Advanced Electrochemical N-N Coupling for High-Purity Hydrazine Intermediates and Commercial Scale-Up
The landscape of organic synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, particularly in the production of critical nitrogen-containing scaffolds. Patent CN112126942A introduces a groundbreaking electrochemical approach for the N-N coupling of secondary aromatic amines, addressing long-standing inefficiencies in the manufacture of polysubstituted hydrazine compounds. This technology leverages electrons as clean reagents to facilitate direct oxidative dehydrogenation coupling, theoretically releasing only hydrogen gas as a byproduct. For R&D directors and process chemists, this represents a significant advancement in atom economy and environmental compliance. The method transforms simple N-methylaniline analogs into valuable tetra-substituted hydrazines through a single-component self-coupling mechanism under mild conditions. By eliminating the need for stoichiometric chemical oxidants and transition metal catalysts, this process not only reduces the environmental footprint but also streamlines the purification workflow. The implications for the supply chain of high-purity pharmaceutical intermediates are profound, offering a route that is both economically viable and technically robust for large-scale adoption.

The limitations of conventional methods for synthesizing N-N bonded hydrazine derivatives are well-documented in industrial chemistry literature. Traditional approaches often rely heavily on transition metal catalysis, which introduces significant challenges regarding metal residue removal and regulatory compliance in pharmaceutical manufacturing. Furthermore, many existing protocols require the use of strong chemical oxidants or harsh basic additives, which can lead to safety hazards, increased waste generation, and limited substrate tolerance. These factors collectively contribute to higher production costs and complex downstream processing requirements. In contrast, the novel electrochemical approach disclosed in the patent circumvents these issues by utilizing an undivided cell setup with inexpensive electrolytes. This method avoids the use of expensive noble metal catalysts and toxic oxidizing agents, thereby simplifying the reaction matrix. The ability to operate under mild electrochemical conditions without the need for inert gas protection further enhances the operational simplicity. This shift from reagent-intensive chemistry to electricity-driven synthesis marks a pivotal improvement in the manufacturing of complex amine derivatives, aligning perfectly with modern green chemistry principles.
Mechanistically, the electrochemical N-N coupling proceeds through a sophisticated sequence of anodic oxidation and cathodic reduction events within the undivided cell. The secondary aromatic amine substrate undergoes anodic oxidation to generate a reactive radical cation intermediate, which subsequently dimerizes to form the N-N bond. Simultaneously, the cathodic reduction process balances the charge, typically evolving hydrogen gas, which ensures high atom efficiency. This dual-electrode mechanism allows for precise control over the reaction potential, minimizing side reactions such as over-oxidation or decomposition of the sensitive hydrazine product. From an impurity control perspective, the absence of metal catalysts is a decisive advantage. In traditional metal-catalyzed couplings, trace metals can coordinate with the product or intermediates, leading to difficult-to-remove impurities that require extensive chromatography or scavenging treatments. Here, the primary impurities are likely to be unreacted starting materials or simple over-oxidized byproducts, which are far easier to separate. This results in a cleaner crude reaction profile, reducing the burden on purification teams and increasing the overall yield of the isolated high-purity intermediate.
How to Synthesize Tetra-substituted Hydrazines Efficiently
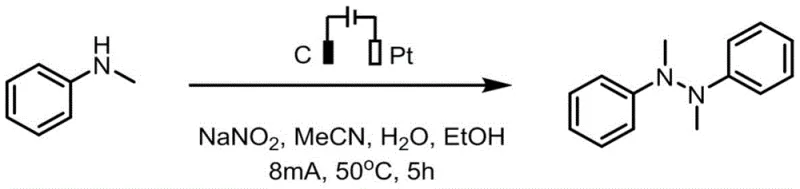
The practical implementation of this electrochemical protocol is designed for ease of operation and reproducibility in both laboratory and pilot plant settings. The synthesis utilizes readily available starting materials such as N-methylaniline and common electrolytes like sodium nitrite, dissolved in a mixed solvent system of acetonitrile, water, and ethanol. The reaction is conducted in a simple undivided cell equipped with a graphite anode and a platinum cathode, applying a constant current under controlled temperature conditions. This straightforward setup eliminates the need for specialized pressure vessels or complex catalyst handling procedures. Detailed standardized synthesis steps, including precise molar ratios, current densities, and workup procedures, are provided in the technical documentation below to ensure consistent results. This accessibility makes the technology highly attractive for process development teams looking to transition from batch to continuous flow electrochemical manufacturing.
- Prepare the electrolytic cell by adding N-methylaniline analogs, sodium nitrite electrolyte, and a mixed solvent system of acetonitrile, water, and ethanol.
- Insert a graphite carbon rod anode and a platinum sheet cathode into the undivided cell and initiate constant current electrolysis at 8mA.
- Maintain the reaction at 50°C with stirring for 5 hours, then purify the resulting tetra-substituted hydrazine via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical methodology offers substantial strategic benefits beyond mere technical novelty. The elimination of transition metal catalysts directly translates to a reduction in raw material costs, as expensive palladium, copper, or nickel complexes are no longer required. Furthermore, the removal of metal catalysts obviates the need for costly metal scavenging resins and extensive testing for heavy metal residues, which are critical bottlenecks in API intermediate supply chains. The use of simple, commodity-grade electrolytes and solvents ensures a stable and resilient supply base, mitigating risks associated with the sourcing of specialized reagents. Additionally, the mild reaction conditions and inherent safety of electrochemical processes reduce energy consumption and waste disposal costs. This holistic improvement in process efficiency supports a more agile and cost-effective supply chain, capable of responding rapidly to market demands for high-quality hydrazine intermediates.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the dependency on precious metal catalysts and stoichiometric chemical oxidants. This reduction in reagent complexity lowers the direct material costs and simplifies the inventory management of hazardous chemicals. Moreover, the simplified workup procedure reduces solvent consumption and labor hours associated with purification, leading to substantial overall manufacturing savings. The economic model shifts from high-cost specialized reagents to low-cost electricity and bulk chemicals, enhancing the margin potential for commercial production.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key inputs are commodity chemicals with multiple global suppliers, unlike specialized catalysts that may have single-source vulnerabilities. The robustness of the electrochemical method against variations in raw material quality ensures consistent output, reducing the risk of batch failures. This reliability is crucial for maintaining continuous production schedules and meeting strict delivery timelines for downstream pharmaceutical clients. The simplified logistics of handling non-hazardous electrolytes compared to strong oxidants further streamline the supply chain operations.
- Scalability and Environmental Compliance: The technology is inherently scalable, with the patent highlighting compatibility with microchannel reactors for intensified production. This facilitates a smoother transition from lab scale to commercial tonnage without the engineering challenges associated with exothermic traditional oxidation reactions. Environmentally, the generation of hydrogen as the sole byproduct aligns with stringent global emissions standards, reducing the regulatory burden and waste treatment costs. This green profile enhances the corporate sustainability metrics, making the supply chain more attractive to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical N-N coupling technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and feasibility for industrial partners. Understanding these details is essential for evaluating the integration of this method into existing manufacturing workflows. The answers reflect the core innovations in catalyst-free synthesis and process safety that define this new standard in hydrazine production.
Q: Does this electrochemical method require transition metal catalysts?
A: No, the patented process (CN112126942A) operates without transition metal catalysts or external chemical oxidants, utilizing electrons as the primary reagent to drive the N-N coupling.
Q: What are the primary advantages regarding impurity profiles?
A: By avoiding metal catalysis, the process eliminates the risk of heavy metal residue contamination, significantly simplifying downstream purification and ensuring high-purity specifications suitable for pharmaceutical applications.
Q: Is this method scalable for industrial production?
A: Yes, the method utilizes simple undivided cells and commercially available electrolytes, and the patent explicitly mentions compatibility with microchannel reaction devices for improved efficiency and industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetra-substituted Hydrazines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facility is equipped with state-of-the-art electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to leveraging technologies like the one described in CN112126942A to deliver superior quality hydrazine derivatives that meet the evolving needs of the pharmaceutical industry.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us to request specific COA data and route feasibility assessments for your target molecules. Together, we can drive efficiency and innovation in your supply chain, ensuring a reliable source of high-purity intermediates for your critical drug development programs.
