Scalable Electrochemical Synthesis of Tetra-Substituted Hydrazines for Commercial Pharmaceutical Applications
Scalable Electrochemical Synthesis of Tetra-Substituted Hydrazines for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to synthesize complex nitrogen-containing scaffolds, particularly hydrazine derivatives which serve as critical building blocks for active pharmaceutical ingredients (APIs). Patent CN112126942B introduces a groundbreaking electrochemical methodology that achieves the N-N coupling of secondary aromatic amines under mild, metal-free conditions. This technology represents a paradigm shift from traditional stoichiometric oxidation methods, utilizing electrons as clean reagents to drive the formation of tetra-substituted hydrazines with high atom economy. By leveraging a simple undivided cell setup and inexpensive electrolytes, this process addresses the growing demand for sustainable manufacturing practices while maintaining rigorous quality standards required for drug substance production.
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the limitations of conventional transition metal catalysis. The described method employs N-methylaniline analogues as readily available starting materials, subjecting them to anodic oxidation followed by cathodic reduction to forge the N-N bond efficiently. This approach not only simplifies the reaction workflow by removing the need for inert atmosphere protection often required in sensitive organometallic chemistry but also drastically reduces the environmental footprint associated with heavy metal waste disposal. As a reliable pharmaceutical intermediate supplier, understanding such innovative synthetic routes is essential for optimizing supply chains and ensuring the continuous availability of high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted hydrazine compounds has relied heavily on transition metal catalysis or the use of strong chemical oxidants, both of which present significant challenges for large-scale manufacturing. Traditional methods often require expensive palladium or copper catalysts that necessitate complex removal steps to meet stringent residual metal specifications mandated by regulatory bodies like the FDA and EMA. Furthermore, the reliance on stoichiometric oxidants generates substantial amounts of hazardous waste, complicating effluent treatment and increasing the overall cost of goods sold (COGS). These conventional routes frequently suffer from limited substrate scope, poor atom economy, and safety concerns related to the handling of energetic oxidizing agents, making them less ideal for the commercial scale-up of complex polymer additives or pharmaceutical intermediates.
The Novel Approach
In stark contrast, the electrochemical strategy disclosed in CN112126942B offers a streamlined alternative that utilizes electricity as the primary driving force for oxidative dehydrogenation. This novel approach operates in an undivided single-compartment cell using a graphite anode and a platinum cathode, eliminating the need for diaphragms and reducing equipment complexity. The reaction proceeds smoothly in a mixed solvent system of acetonitrile, water, and ethanol, utilizing sodium nitrite as a cost-effective electrolyte mediator. By avoiding external chemical oxidants and metal catalysts, this method inherently produces a cleaner crude reaction mixture, thereby facilitating easier purification and enhancing the overall sustainability profile of the manufacturing process.

The versatility of this electrochemical protocol is further demonstrated by its compatibility with a wide range of substrates, including symmetric and asymmetric diaryl amines as well as heteroaryl variants. The general reaction scheme illustrates how N-methylaniline analogues (Formula I) are efficiently converted into the corresponding tetra-substituted hydrazines (Formula II) through a direct oxidative coupling mechanism. This broad substrate tolerance is crucial for medicinal chemists who require diverse structural motifs for structure-activity relationship (SAR) studies. Moreover, the mild reaction conditions (50°C) ensure that sensitive functional groups remain intact, preventing decomposition and preserving the integrity of the molecular scaffold throughout the synthesis.
Mechanistic Insights into Electrochemical Oxidative Coupling
The core of this technology relies on the precise control of electrochemical potential to generate reactive radical intermediates at the anode surface without over-oxidizing the substrate. During the electrolysis, the secondary amine undergoes a single-electron transfer (SET) at the graphite anode to form a radical cation, which subsequently dimerizes to form the N-N bond. Simultaneously, protons generated during the oxidation process are reduced at the platinum cathode to evolve hydrogen gas, which serves as the sole theoretical byproduct of the reaction. This elegant redox-neutral balance ensures high atom efficiency and minimizes the formation of unwanted side products that typically plague thermal oxidation methods. Understanding this mechanism allows process engineers to fine-tune current density and electrolyte concentration to maximize yield and selectivity.
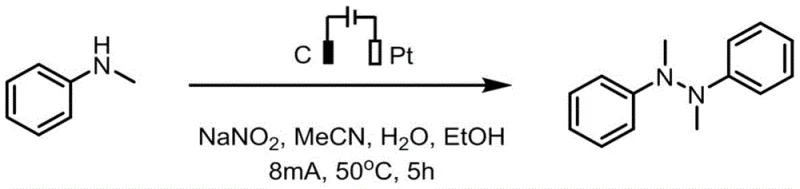
Impurity control is another critical aspect where this electrochemical method excels, particularly for applications requiring high-purity OLED material or API intermediates. The use of sodium nitrite as an electrolyte not only enhances conductivity but may also play a mediating role in the electron transfer process, smoothing the oxidation potential and preventing localized hotspots that could lead to degradation. The specific synthesis example shown above highlights the conversion of N-methylaniline into its dimeric hydrazine form with an impressive 80% isolated yield. Such high efficiency, combined with the absence of metal contaminants, significantly reduces the burden on downstream purification units, allowing for a more robust and predictable manufacturing campaign that aligns with modern green chemistry principles.
How to Synthesize Tetra-Substituted Hydrazines Efficiently
To implement this synthesis effectively, operators must adhere to specific parameters regarding solvent composition and current density to ensure reproducibility and safety. The patent outlines a standardized procedure involving the dissolution of the amine substrate and sodium nitrite in a ternary solvent mixture, followed by constant current electrolysis at 8mA. Detailed operational guidelines emphasize the importance of maintaining a temperature of 50°C and a stirring rate of 450 rpm to ensure uniform mass transfer and heat dissipation throughout the reaction vessel. For a comprehensive breakdown of the experimental protocol, please refer to the step-by-step guide below.
- Prepare the electrolytic cell by adding N-methylaniline analogue, sodium nitrite electrolyte, and a mixed solvent of acetonitrile, water, and ethanol.
- Insert a graphite carbon rod anode and a platinum sheet cathode into the undivided cell and initiate constant current electrolysis at 8mA.
- Maintain the reaction at 50°C for 5 hours with stirring, then purify the resulting tetra-substituted hydrazine via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this electrochemical technology offers substantial strategic benefits by decoupling production costs from the volatile pricing of precious metal catalysts. The elimination of palladium or copper catalysts removes a major cost driver and mitigates supply chain risks associated with the sourcing of these critical raw materials. Additionally, the use of commodity chemicals like sodium nitrite and common solvents ensures a stable and resilient supply base, reducing the likelihood of production delays due to material shortages. This stability is paramount for supply chain heads managing the commercial scale-up of complex organic intermediates where continuity of supply is non-negotiable.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and stoichiometric oxidants leads to a drastic simplification of the bill of materials (BOM). Without the need for costly metal scavengers or extensive washing steps to remove metal residues, the overall processing time and utility consumption are significantly lowered. This streamlined workflow translates directly into improved margin profiles and a more competitive pricing structure for the final hydrazine products, enabling cost reduction in fine chemical manufacturing without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on widely available, non-hazardous reagents such as sodium nitrite and graphite electrodes enhances the robustness of the supply chain. Unlike specialized organometallic reagents that may have long lead times or strict storage requirements, the inputs for this electrochemical process are stable and easily sourced from multiple vendors. This flexibility allows procurement teams to negotiate better terms and maintain optimal inventory levels, effectively reducing lead time for high-purity hydrazine derivatives and ensuring timely delivery to downstream customers.
- Scalability and Environmental Compliance: The simplicity of the undivided cell design facilitates easy scale-up from laboratory benchtop to pilot plant and eventually to full commercial production. The inherent safety of operating at mild temperatures and ambient pressure, combined with the generation of hydrogen gas as the only byproduct, simplifies regulatory compliance and waste management. This alignment with environmental, social, and governance (ESG) goals makes the process highly attractive for companies aiming to reduce their carbon footprint and adhere to increasingly strict environmental regulations in the chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical N-N coupling technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on process capabilities and product quality. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this method into existing production lines.
Q: What are the primary advantages of this electrochemical method over traditional metal-catalyzed coupling?
A: This method eliminates the need for expensive transition metal catalysts and harsh chemical oxidants, significantly reducing heavy metal contamination risks and simplifying downstream purification processes.
Q: Is this process suitable for large-scale industrial production of hydrazine derivatives?
A: Yes, the use of simple undivided cells, inexpensive electrolytes like sodium nitrite, and mild reaction conditions (50°C) makes the process highly scalable and economically viable for commercial manufacturing.
Q: What is the expected purity profile of the synthesized tetra-substituted hydrazines?
A: The mild electrochemical conditions minimize side reactions and over-oxidation, allowing for the isolation of high-purity products (up to 80% yield in optimized examples) after standard chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetra-Substituted Hydrazines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of high-value pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112126942B can be successfully translated into robust industrial processes. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of tetra-substituted hydrazines adheres to the highest international standards for safety and efficacy.
We invite global partners to collaborate with us to leverage this cutting-edge technology for their specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can drive value and efficiency in your supply chain.
