Revolutionizing Pharmaceutical Intermediate Production Through FeCl3-Catalyzed One-Pot Synthesis of High-Purity Naphthoquinocyclopropane Compounds at Commercial Scale
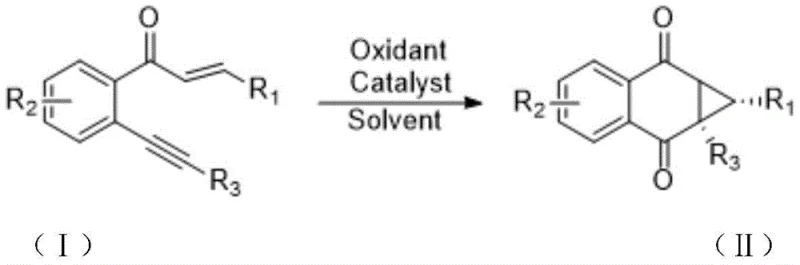
The recently granted Chinese patent CN109896944B represents a significant advancement in the synthesis of structurally complex naphthoquinone derivatives through an innovative catalytic approach that addresses longstanding challenges in fine chemical manufacturing. This methodology provides a sustainable pathway for producing high-value pharmaceutical intermediates by leveraging iron-based catalysis under environmentally benign conditions. The invention specifically targets the critical need for efficient cyclopropanation techniques within medicinal chemistry applications where structural complexity directly correlates with biological activity. By establishing a one-pot transformation from readily accessible o-alkynyl chalcone precursors to functionalized naphthoquinocyclopropane scaffolds, this process delivers both scientific novelty and immediate industrial applicability. The patent demonstrates rigorous experimental validation across multiple substrate variations while maintaining consistent operational parameters that facilitate straightforward technology transfer to manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing naphthoquinone and cyclopropane structures suffer from significant operational constraints that hinder their industrial adoption despite their chemical importance in pharmaceutical applications. Chromium trioxide oxidation methods generate substantial toxic waste streams requiring expensive treatment protocols while operating under highly corrosive acidic conditions that demand specialized equipment and safety measures. Liquid phase oxidation techniques using naphthalene precursors involve energy-intensive processes with poor selectivity leading to complex product mixtures that complicate purification and reduce overall yield efficiency. Conventional cyclopropanation methods such as Freund reactions require hazardous metallic sodium handling under strictly controlled environments due to explosion risks during alkyl halide reactions. Ylide-based cyclopropanation generates considerable stoichiometric waste with low atom economy while carbene approaches suffer from inconsistent stereoselectivity and require expensive transition metal catalysts that necessitate additional removal steps to meet pharmaceutical purity standards. These cumulative limitations create substantial barriers to commercial implementation particularly when scaling production for global pharmaceutical supply chains.
The Novel Approach
The patented methodology overcomes these historical challenges through an elegant iron-catalyzed oxidative cyclization process that operates under remarkably mild conditions while delivering superior environmental performance. By utilizing ferric chloride or ferric nitrate as catalysts in combination with diiodo pentoxide as a green oxidant in standard organic solvents like dioxane at temperatures between 80°C and 120°C, this approach eliminates hazardous reagents and generates minimal waste byproducts. The one-pot reaction sequence completes within thirty minutes to one hour without requiring inert atmosphere or specialized equipment, significantly reducing operational complexity compared to conventional methods. Crucially, the process demonstrates exceptional substrate tolerance across diverse functional groups including halogens and heterocyclic systems while maintaining consistent yield profiles between forty percent and seventy-eight percent across multiple examples. This robustness enables direct adaptation to various pharmaceutical intermediate requirements without extensive process re-engineering.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The catalytic cycle begins with Lewis acid activation of the alkyne moiety by ferric chloride, facilitating nucleophilic attack from the adjacent carbonyl oxygen to form a key oxonium intermediate. This critical step initiates an intramolecular cyclization cascade where the activated alkyne undergoes electrophilic addition followed by oxidative rearomatization mediated by diiodo pentoxide as the terminal oxidant. The iron catalyst serves dual functions by coordinating both the alkyne and carbonyl groups while simultaneously activating the oxidant through redox cycling between Fe(III) and Fe(II) states. This concerted mechanism enables direct formation of the strained cyclopropane ring fused to the quinone system without requiring external radical initiators or high-energy intermediates that typically compromise selectivity in alternative approaches.
Impurity profile management is achieved through precise control of catalyst loading between five percent and thirty percent relative to substrate concentration, which prevents overoxidation side reactions while maintaining optimal reaction kinetics. The solvent system selection—particularly the preference for dioxane—creates an ideal polarity environment that stabilizes reactive intermediates without promoting decomposition pathways observed in more polar media like acetonitrile. Temperature regulation within the eighty to one hundred twenty degree Celsius range ensures sufficient activation energy for cyclization while avoiding thermal degradation that would generate polymeric byproducts. This careful balance of reaction parameters results in consistently high purity profiles across all synthesized derivatives as confirmed by comprehensive NMR characterization data presented in the patent examples.
How to Synthesize Naphthoquinocyclopropane Compounds Efficiently
This patented methodology represents a paradigm shift in naphthoquinone derivative production by enabling direct access to complex cyclopropanated structures through a streamlined catalytic process that eliminates multiple synthetic steps required by conventional approaches. The following standardized procedure provides a reliable framework for manufacturing teams seeking to implement this technology at scale while maintaining strict quality control parameters essential for pharmaceutical applications. Detailed operational guidelines below outline critical process parameters validated through extensive experimental work described in the patent documentation.
- Combine o-alkynyl chalcone compound with ferric chloride catalyst and diiodo pentoxide oxidant in optimal solvent ratios within a pressure-resistant reaction tube.
- Maintain precise thermal control at specified temperature range for defined duration under magnetic stirring to ensure complete cyclization without decomposition.
- Execute purification through silica gel-assisted solvent removal followed by column chromatography using petroleum ether/ethyl acetate eluent to isolate target compounds.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate procurement by transforming traditionally complex manufacturing processes into streamlined operations that enhance supply chain resilience while reducing total cost of ownership. The elimination of hazardous reagents and multi-step sequences creates immediate opportunities for cost optimization without compromising product quality or regulatory compliance requirements essential for global pharmaceutical partners.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts and associated purification steps significantly reduces raw material expenses while eliminating costly waste treatment procedures required for heavy metal contamination control. The simplified one-pot process architecture decreases capital investment needs by avoiding specialized equipment typically required for cryogenic operations or high-pressure reactions found in conventional cyclopropanation methods.
- Enhanced Supply Chain Reliability: Utilization of commercially available starting materials with broad supplier options minimizes single-source dependencies while the robust reaction profile maintains consistent output quality across varying batch sizes. The short reaction time and ambient pressure operation enable flexible production scheduling that accommodates urgent demand fluctuations without requiring extensive revalidation protocols.
- Scalability and Environmental Compliance: The process demonstrates seamless scalability from laboratory validation to commercial production volumes due to its straightforward thermal management requirements and absence of sensitive reaction parameters that typically complicate scale-up operations. The environmentally benign profile eliminates regulatory hurdles associated with hazardous waste disposal while supporting corporate sustainability initiatives through reduced energy consumption and minimal solvent usage.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of patent CN109896944B's experimental data and implementation requirements. These insights reflect practical experience from similar catalytic process deployments within our global manufacturing network.
Q: How does this catalytic method eliminate environmental hazards associated with traditional naphthoquinone synthesis?
A: The FeCl3-catalyzed process operates under mild conditions without generating hazardous waste streams or requiring toxic heavy metal oxidants. By utilizing diiodo pentoxide as a clean oxidant and avoiding chromium-based systems that produce persistent pollutants, this methodology achieves inherent environmental safety while maintaining high reaction efficiency across diverse substrates.
Q: What substrate flexibility enables this process to support complex pharmaceutical intermediate manufacturing?
A: The reaction demonstrates exceptional tolerance for various functional groups including halogens, alkyl chains, and heterocyclic moieties as evidenced by successful synthesis across fifteen derivative examples. This broad substrate scope allows customization of naphthoquinocyclopropane structures while maintaining consistent yield profiles through the one-pot cyclization mechanism.
Q: How does the one-step cyclization improve commercial scalability compared to conventional cyclopropanation techniques?
A: The direct oxidative cyclization eliminates multi-step sequences required in traditional methods like Freund reactions or ylide cyclopropanation. This simplification reduces intermediate handling risks and equipment requirements while enabling seamless transition from laboratory-scale to industrial production volumes through standardized process parameters.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthoquinocyclopropane Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global pharmaceutical clients through rigorous QC labs equipped with advanced analytical capabilities. We have successfully implemented similar iron-catalyzed processes across multiple therapeutic compound classes, demonstrating our ability to translate patented methodologies into reliable manufacturing operations that meet demanding regulatory standards worldwide. Our technical team specializes in optimizing catalytic processes for maximum efficiency while ensuring consistent product quality through comprehensive process analytical technology monitoring.
Leverage our expertise through a Customized Cost-Saving Analysis tailored to your specific production requirements by contacting our technical procurement team today to request detailed COA data and route feasibility assessments for your next-generation pharmaceutical intermediate needs.
