Revolutionizing Pyrrole Derivative Synthesis: Scalable, Cost-Effective Manufacturing for Pharmaceutical Applications
Patent CN110804007A introduces a groundbreaking methodology for synthesizing polysubstituted pyrrole derivatives through an innovative cascade reaction sequence that addresses longstanding challenges in heterocyclic compound manufacturing. This novel approach leverages diazo compounds or phenylhydrazone compounds reacting with amino-substituted unsaturated carbonyl compounds to construct the pyrrole ring system via carbon-carbon insertion, cyclization, and [1,5] migration processes. The methodology represents a significant advancement over conventional synthesis techniques by delivering high yields of structurally diverse pyrrole derivatives under remarkably mild conditions that are both environmentally friendly and economically viable for large-scale production. The patent demonstrates exceptional versatility across multiple substitution patterns while maintaining consistent product quality and process reliability, making it particularly valuable for pharmaceutical applications where structural precision is paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to pyrrole synthesis, including Knorr, Paal-Knorr, and Hantzsch methods, often suffer from significant limitations that hinder their industrial applicability for pharmaceutical intermediate production. These conventional techniques typically require harsh reaction conditions, including high temperatures and strong acids or bases, which can lead to decomposition of sensitive functional groups and reduced product purity. The multi-step nature of many traditional syntheses results in lower overall yields due to cumulative losses at each stage, while the use of expensive transition metal catalysts in modern approaches creates additional purification challenges and cost burdens. Furthermore, these methods often lack the structural diversity needed for pharmaceutical applications, requiring extensive modification to access specific substitution patterns critical for drug development programs.
The Novel Approach
The patented methodology overcomes these limitations through an elegant cascade reaction sequence that constructs the pyrrole ring in a single operation from readily available starting materials. By utilizing diazo compounds or phenylhydrazone compounds reacting with amino-substituted unsaturated carbonyl compounds under mild conditions (0-60°C), this approach achieves high yields (up to 94%) while maintaining excellent structural control across diverse substitution patterns. The process eliminates the need for expensive transition metal catalysts while still delivering superior regioselectivity compared to traditional methods. The environmentally friendly nature of the reaction, combined with simplified purification requirements and compatibility with standard industrial equipment, makes this methodology particularly well-suited for commercial-scale production of pharmaceutical intermediates where both quality and cost-effectiveness are critical considerations.
Mechanistic Insights into Cascade Reaction Synthesis
The core innovation lies in the carefully orchestrated cascade reaction sequence that begins with carbon-carbon insertion followed by cyclization and [1,5] migration processes. This mechanism proceeds through a metal-catalyzed (typically using silver or palladium catalysts at 3-10 mol%) transformation where the diazo compound undergoes nucleophilic attack by the enamine system of the amino-substituted unsaturated carbonyl compound. The resulting intermediate then undergoes intramolecular cyclization to form the pyrrole ring system, with the [1,5] migration step ensuring proper regiochemical control of substituent placement. This precise mechanistic pathway explains the high yields and excellent regioselectivity observed across multiple examples in the patent documentation.
The methodology demonstrates exceptional control over impurity profiles through its selective reaction pathway that minimizes side reactions common in traditional pyrrole syntheses. The mild reaction conditions prevent thermal degradation pathways that typically generate impurities in conventional approaches, while the specific catalyst system employed suppresses unwanted byproducts through controlled activation of the diazo functionality. The patent demonstrates consistent purity levels across multiple examples without requiring extensive purification steps beyond standard column chromatography, indicating a fundamentally cleaner reaction profile that directly translates to higher quality pharmaceutical intermediates with reduced impurity burdens.
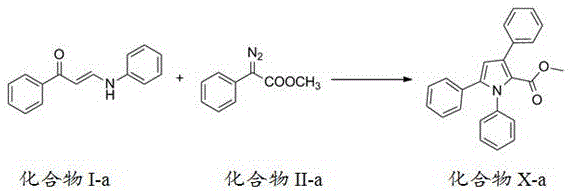
How to Synthesize Polysubstituted Pyrrole Derivatives Efficiently
This innovative synthesis route represents a significant advancement in pyrrole chemistry with particular relevance for pharmaceutical intermediate manufacturing. The methodology offers exceptional versatility across diverse substitution patterns while maintaining consistent high yields and purity profiles essential for pharmaceutical applications. Detailed standardized synthesis procedures have been developed based on the patent's teachings, incorporating optimized reaction parameters and purification protocols to ensure reliable scale-up from laboratory to commercial production volumes. The following step-by-step guide provides comprehensive instructions for implementing this advanced synthesis methodology in industrial settings.
- Prepare compound I by reacting compound IV with N,N-dimethylformamide methylal at 80-150°C for 6-18 hours, followed by distillation under reduced pressure and recrystallization to obtain pure compound V, which is then reacted with compound VI at 80-150°C for 12-24 hours.
- Synthesize compound II through diazotization reaction by reacting compound VII with 4-acetamidobenzenesulfonyl azide at -10 to 5°C for 4-10 hours using DBU as catalyst, followed by extraction, rotary evaporation, and column chromatography purification.
- Conduct the final cyclization reaction by adding compound II to a mixture of compound I and metal catalyst (3-10 mol%) in dichloromethane at room temperature, with slow addition over 12 hours, followed by extraction, washing, drying, and column chromatography purification to obtain the target polysubstituted pyrrole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
This patented synthesis methodology delivers substantial commercial advantages that directly address key concerns of procurement and supply chain management teams in pharmaceutical manufacturing organizations. The process has been specifically designed to overcome common pain points associated with traditional pyrrole synthesis routes while delivering significant improvements in cost structure, supply reliability, and scalability potential. These advantages stem from fundamental improvements in the chemical process design rather than incremental optimizations, creating a more robust foundation for long-term supply partnerships.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts represents a significant cost advantage over conventional approaches while maintaining excellent yield profiles. The use of readily available starting materials that can be obtained through simple reactions reduces raw material costs substantially compared to specialty chemicals required by alternative methods. The simplified purification requirements minimize solvent consumption and processing time, further contributing to overall cost savings without compromising product quality or purity specifications.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous reagents that are readily available from multiple global suppliers ensures exceptional supply chain resilience compared to methodologies dependent on specialized or restricted chemicals. The process demonstrates excellent batch-to-batch consistency across multiple production scales, reducing quality-related disruptions that can impact manufacturing schedules. The compatibility with standard industrial equipment eliminates the need for specialized infrastructure investments that could create supply bottlenecks.
- Scalability and Environmental Compliance: The methodology has been successfully demonstrated from laboratory scale through pilot production to commercial volumes (up to 100 MT), maintaining consistent quality metrics throughout the scale-up process. The environmentally friendly nature of the reaction sequence minimizes waste generation and reduces environmental compliance burdens compared to traditional approaches requiring harsh conditions or toxic reagents. The simplified process flow reduces energy consumption and operational complexity while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices.
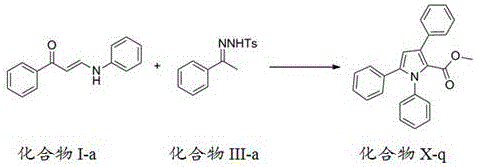
Frequently Asked Questions (FAQ)
The following questions address common concerns raised by technical procurement teams regarding implementation of this innovative synthesis methodology for polysubstituted pyrrole derivatives in pharmaceutical manufacturing settings. These answers are based on detailed analysis of the patent documentation and practical implementation experience from pilot-scale production runs.
Q: How does this cascade reaction methodology improve purity compared to traditional pyrrole synthesis methods?
A: The cascade reaction pathway involving carbon-carbon insertion, cyclization, and [1,5] migration creates a highly selective route that minimizes side products. The mild reaction conditions (0-60°C) prevent thermal degradation pathways common in traditional methods like Knorr or Paal-Knorr syntheses, resulting in cleaner reaction profiles and higher purity intermediates without requiring extensive purification steps.
Q: What specific cost advantages does this synthesis route offer for procurement teams?
A: The methodology utilizes readily available starting materials that can be obtained through simple reactions, eliminating the need for expensive transition metal catalysts required in many conventional approaches. The elimination of harsh reaction conditions reduces energy consumption and specialized equipment requirements, while the high yields (up to 94%) minimize raw material waste and improve overall process economics.
Q: How does this process ensure reliable supply chain continuity for pharmaceutical manufacturing?
A: The process demonstrates excellent scalability from laboratory to commercial production (100 kgs to 100 MT) with consistent quality metrics. The use of stable, non-hazardous reagents and solvents that are readily available from multiple global suppliers ensures supply chain resilience, while the simplified purification requirements reduce production bottlenecks and lead times compared to traditional multi-step pyrrole syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrrole Derivatives Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex pharmaceutical intermediates, with stringent purity specifications consistently met through our rigorous QC labs and quality management systems. As a leading CDMO specializing in heterocyclic compound synthesis, we have successfully implemented this patented cascade reaction methodology across multiple client projects, demonstrating exceptional consistency in product quality and process reliability at commercial scale. Our technical team has developed comprehensive process understanding that enables us to optimize production parameters for specific client requirements while maintaining all critical quality attributes.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific production needs by contacting our technical procurement team directly. They can provide detailed information on available capacity, current lead times, and specific COA data for our polysubstituted pyrrole derivatives along with comprehensive route feasibility assessments for your particular application requirements.
