Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Scale-Up
Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,3-Triazoles for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those functionalized with fluorine atoms to enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in Chinese Patent CN113121462B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds. This technology represents a paradigm shift from traditional transition-metal catalysis to a more sustainable, base-promoted organic synthesis. For R&D directors and procurement strategists, this patent offers a compelling alternative to legacy routes, promising streamlined operations and reduced regulatory burdens associated with heavy metal residues. The ability to access these privileged scaffolds efficiently is critical for the development of next-generation active pharmaceutical ingredients (APIs) and crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on copper-catalyzed azide-alkyne cycloaddition (CuAAC) reactions or organocatalytic variants involving trifluoromethyl ketones. As illustrated in the reaction schemes below, these conventional pathways often necessitate the use of organic azides, which are notoriously toxic, unstable, and prone to explosive decomposition, posing severe safety risks during scale-up. Furthermore, the reliance on copper catalysts introduces a significant downstream purification challenge, requiring rigorous metal scavenging steps to meet the stringent ppm limits imposed by global health authorities for pharmaceutical products. These additional processing steps not only inflate the cost of goods sold (COGS) but also extend the overall production lead time, creating bottlenecks in the supply chain for high-purity intermediates.
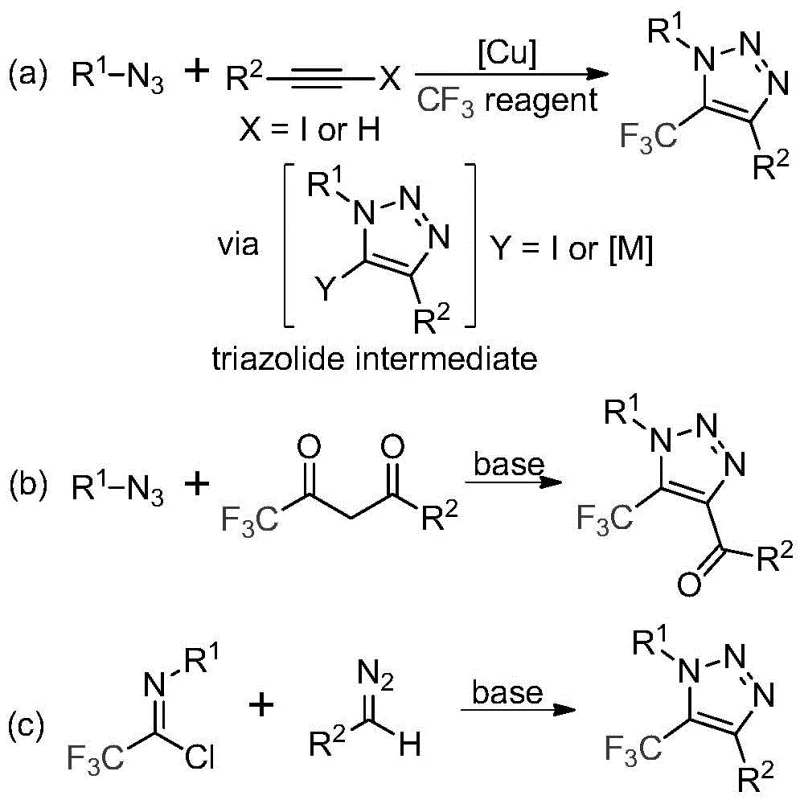
The Novel Approach
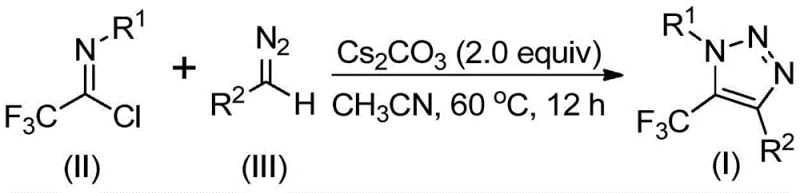
In stark contrast to these hazardous legacy methods, the technology described in patent CN113121462B utilizes a metal-free, azide-free strategy that leverages readily available diazo compounds and trifluoroethylimidoyl chlorides. This innovative route eliminates the need for explosive reagents and expensive transition metals, fundamentally altering the risk profile of the manufacturing process. By employing a simple base promoter like cesium carbonate in a polar aprotic solvent, the reaction achieves high efficiency under mild thermal conditions. This approach not only simplifies the reaction setup but also drastically reduces the complexity of the workup procedure, allowing for a more direct path to the final product. The versatility of this method is evident in its tolerance for various functional groups, enabling the synthesis of diverse derivatives suitable for medicinal chemistry optimization campaigns without the baggage of metal contamination.
Mechanistic Insights into Base-Promoted Cyclization
The core of this synthetic innovation lies in a unique mechanistic pathway that diverges from the classic 1,3-dipolar cycloaddition. The reaction is hypothesized to initiate with a base-promoted intermolecular nucleophilic addition-elimination sequence, where the diazo species attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step facilitates the formation of a critical carbon-carbon bond, setting the stage for the subsequent ring closure. Following this initial coupling, the intermediate undergoes an intramolecular 5-endo-dig cyclization, a process that is thermodynamically favorable under the specified reaction conditions. This mechanism avoids the formation of stable metal-triazolide complexes that often require harsh protonolysis or transmetallation to release the product, thereby preserving sensitive functional groups that might otherwise degrade under stronger acidic or basic conditions typically associated with metal catalysis.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring product quality. The absence of transition metals removes a major source of colored impurities and potential genotoxic contaminants, simplifying the purification landscape significantly. The use of cesium carbonate as a mild base ensures that the reaction environment remains controlled, minimizing side reactions such as hydrolysis of the imidoyl chloride or decomposition of the diazo compound. Experimental data from the patent indicates that optimizing the stoichiometry, specifically using a slight excess of the diazo compound relative to the imidoyl chloride, drives the reaction to completion while suppressing dimerization byproducts. This precise control over the reaction trajectory results in cleaner crude profiles, which translates directly to higher isolated yields and reduced solvent consumption during the final chromatographic purification steps.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and reproducibility. The process is designed to be operationally simple, utilizing standard glassware and common organic solvents. The key to success lies in the careful selection of the base and the maintenance of anhydrous conditions to prevent the hydrolysis of the reactive imidoyl chloride starting material. Detailed procedural guidelines ensure that chemists can replicate the high yields reported in the patent examples, ranging from moderate to excellent depending on the electronic nature of the substituents. For a comprehensive, step-by-step technical guide on executing this transformation, please refer to the standardized protocol section below.
- Mix cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in acetonitrile solvent.
- Heat the reaction mixture to 60°C and stir for approximately 12 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via column chromatography to obtain the final triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers transformative economic and logistical benefits. The shift away from specialized catalysts and hazardous reagents aligns perfectly with modern green chemistry principles while delivering tangible bottom-line improvements. By simplifying the raw material portfolio and reducing the number of unit operations required for purification, manufacturers can achieve substantial cost savings and enhanced supply reliability. This methodology transforms the production of these valuable heterocycles from a high-risk, high-cost endeavor into a routine, scalable commodity process.
- Cost Reduction in Manufacturing: The elimination of copper catalysts and ligands removes a significant cost center from the bill of materials. Transition metals, especially those required for high-turnover catalysis, can be prohibitively expensive, and their removal often necessitates costly scavenger resins or extensive recrystallization processes. Furthermore, avoiding organic azides mitigates the need for specialized safety infrastructure and explosion-proof equipment, lowering capital expenditure requirements. The use of cesium carbonate, a relatively inexpensive and stable inorganic base, further drives down reagent costs compared to exotic organocatalysts. Consequently, the overall cost reduction in API manufacturing is achieved through both lower direct material costs and reduced processing overheads associated with safety and purification.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, specifically trifluoroethylimidoyl chlorides and diazo compounds, are derived from widely available commodity chemicals such as aromatic amines and acid chlorides. This reliance on bulk chemical feedstocks ensures a stable and resilient supply chain, less susceptible to the bottlenecks often seen with niche catalysts or custom-synthesized reagents. The robustness of the reaction conditions, which tolerate a wide range of functional groups, means that suppliers can source diverse substrates without needing to qualify new, fragile reagents for every analog. This flexibility allows for rapid response to changing market demands and ensures continuity of supply for critical pharmaceutical intermediates even during periods of raw material volatility.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but this base-promoted method is inherently scalable due to its mild thermal profile and lack of exothermic hazards associated with azide handling. The reaction operates effectively at moderate temperatures (around 60°C), reducing energy consumption for heating and cooling compared to cryogenic or high-temperature alternatives. Additionally, the absence of heavy metals simplifies waste stream management, facilitating easier compliance with increasingly strict environmental regulations regarding metal discharge. The commercial scale-up of complex heterocycles via this route is therefore not only technically feasible but also environmentally sustainable, positioning manufacturers favorably for audits and regulatory approvals in major markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is essential for project managers evaluating the feasibility of integrating this route into their existing production portfolios.
Q: Does this synthesis method require toxic azide reagents?
A: No, unlike traditional copper-catalyzed methods, this patent describes a metal-free process that avoids the use of hazardous and explosive organic azides, significantly improving operational safety.
Q: What are the typical reaction conditions for this triazole formation?
A: The reaction typically proceeds in acetonitrile at mild temperatures around 60°C using cesium carbonate as a promoter, completing within 8 to 16 hours.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process utilizes cheap, commercially available starting materials and avoids expensive transition metal catalysts, making it highly scalable and cost-effective for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient heterocyclic synthesis in accelerating drug discovery and development pipelines. Our team of expert process chemists has extensively evaluated the methodology described in CN113121462B and possesses the technical capability to adapt and optimize this route for your specific project needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole delivered meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming programs. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and target timelines. We encourage you to reach out today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us help you secure a reliable supply of high-quality intermediates while optimizing your overall manufacturing economics.
