Scalable Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Advanced Pharmaceutical Intermediates
Introduction to Advanced Triazole Synthesis Technology
The pharmaceutical and agrochemical industries continuously demand robust synthetic methodologies for constructing nitrogen-containing heterocycles, particularly 1,2,3-triazoles, which serve as privileged scaffolds in drug discovery. Patent CN113121462B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing critical limitations in existing synthetic routes. The introduction of a trifluoromethyl group is strategically vital for enhancing the metabolic stability, lipophilicity, and bioavailability of parent molecules, making these intermediates indispensable for modern medicinal chemistry programs. This novel technology leverages a base-promoted cyclization strategy that circumvents the use of hazardous azides and expensive transition metal catalysts, offering a safer and more economically viable pathway for the commercial scale-up of complex pharmaceutical intermediates. By utilizing readily available trifluoroethylimidoyl chlorides and diazo compounds, this process represents a significant leap forward in green chemistry and process efficiency.
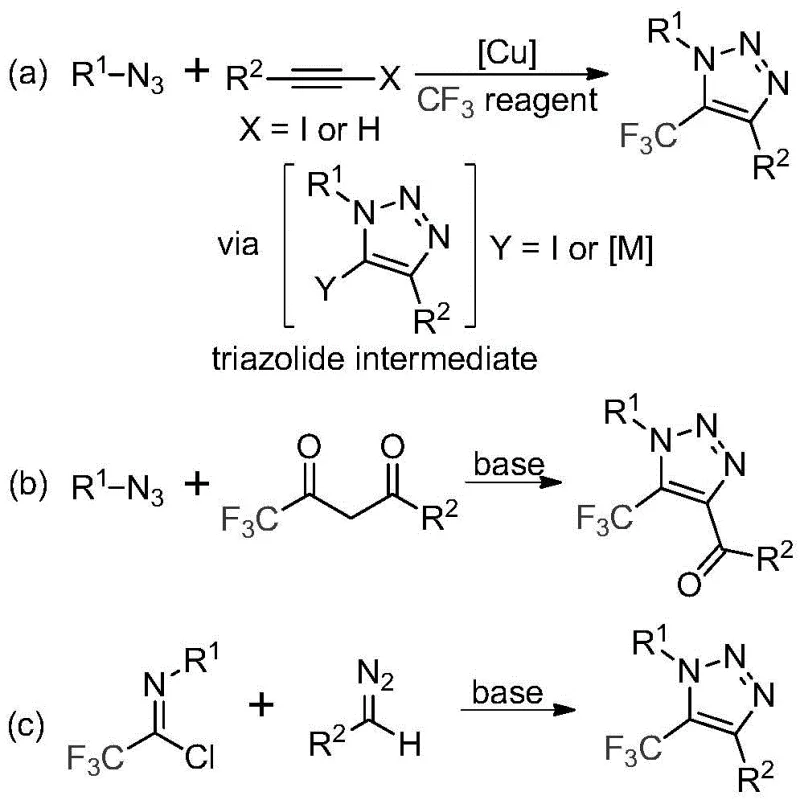
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on two primary strategies, both of which present substantial operational and safety challenges for industrial manufacturers. The first conventional approach involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation; this route necessitates the handling of highly explosive organic azides and introduces toxic copper residues that require rigorous and costly purification steps to meet stringent pharmaceutical standards. The second traditional method employs organocatalytic 1,3-dipolar cycloaddition between azides and trifluoromethyl ketones, which similarly suffers from the inherent safety risks associated with azide chemistry and often requires harsh reaction conditions that limit functional group tolerance. These legacy methods not only pose significant safety hazards regarding explosion risks but also complicate the supply chain due to the specialized handling requirements for energetic materials and the environmental burden of heavy metal waste disposal.
The Novel Approach
In stark contrast to these hazardous legacy protocols, the technology described in patent CN113121462B introduces a transformative metal-free and azide-free synthetic route that fundamentally reshapes the manufacturing landscape for these valuable heterocycles. As illustrated in the reaction scheme below, the novel method utilizes a simple yet effective base-promoted reaction between trifluoroethylimidoyl chlorides and diazo compounds to construct the triazole ring directly. This approach completely eliminates the need for explosive azide precursors and toxic transition metal catalysts, thereby drastically simplifying the safety protocols required for production. Furthermore, the reaction proceeds under mild thermal conditions using inexpensive inorganic bases like cesium carbonate, which facilitates a cleaner reaction profile with fewer byproducts. This paradigm shift enables manufacturers to achieve cost reduction in pharmaceutical intermediate manufacturing by removing expensive catalyst recovery steps and minimizing waste treatment costs associated with heavy metals.
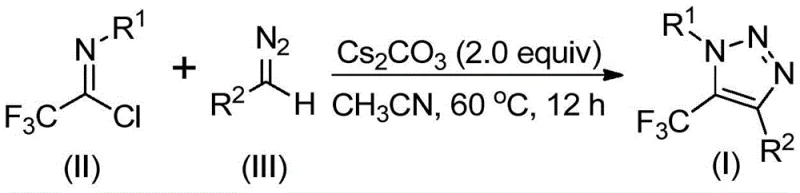
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic pathway of this innovative synthesis involves a sophisticated sequence of base-mediated transformations that ensure high regioselectivity and yield. The reaction is initiated by the deprotonation of the diazo compound or the activation of the imidoyl chloride by the cesium carbonate base, promoting an intermolecular nucleophilic addition-elimination process. This initial step facilitates the formation of a critical carbon-carbon bond between the diazo species and the imidoyl chloride framework. Subsequently, the intermediate undergoes an intramolecular 5-endo-dig cyclization, a kinetically favorable process that closes the five-membered triazole ring with high precision. The use of polar aprotic solvents such as acetonitrile is crucial in this mechanism, as it effectively solvates the ionic intermediates and stabilizes the transition states, thereby driving the reaction to completion with remarkable efficiency. This mechanistic understanding allows process chemists to fine-tune reaction parameters to maximize throughput while maintaining the structural integrity of sensitive functional groups attached to the R1 and R2 positions.
From an impurity control perspective, the absence of transition metals and the mild basic conditions contribute to an exceptionally clean impurity profile, which is a paramount concern for high-purity pharmaceutical intermediates. Traditional copper-catalyzed routes often generate difficult-to-remove metal complexes and homocoupling byproducts that can persist through multiple purification stages, potentially compromising the safety of the final drug substance. In this novel base-promoted system, the primary byproducts are typically inorganic salts and benign organic fragments that are easily removed during standard aqueous workup or silica gel chromatography. The high functional group tolerance observed across various substrates, including those with electron-withdrawing halogens and electron-donating alkoxy groups, indicates that the reaction mechanism is robust against electronic perturbations. This reliability ensures consistent batch-to-batch quality, a critical metric for reliable pharmaceutical intermediate suppliers serving regulated markets where impurity thresholds are strictly enforced by global health authorities.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazoles Efficiently
The practical implementation of this synthesis protocol is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot-plant production. The standard procedure involves charging a reaction vessel with cesium carbonate, trifluoroethylimidoyl chloride, and the corresponding diazo compound in anhydrous acetonitrile. The mixture is then heated to a moderate temperature range of 50-70°C, typically maintained at 60°C for optimal kinetics, and stirred for a duration of 8 to 16 hours. Upon completion, the reaction mixture is filtered to remove inorganic salts, and the filtrate is concentrated to afford the crude product, which is subsequently purified via column chromatography. For detailed standardized operating procedures and specific stoichiometric ratios tailored to different substrates, please refer to the technical guide below.
- Combine trifluoroethylimidoyl chloride, diazo compound, and cesium carbonate in anhydrous acetonitrile under inert atmosphere.
- Heat the reaction mixture to 60°C and stir for 12 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route offers compelling strategic advantages that extend beyond mere technical feasibility. The elimination of copper catalysts and explosive azides translates directly into a simplified logistics framework, as the raw materials involved—such as cesium carbonate and acetonitrile—are commodity chemicals with stable global supply chains and predictable pricing structures. This stability mitigates the risk of supply disruptions often associated with specialized reagents or precious metals, ensuring enhanced supply chain reliability for long-term production contracts. Moreover, the mild reaction conditions reduce the energy consumption required for heating and cooling, contributing to a lower overall carbon footprint and aligning with increasingly stringent environmental, social, and governance (ESG) mandates imposed by multinational corporations.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of expensive noble metal catalysts and the associated downstream purification costs. In traditional methods, the removal of trace copper to parts-per-million levels often requires specialized scavengers or additional chromatography steps, which significantly inflate the cost of goods sold (COGS). By adopting this base-promoted method, manufacturers can bypass these costly remediation steps entirely. Additionally, the high atom economy and excellent yields reported in the patent data suggest that less raw material is wasted, further optimizing the material cost per kilogram of the final product. The use of inexpensive inorganic bases instead of proprietary organocatalysts also contributes to a substantial reduction in reagent costs, making the overall process financially attractive for large-volume production.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials ensures that production schedules are not held hostage by the lead times of exotic reagents. Trifluoroethylimidoyl chlorides and diazo compounds used in this method can be sourced from multiple vendors or synthesized in-house using well-established protocols, creating a resilient supply network. This diversification of supply sources reduces the vulnerability to single-source bottlenecks, a critical consideration for maintaining continuous manufacturing operations. Furthermore, the non-hazardous nature of the reagents simplifies storage and transportation requirements, lowering insurance premiums and logistical complexities associated with shipping dangerous goods across international borders.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, having been successfully validated from milligram to gram scales in the patent examples, with clear pathways for extension to multi-kilogram and ton-scale production. The absence of heavy metals simplifies wastewater treatment and waste disposal, as the effluent does not require complex heavy metal precipitation or recovery systems. This ease of waste management facilitates compliance with environmental regulations in various jurisdictions, reducing the administrative burden and potential fines associated with hazardous waste discharge. The straightforward workup procedure, involving simple filtration and concentration, is inherently easier to automate and scale in continuous flow reactors or large batch vessels compared to complex multi-step catalytic cycles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this technology within their existing manufacturing portfolios and R&D pipelines.
Q: Why is this metal-free method superior to copper-catalyzed routes?
A: This method eliminates the need for toxic copper catalysts and explosive organic azides, significantly improving safety profiles and reducing heavy metal impurity risks in the final API.
Q: What is the substrate scope for R1 and R2 groups?
A: The process demonstrates excellent tolerance for various substituents, including substituted aryl groups (methyl, methoxy, halogens) and functional groups like esters, ketones, and phosphonates.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of inexpensive bases like cesium carbonate and common solvents like acetonitrile, combined with mild temperatures (60°C), makes the process highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to leverage this advanced synthesis technology for your specific project needs. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole intermediates meets the highest industry standards for potency and impurity profiles. We understand the critical nature of timeline and quality in the pharmaceutical sector and are committed to delivering consistent results that support your drug development goals.
We invite you to engage with our technical procurement team to discuss how this metal-free route can be integrated into your supply chain to drive efficiency and value. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence and our proven track record in delivering complex organic intermediates.
