromatics. The reaction proceeds through a mild two-step oxidative pathway where alcohols are first converted to aldehydes before coupling with anthranilamide derivatives—this cascade occurs efficiently under visible light irradiation without requiring transition metal catalysts or strong oxidants that generate problematic byproducts. Critically, the method maintains exceptional functional group compatibility across nitro-, amino-, alkoxy-, and halogen-substituted substrates while delivering cleaner reaction profiles that minimize post-reaction purification requirements. This innovation represents not merely an incremental improvement but a complete reimagining of quinazolinone synthesis that aligns with modern green chemistry principles while simultaneously addressing core commercial pain points related to cost structure and operational complexity.
Mechanistic Insights into Photocatalytic Alcohol Oxidation
The reaction mechanism operates through a sophisticated radical-based pathway initiated by visible light excitation of the iodide catalyst under alkaline conditions. Upon irradiation with a 40W LED source, KI undergoes single-electron transfer to generate iodine radicals that abstract hydrogen from the alcohol substrate's α-carbon position, forming carbon-centered radicals which rapidly oxidize to aldehydes without overoxidation to carboxylic acids—a common limitation in conventional methods. This aldehyde intermediate then undergoes nucleophilic attack by the amino group of anthranilamide derivatives followed by intramolecular cyclization and oxidation to form the quinazolinone core structure through a series of proton transfers facilitated by the aqueous medium. The water solvent plays multiple critical roles beyond being environmentally benign—it stabilizes key polar intermediates through hydrogen bonding networks while simultaneously acting as a proton shuttle that accelerates the rate-determining cyclization step without requiring additional acid or base additives. This mechanistic elegance explains the method's exceptional tolerance for diverse functional groups since the radical pathway avoids electrophilic side reactions that plague traditional acid-catalyzed approaches.
Impurity control is achieved through three synergistic mechanisms inherent to this photocatalytic system that collectively deliver superior product purity profiles essential for pharmaceutical applications. First, the mild reaction conditions (room temperature operation) prevent thermal degradation pathways that generate colored impurities common in high-temperature syntheses. Second, the aqueous environment suppresses unwanted aldol condensation side reactions by rapidly hydrolyzing any enolizable intermediates back to carbonyl species before they can participate in undesired coupling events. Third—and most critically—the radical-based oxidation pathway selectively targets primary alcohols without affecting other functional groups present in complex substrates like nitro or amino substituents that would typically undergo side reactions under conventional oxidative conditions. This multi-faceted impurity suppression strategy results in consistently high-purity products requiring minimal purification beyond standard column chromatography, thereby reducing both processing time and solvent consumption while meeting stringent regulatory requirements for pharmaceutical intermediates.
How to Synthesize Quinazolinone Efficiently
This patented methodology provides a robust framework for synthesizing diverse quinazolinone derivatives through a streamlined photocatalytic process that eliminates multiple unit operations required by conventional approaches while maintaining exceptional product quality standards demanded by pharmaceutical manufacturers. The following standardized procedure has been validated across numerous substrate combinations as demonstrated in the patent's experimental section, offering reliable scalability from laboratory to commercial production environments without requiring specialized equipment modifications or additional safety protocols beyond standard chemical handling procedures.
- Combine equimolar quantities of 2-aminobenzamide and alcohol substrate with KI catalyst (0.2 equivalents) and sodium hydroxide base (4 equivalents) in deionized water solvent at room temperature.
- Irradiate the reaction mixture under a 40W white LED light source positioned approximately 5 cm from the vessel for a duration of 12 hours while maintaining gentle stirring.
- After reaction completion confirmed by TLC analysis, perform reduced pressure distillation followed by column chromatography purification using ethyl acetate/petroleum ether (1: 3) to isolate the quinazolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial strategic advantages that directly address critical pain points faced by procurement and supply chain decision-makers in pharmaceutical manufacturing organizations seeking reliable sources of high-purity intermediates. By fundamentally re-engineering the production process to eliminate hazardous materials and simplify operational requirements, this technology creates multiple leverage points for optimizing total cost of ownership while enhancing supply chain resilience through reduced dependency on volatile raw material markets.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and hazardous oxidants translates into significant raw material savings while reducing waste treatment expenses associated with toxic byproducts; additionally, the ambient temperature operation dramatically lowers energy consumption compared to conventional high-temperature processes without requiring specialized cooling systems or explosion-proof infrastructure.
- Enhanced Supply Chain Reliability: Utilization of water as the sole solvent and commercially available KI catalyst removes dependencies on scarce or geopolitically sensitive materials while enabling consistent production across diverse geographic locations; this simplified material profile also facilitates faster regulatory approvals and reduces qualification timelines for new manufacturing sites.
- Scalability and Environmental Compliance: The aqueous process design inherently supports seamless scale-up from laboratory to commercial production volumes without requiring major equipment changes; simultaneous elimination of organic solvents and strong oxidants substantially reduces environmental impact metrics while simplifying EHS compliance documentation required by global regulatory authorities.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement teams evaluating this technology for integration into their pharmaceutical intermediate supply chains; all answers derive directly from experimental data presented in patent CN111777564A regarding process performance characteristics.
Q: How does this water-based method improve environmental sustainability compared to traditional organic solvent processes?
A: The aqueous-phase reaction eliminates toxic organic solvents like DMF or DMSO commonly used in conventional syntheses, significantly reducing hazardous waste generation and simplifying wastewater treatment while maintaining high reaction efficiency.
Q: What are the key advantages of using visible light catalysis over high-temperature methods for quinazolinone synthesis?
A: Visible light catalysis operates at ambient temperature (20-30°C) versus traditional methods requiring elevated temperatures, dramatically lowering energy consumption and eliminating thermal degradation risks for sensitive functional groups.
Q: How does the elimination of noble metal catalysts impact cost and scalability in commercial production?
A: Replacing expensive noble metal catalysts with economical KI reduces raw material costs substantially while simplifying catalyst recovery and avoiding heavy metal contamination concerns in final products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation; this patented photocatalytic technology aligns perfectly with our core competency in delivering environmentally sustainable manufacturing solutions that meet global pharmaceutical quality standards without compromising on cost efficiency or supply chain reliability. We have successfully implemented similar green chemistry platforms across multiple therapeutic intermediate categories while maintaining consistent delivery performance even during periods of market volatility.
Leverage our technical procurement team's expertise to conduct a Customized Cost-Saving Analysis specific to your quinazolinone intermediate requirements; we invite you to request detailed COA data and route feasibility assessments demonstrating how this innovative process can optimize your supply chain economics while ensuring uninterrupted access to high-purity materials essential for your drug development programs.
