Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activities. Patent CN112239456B introduces a robust methodology for the preparation of substituted 2,3-dihydroquinolone compounds, a privileged structure found in numerous bioactive molecules ranging from antitumor agents to analgesics. As illustrated in the structural diversity of known bioactive agents, the 2,3-dihydroquinolone core is essential for interacting with various biological targets, necessitating reliable and scalable access to these intermediates for drug discovery pipelines. This patent discloses a transition metal palladium-catalyzed carbonylation reaction that utilizes N-pyridine sulfonyl-o-iodoaniline and olefins as key starting materials, offering a significant advancement over traditional synthetic approaches by employing a solid carbon monoxide substitute.
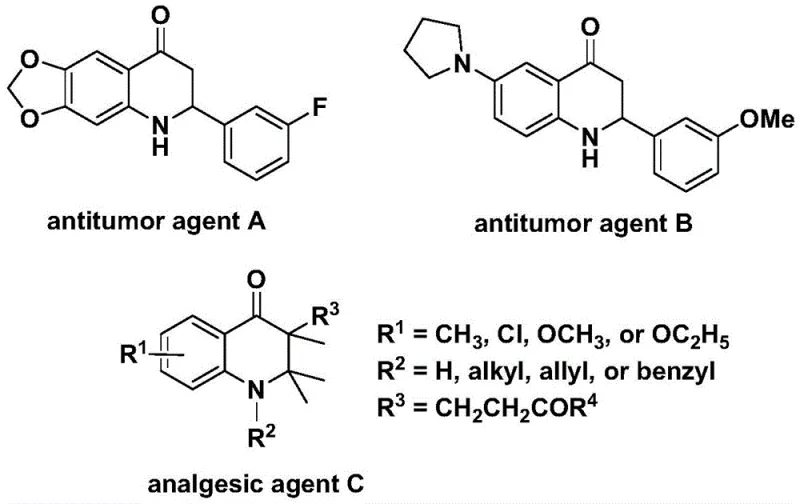
The strategic importance of this technology lies in its ability to construct the carbonyl-containing six-membered heterocyclic ring directly and efficiently. Unlike conventional methods that may rely on hazardous gaseous carbon monoxide or multi-step sequences with poor atom economy, this invention leverages a carbonylative cyclization strategy that is both operationally simple and highly adaptable. For R&D directors overseeing process development, the ability to access diverse 2-aryl and 3-alkyl substituted variants through simple substrate design represents a critical capability for rapid structure-activity relationship (SAR) studies. Furthermore, the method's compatibility with gram-level expansion suggests a clear pathway toward industrial application, addressing the growing demand for high-purity pharmaceutical intermediates in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-dihydroquinolone skeletons has faced significant hurdles regarding safety, efficiency, and substrate tolerance. Traditional carbonylation reactions often require the use of pressurized carbon monoxide gas, which poses severe safety risks due to its high toxicity and flammability, necessitating specialized high-pressure reactors and rigorous safety protocols that increase capital expenditure and operational complexity. Additionally, many existing synthetic routes suffer from limited substrate scope, failing to accommodate sensitive functional groups or sterically hindered olefins, which restricts the chemical space available for medicinal chemists to explore. The reliance on harsh reaction conditions can also lead to the formation of complex impurity profiles, complicating downstream purification and reducing overall process yield, which is a major concern for procurement teams focused on cost-effective manufacturing.
The Novel Approach
The methodology described in patent CN112239456B overcomes these challenges by introducing a mild and safe carbonylation protocol using 1,3,5-trimesic acid phenol ester (TFBen) as a solid carbon monoxide surrogate. This innovation eliminates the need for handling toxic CO gas, allowing the reaction to proceed under standard atmospheric pressure conditions in common organic solvents like 1,4-dioxane. The use of a palladium catalyst system comprising Pd(acac)2 and dppp ligand ensures high catalytic activity and selectivity, enabling the efficient coupling of N-pyridine sulfonyl-o-iodoaniline with a wide array of olefins. This approach not only enhances operational safety but also simplifies the reaction setup, making it accessible for both laboratory-scale optimization and large-scale commercial production without the need for specialized high-pressure infrastructure.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The reaction mechanism involves a sophisticated catalytic cycle initiated by the oxidative addition of the palladium catalyst into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This step generates a reactive aryl-palladium intermediate, which subsequently undergoes migratory insertion of carbon monoxide released in situ from the decomposition of the TFBen additive. The resulting acyl-palladium species is then intercepted by the coordinated olefin, leading to the formation of a palladium-alkyl intermediate through alkene insertion. Finally, a reductive elimination step closes the catalytic cycle, releasing the substituted 2,3-dihydroquinolone product and regenerating the active palladium species for further turnover. This mechanistic pathway ensures high regioselectivity and minimizes side reactions, which is crucial for maintaining high purity standards required in pharmaceutical manufacturing.
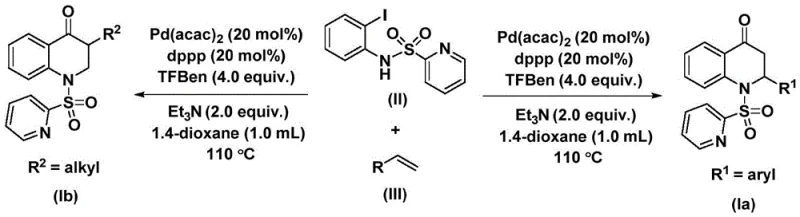
Impurity control is inherently managed through the precise tuning of reaction parameters and the selection of high-quality reagents. The use of triethylamine as an additive helps to neutralize acidic byproducts and stabilize the catalytic species, preventing catalyst deactivation and the formation of palladium black. Furthermore, the specific choice of the dppp ligand creates an optimal steric and electronic environment around the palladium center, favoring the desired carbonylative cyclization over competing pathways such as direct Heck coupling or homocoupling of the aryl iodide. This level of mechanistic control allows for the synthesis of complex derivatives, including those with silyl groups or halogen substituents, with minimal formation of difficult-to-remove impurities, thereby streamlining the purification process and improving overall process mass intensity.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The synthesis protocol outlined in the patent provides a straightforward procedure for accessing these valuable heterocycles. By combining the iodoaniline derivative, the chosen olefin, the palladium catalyst, ligand, and the solid CO source in a suitable solvent, chemists can achieve high conversions under thermal conditions. The reaction typically proceeds at temperatures around 110 °C for a duration of 24 to 48 hours, depending on the specific electronic and steric nature of the olefin substrate. Detailed standardized synthesis steps for implementing this protocol in a GMP environment are provided below.
- Combine palladium bis(acetylacetonate), dppp ligand, triethylamine, and the solid carbon monoxide substitute (TFBen) with the starting materials in an organic solvent like dioxane.
- Heat the reaction mixture to 110 °C and maintain stirring for 24 to 48 hours to ensure complete conversion of the iodoaniline and olefin substrates.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 2,3-dihydroquinolone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits in terms of cost stability and supply reliability. The elimination of high-pressure gas handling equipment significantly reduces the capital investment required for manufacturing facilities, while the use of commercially available starting materials ensures a stable supply chain不受 geopolitical fluctuations affecting specialty gases. The simplicity of the post-treatment process, involving filtration and standard column chromatography, reduces labor costs and processing time, contributing to a more lean and efficient production workflow. Moreover, the high substrate compatibility means that a single platform technology can be used to produce a wide library of intermediates, maximizing asset utilization and reducing the need for multiple dedicated production lines.
- Cost Reduction in Manufacturing: The replacement of toxic carbon monoxide gas with a solid surrogate drastically simplifies the engineering controls required for the reaction, leading to substantial cost savings in facility maintenance and safety compliance. By avoiding the need for specialized high-pressure autoclaves, manufacturers can utilize standard glass-lined or stainless steel reactors, which lowers the barrier to entry for production and reduces the overall cost of goods sold (COGS). Additionally, the high reaction efficiency and yield minimize raw material waste, further enhancing the economic viability of the process for large-scale API intermediate manufacturing.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable solid reagents like TFBen and commercially sourced olefins mitigates the risks associated with the logistics of hazardous gases. This stability ensures consistent production schedules and reduces the likelihood of supply disruptions caused by transportation restrictions on dangerous goods. The robustness of the catalytic system also means that the process is less sensitive to minor variations in raw material quality, providing a more predictable and reliable output for downstream customers requiring just-in-time delivery of critical pharmaceutical building blocks.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic gas emissions align perfectly with modern green chemistry principles and stringent environmental regulations. Scaling this process from gram to kilogram or tonne levels does not introduce significant new safety hazards, facilitating a smoother technology transfer from R&D to commercial production. The simplified waste stream, devoid of high-pressure gas residues, makes disposal and treatment more straightforward, helping companies meet their sustainability goals and reduce the environmental footprint of their chemical manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What are the safety advantages of this carbonylation method compared to traditional CO gas methods?
A: This protocol utilizes 1,3,5-trimesic acid phenol ester (TFBen) as a solid carbon monoxide substitute, eliminating the need for handling toxic and high-pressure carbon monoxide gas, thereby significantly enhancing operational safety and simplifying equipment requirements for scale-up.
Q: What is the substrate scope for the olefin component in this synthesis?
A: The method demonstrates broad compatibility, successfully accommodating both substituted aryl olefins (including those with methyl, chloro, fluoro, and tert-butyl groups) and various alkyl olefins such as linear, branched, and silyl-substituted alkenes, yielding products with good to excellent efficiency.
Q: How does the choice of catalyst and ligand impact the reaction efficiency?
A: The use of palladium bis(acetylacetonate) paired with 1,3-bis(diphenylphosphino)propane (dppp) has been optimized to provide superior reaction efficiency and yield compared to other palladium sources, ensuring robust catalytic turnover for the carbonylative cyclization process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and potency. By leveraging innovative technologies like the one described in CN112239456B, we can offer cost-effective and sustainable manufacturing solutions tailored to your specific project needs.
We invite you to contact our technical procurement team to discuss how we can support your supply chain with customized synthesis services. Whether you require a Customized Cost-Saving Analysis for your current route or need to request specific COA data and route feasibility assessments for new targets, our experts are ready to provide the data-driven insights you need to make informed decisions. Partner with us to secure a reliable supply of complex pharmaceutical intermediates and accelerate your path to clinical and commercial success.
