Advanced Palladium-Catalyzed Carbonylation for Scalable 2-Trifluoromethyl Imidazole Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 2-Trifluoromethyl Imidazole Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access complex heterocyclic scaffolds that offer enhanced biological profiles. A significant breakthrough in this domain is detailed in patent CN111423381B, which discloses a highly efficient preparation method for 2-trifluoromethyl substituted imidazole compounds. These nitrogen-containing five-membered heterocycles are pivotal structural motifs found in numerous bioactive molecules, serving as key intermediates for active pharmaceutical ingredients (APIs) and functional materials. The introduction of the trifluoromethyl group is particularly strategic, as it imparts superior metabolic stability and lipophilicity to the molecular backbone. This patent outlines a transition metal palladium-catalyzed carbonylation series reaction that operates under remarkably mild conditions, utilizing cheap and easily obtainable starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts. For R&D directors and procurement specialists alike, this represents a paradigm shift towards more sustainable and cost-effective manufacturing of high-value heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with significant challenges that hinder large-scale adoption. Traditional literature methods often rely on the direct reaction of a synthon containing a trifluoromethyl group with a suitable substrate, frequently employing reagents like trifluorodiazoethane. While effective in specific contexts, these synthons are often unstable, hazardous to handle, and not widely available commercially, leading to supply chain bottlenecks. Furthermore, many existing protocols require harsh reaction conditions, including elevated temperatures or the use of high-pressure carbon monoxide gas, which necessitates specialized equipment and rigorous safety protocols. The limited substrate compatibility of these older methods often results in poor yields when dealing with diverse functional groups, forcing chemists to employ lengthy protection-deprotection sequences. Consequently, the cost of goods sold (COGS) for trifluoromethylated imidazoles remains prohibitively high for many applications, limiting their utility in early-stage drug discovery and commercial manufacturing.
The Novel Approach
The methodology described in the patent data offers a transformative solution by leveraging a three-component coupling reaction catalyzed by palladium. This novel approach utilizes trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts as the foundational building blocks, all of which are relatively inexpensive and commercially accessible. The reaction proceeds efficiently at a mild temperature of 30°C, drastically reducing energy consumption compared to traditional high-temperature protocols. Crucially, the system employs formic acid and acetic anhydride as a carbon monoxide alternative, eliminating the need for handling toxic, high-pressure CO gas cylinders, thereby enhancing operational safety and simplifying reactor requirements. The scope of this reaction is exceptionally broad, accommodating various substituents on the aryl rings, including methyl, tert-butyl, halogens, and nitro groups, as demonstrated by the successful synthesis of diverse derivatives.
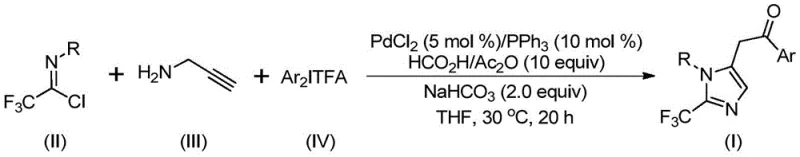
The versatility of this synthetic route is further evidenced by the successful preparation of compounds I-1 through I-5, showcasing excellent tolerance for different electronic and steric environments. This flexibility allows medicinal chemists to rapidly generate libraries of analogs for structure-activity relationship (SAR) studies without being constrained by synthetic feasibility. By streamlining the synthesis into a single pot with high atom economy, this method significantly reduces waste generation and purification complexity. For a reliable pharmaceutical intermediate supplier, adopting such a streamlined process translates directly into improved throughput and reduced lead times for custom synthesis projects. The ability to tune the R and Ar groups independently provides a powerful tool for designing molecules with precise physicochemical properties, meeting the stringent demands of modern drug development.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
Understanding the mechanistic underpinnings of this transformation is essential for R&D teams aiming to optimize the process for specific substrates. The reaction initiates with the formation of an intermolecular carbon-nitrogen bond promoted by the alkali base, yielding a trifluoroacetamidine intermediate. This species then undergoes isomerization, setting the stage for the palladium catalytic cycle. The palladium catalyst, typically palladium chloride coordinated with triphenylphosphine, facilitates the palladation of the alkyne moiety in the propargylamine component, generating a key alkenyl palladium intermediate. Subsequent isomerization converts this into an alkyl palladium species, which is primed for the carbonylation step. Uniquely, the carbon monoxide required for this insertion is generated in situ from the decomposition of formic acid in the presence of acetic anhydride, ensuring a steady, low-concentration supply of CO that drives the reaction forward without the risks associated with external gas feeding.
Following carbonylation, an acyl palladium intermediate is formed, which then undergoes oxidative addition with the diaryl iodonium salt. This step generates a high-valent tetravalent palladium intermediate, a critical juncture that determines the efficiency of the aryl group incorporation. The cycle concludes with a reductive elimination step, releasing the final 2-trifluoromethyl-substituted imidazole product and regenerating the active palladium catalyst. From an impurity control perspective, the mild reaction temperature of 30°C plays a vital role in suppressing side reactions such as polymerization of the alkyne or decomposition of the sensitive imidoyl chloride. The use of sodium bicarbonate as a mild base further ensures that acid-sensitive functional groups on the substrate remain intact. This mechanistic elegance ensures that the crude reaction mixture is relatively clean, simplifying downstream purification and enhancing the overall purity profile of the final API intermediate.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and mixing protocols to maximize yield and reproducibility. The patent specifies a molar ratio of palladium chloride to triphenylphosphine to sodium bicarbonate of approximately 0.05:0.1:2, ensuring sufficient catalytic activity and neutralization of acidic byproducts. The reaction is typically conducted in tetrahydrofuran (THF), which effectively dissolves all components and promotes the catalytic cycle, although other aprotic solvents like acetonitrile or dioxane can also be employed. The detailed standardized synthesis steps below outline the precise procedure for achieving high conversion rates, guiding process chemists from raw material charging to final isolation. Adhering to these parameters allows for the consistent production of high-purity material suitable for subsequent biological evaluation or formulation.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, formic acid, trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt in an organic solvent such as THF.
- Stir the reaction mixture at a mild temperature of 30°C for 16 to 24 hours to allow the carbonylation and cyclization to proceed efficiently.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity 2-trifluoromethyl substituted imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic and logistical implications of this patented method are profound. The shift from hazardous, specialty reagents to commodity chemicals fundamentally alters the cost structure of producing trifluoromethylated imidazoles. By utilizing starting materials that are widely available in the global chemical market, manufacturers can mitigate the risk of supply disruptions that often plague niche synthetic routes. The elimination of high-pressure gas infrastructure not only reduces capital expenditure (CAPEX) for new production lines but also lowers the operational overhead associated with safety compliance and maintenance. Furthermore, the simplicity of the post-treatment process, which involves basic filtration and standard column chromatography, reduces the demand for specialized purification equipment and skilled labor hours. These factors collectively contribute to a more resilient and agile supply chain capable of responding quickly to fluctuating market demands.
- Cost Reduction in Manufacturing: The utilization of inexpensive catalysts like palladium chloride and readily available organic salts drastically lowers the raw material costs compared to methods requiring precious metal complexes or exotic fluorinating agents. The avoidance of high-pressure carbon monoxide cylinders removes a significant safety and logistics cost burden, while the mild reaction temperature minimizes energy consumption for heating and cooling. Additionally, the high reaction efficiency and yield reported in the examples mean less raw material is wasted, directly improving the mass balance and reducing the cost per kilogram of the final product. This economic efficiency makes the commercial scale-up of complex pharmaceutical intermediates much more viable for mid-sized manufacturers.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical metric for any supply chain负责人,and this method excels by relying on bench-stable solids and liquids rather than gases or cryogenic fluids. Trifluoroethylimidoyl chloride and diaryl iodonium salts can be stockpiled safely, allowing manufacturers to build inventory buffers against market volatility. The robustness of the reaction conditions means that production is less susceptible to minor variations in environmental controls, ensuring consistent batch-to-bquality. This reliability translates into shorter lead times for customers, as production schedules can be optimized without the delays associated with complex safety checks or specialized reagent procurement. It positions the manufacturer as a dependable partner for long-term API supply agreements.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated effectively at the gram level with clear pathways to kilogram and tonne scales. The use of formic acid as a CO surrogate aligns with green chemistry principles by avoiding the direct use of toxic gases, thereby simplifying environmental permitting and waste treatment processes. The solvent system, primarily THF, is well-understood in industrial settings with established recovery and recycling protocols, minimizing the environmental footprint of the manufacturing process. This alignment with sustainability goals is increasingly important for multinational corporations seeking to reduce the carbon footprint of their supply chains, making this technology attractive for ESG-focused procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and procedural details outlined in the patent documentation, providing clarity for technical teams evaluating this route for their own pipelines. Understanding these nuances helps in making informed decisions about process adoption and vendor selection.
Q: What are the advantages of introducing a trifluoromethyl group into imidazole compounds?
A: Introducing a trifluoromethyl group significantly improves the electronegativity, bioavailability, metabolic stability, and lipophilicity of the parent molecule, making these compounds highly valuable in drug discovery and functional material development.
Q: How does this novel method improve upon conventional synthesis routes?
A: Unlike conventional methods that may require harsh conditions or unstable synthons like trifluorodiazoethane, this method utilizes cheap, readily available starting materials and operates under mild conditions (30°C) with high reaction efficiency and broad substrate compatibility.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It uses inexpensive catalysts and reagents, avoids high-pressure carbon monoxide gas by using formic acid/acetic anhydride as a CO source, and features a simple post-treatment procedure, facilitating expansion from gram to industrial scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into robust manufacturing processes. We are committed to delivering high-purity 2-trifluoromethyl imidazole intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex fluorination chemistries safely and efficiently, adhering to the highest international standards for quality and safety.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your overall project costs. Let us be your partner in turning complex chemical challenges into commercial successes.
