Revolutionizing Pyrrole Derivative Synthesis: Scalable, Eco-Friendly Production for Pharmaceutical Applications
The patent CN110804007A introduces a groundbreaking methodology for synthesizing polysubstituted pyrrole derivatives, representing a significant advancement in heterocyclic chemistry with profound implications for pharmaceutical manufacturing. This innovative approach utilizes diazo compounds or phenylhydrazone compounds reacting with amino-substituted unsaturated carbonyl compounds through a sophisticated cascade mechanism involving carbon-carbon insertion, cyclization, and [1,5] migration reactions. The process achieves high efficiency and yield while operating under remarkably mild conditions, addressing long-standing challenges in pyrrole ring construction that have constrained pharmaceutical development pipelines. This patent represents a strategic breakthrough that aligns perfectly with industry demands for sustainable, scalable synthesis routes for complex heterocyclic intermediates essential in modern drug discovery and development programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing polysubstituted pyrroles have been plagued by multiple limitations that hinder their industrial applicability. Conventional methods such as Knorr synthesis, Paal-Knorr synthesis, and Hantzsch synthesis often require harsh reaction conditions including high temperatures and strong acids or bases, which can lead to decomposition of sensitive functional groups and reduced product yields. Transition metal-catalyzed cyclization and multi-component coupling reactions ([4+1], [3+2], [2+2+1] additions) frequently employ expensive and toxic catalysts that necessitate complex removal procedures, significantly increasing production costs and environmental impact. The preparation of starting materials for these conventional routes is often complicated and multi-step, resulting in extended production timelines and inconsistent quality. Furthermore, these methods typically suffer from poor regioselectivity and generate difficult-to-remove impurities that compromise the purity required for pharmaceutical applications, creating substantial barriers to commercial scale-up and regulatory approval.
The Novel Approach
The patented methodology overcomes these limitations through an elegant cascade reaction sequence that begins with readily accessible starting materials. By utilizing diazo compounds or phenylhydrazone compounds reacting with amino-substituted unsaturated carbonyl compounds, the process achieves pyrrole ring construction through a series of well-defined steps including carbon-carbon insertion, cyclization, and [1,5] migration reactions. This approach operates under mild conditions (0-60°C) without requiring extreme pH environments, preserving sensitive functional groups while delivering high yields of the target compounds. The metal catalysts employed (such as palladium acetate or silver trifluoromethanesulfonate at only 3-10 mol%) are both effective and relatively inexpensive compared to traditional transition metal catalysts. Crucially, the reaction pathway minimizes side product formation through its inherent selectivity, resulting in cleaner reactions that simplify downstream purification and enhance overall process efficiency for pharmaceutical manufacturing applications.
Mechanistic Insights into Diazo-Mediated Pyrrole Ring Construction
The core innovation lies in the sophisticated cascade mechanism that enables efficient pyrrole ring formation. The process begins with the reaction between an amino-substituted unsaturated carbonyl compound (compound I) and a diazo compound (compound II), facilitated by metal catalysts such as palladium acetate or silver trifluoromethanesulfonate. The initial step involves carbon-carbon insertion where the diazo compound inserts into the carbon-nitrogen bond of the enone system, forming a key intermediate that subsequently undergoes intramolecular cyclization. This cyclization step is followed by a [1,5] migration process that rearranges the molecular structure to form the stable pyrrole ring system with precise regiocontrol. The metal catalysts play a critical role in facilitating the carbene transfer from the diazo compound while maintaining the mild reaction conditions necessary for preserving sensitive functional groups throughout the transformation.
The mechanism inherently promotes high purity through its selective transformation pathway that minimizes competing side reactions. The cascade nature of the reaction ensures that intermediates are rapidly converted to the final product, reducing opportunities for decomposition or side product formation. The mild temperature range (0-60°C) prevents thermal degradation pathways that commonly plague traditional pyrrole syntheses requiring higher temperatures. Additionally, the use of environmentally benign solvents such as dichloromethane or toluene combined with straightforward purification protocols (column chromatography with petroleum ether/ethyl acetate) enables efficient removal of catalyst residues and minor impurities. This inherent selectivity and clean reaction profile directly translates to superior product purity without requiring additional purification steps that would increase production costs and time.
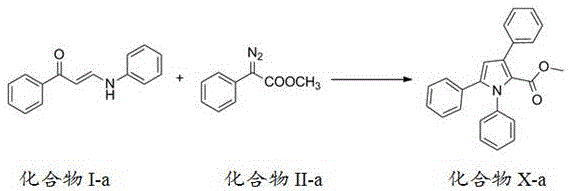
How to Synthesize Polysubstituted Pyrrole Derivatives Efficiently
This patented methodology provides a robust pathway for producing high-purity polysubstituted pyrrole derivatives suitable for pharmaceutical applications. The process begins with the preparation of key intermediates through straightforward synthetic routes using commercially available starting materials. The core transformation leverages a cascade reaction mechanism that efficiently constructs the pyrrole ring system with excellent regiocontrol and high yield. Detailed standardized synthesis steps for implementing this technology in manufacturing environments are outlined in the following section.
- Prepare compound I by reacting compound IV with N,N-dimethylformamide methylal at 80-150°C for 6-18 hours, followed by reaction with compound VI at the same temperature range for 12-24 hours, with purification through distillation and recrystallization.
- Prepare compound II by reacting compound VII with 4-acetamidobenzenesulfonyl azide at -10 to 5°C for 4-10 hours using DBU as catalyst, followed by extraction, rotary evaporation, and column chromatography purification.
- Conduct the cyclization reaction by adding compound II to a mixture of compound I and metal catalyst (3-10 mol%) in dichloromethane at room temperature over 12 hours, followed by extraction, washing, drying, and purification through column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points faced by procurement and supply chain teams in pharmaceutical manufacturing organizations. By eliminating complex multi-step sequences and harsh reaction conditions found in traditional approaches, the process delivers substantial operational improvements that enhance supply chain resilience while reducing overall production costs. The strategic advantages of this methodology extend beyond mere chemical efficiency to encompass broader business benefits that strengthen competitive positioning in the pharmaceutical intermediates market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and high-temperature processing requirements significantly reduces raw material costs while minimizing energy consumption during production. The use of readily available starting materials that can be obtained through simple reactions eliminates dependency on specialized or constrained supply chains, creating substantial cost savings through simplified sourcing and reduced processing complexity.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available precursors ensures consistent material availability regardless of market fluctuations. The simplified reaction sequence with fewer processing steps reduces production time variability while maintaining high product quality across different scales. This process robustness translates to predictable lead times and reliable delivery schedules that strengthen supply chain continuity for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions (0-60°C) and environmentally friendly solvent systems enable seamless scale-up from laboratory to commercial production without requiring specialized equipment modifications. The reduced generation of hazardous waste streams compared to traditional methods significantly lowers environmental impact while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding the implementation and benefits of this patented technology for pharmaceutical manufacturing applications. These answers are based on detailed analysis of the patent's technical specifications and demonstrated performance characteristics.
Q: How does this synthesis method improve purity compared to traditional approaches?
A: The cascade reaction mechanism involving carbon-carbon insertion, cyclization, and [1,5] migration creates a highly selective pathway that minimizes side products. The mild reaction conditions (0-60°C) prevent thermal degradation that commonly causes impurities in conventional high-temperature pyrrole syntheses.
Q: What makes this process suitable for large-scale pharmaceutical manufacturing?
A: The process utilizes readily available starting materials that can be obtained through simple reactions, operates under mild and environmentally friendly conditions, and employs catalysts that are easily removable. The demonstrated scalability from laboratory to commercial production (100 kgs to 100 MT) ensures consistent quality at scale.
Q: How does this method address supply chain concerns for pharmaceutical intermediates?
A: The synthesis relies on stable, commercially available precursors that are not subject to supply constraints. The simplified reaction sequence with fewer processing steps reduces production time variability, while the robust purification protocol ensures consistent quality across batches regardless of scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrrole Derivatives Supplier
Our company stands at the forefront of advanced heterocyclic chemistry manufacturing, offering unparalleled expertise in producing complex pyrrole-based intermediates with exceptional purity and consistency. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs. Our commitment to quality assurance ensures that every batch meets or exceeds the rigorous requirements of global pharmaceutical regulatory bodies, providing our partners with complete confidence in their supply chain continuity.
We invite you to leverage our technical expertise through a Customized Cost-Saving Analysis tailored to your specific manufacturing needs. Contact our technical procurement team today to request detailed COA data and route feasibility assessments that will demonstrate how our patented methodology can enhance your production efficiency while ensuring reliable supply of high-purity polysubstituted pyrrole derivatives for your pharmaceutical development programs.
