Revolutionizing Evodiamine Production Advanced Catalytic Process for High-Purity Pharmaceutical Intermediates at Commercial Scale
The present analysis examines Chinese Patent CN110092786B which discloses an innovative method for synthesizing evodiamine—a critical pharmaceutical intermediate with significant applications in cardiovascular and analgesic drug development as evidenced by its inclusion in Japanese Pharmacopoeia quality control standards. This patented process addresses longstanding challenges in traditional synthesis routes by introducing a streamlined two-step approach that significantly enhances yield stability while reducing operational complexity through elimination of multiple purification stages that typically plague conventional methods. The methodology leverages non-reducing acid catalysis followed by organic base-mediated cyclization to achieve efficient conversion without requiring extensive isolation procedures thereby directly addressing purity concerns critical for pharmaceutical applications. Notably the process enables direct crystallization of high-purity evodiamine through refluxing techniques which simultaneously serves as both reaction completion indicator and purification mechanism thus substantially improving manufacturing efficiency. This advancement represents a substantial leap forward in natural product-derived pharmaceutical manufacturing offering tangible benefits across R&D procurement and supply chain operations within global pharmaceutical enterprises by transforming what was previously considered a complex extraction challenge into a scalable synthetic pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional evodiamine production methods primarily rely on complex extraction and purification processes from natural sources such as Evodia rutaecarpa which inherently suffer from inconsistent raw material quality seasonal availability constraints and extensive chromatographic separation requirements that severely limit scalability while introducing significant batch-to-batch variability in purity profiles. These conventional approaches typically involve multi-step isolation procedures requiring specialized equipment for handling unstable intermediates that degrade rapidly under standard processing conditions thereby necessitating costly cryogenic storage and immediate processing protocols that dramatically increase operational expenses. Furthermore the inherent structural complexity of indoloquinoline alkaloids like evodiamine makes traditional synthetic routes prone to side reactions generating difficult-to-remove impurities that compromise final product quality and necessitate additional purification stages which collectively reduce overall process efficiency below commercially viable thresholds for large-scale manufacturing operations. The cumulative effect of these limitations manifests as prohibitively long lead times inconsistent supply volumes and elevated costs that prevent reliable integration into modern pharmaceutical supply chains where stringent quality specifications must be met consistently across all production scales.
The Novel Approach
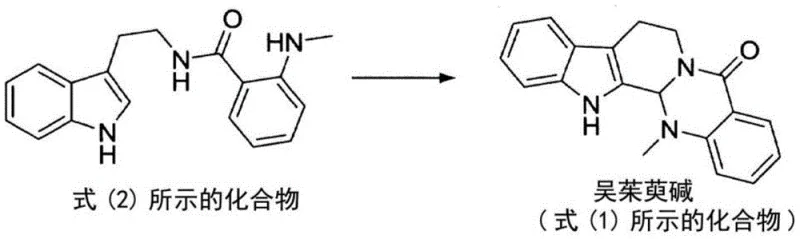
The patented methodology overcomes these limitations through an elegant two-stage synthetic pathway beginning with condensation of readily available tryptamine and N-methylisatoic anhydride followed by acid-catalyzed formylation and base-mediated cyclization which eliminates intermediate purification requirements while maintaining exceptional product quality through integrated crystallization design. By utilizing non-reducing acid catalysts such as acetic acid or scandium trifluoromethanesulfonate under precisely controlled thermal conditions this process achieves selective formylation without triggering decomposition pathways that commonly occur with traditional strong acid systems thereby preserving molecular integrity throughout the reaction sequence. The subsequent organic base treatment—particularly when conducted under reflux conditions—induces spontaneous crystallization of high-purity evodiamine directly from the reaction mixture eliminating multiple downstream processing steps that traditionally account for significant cost drivers in pharmaceutical intermediate manufacturing. This innovative approach transforms what was previously considered a complex extraction challenge into a robust synthetic pathway that demonstrates exceptional reproducibility across different production scales while maintaining stringent purity specifications required by global regulatory authorities.
Mechanistic Insights into Non-Reducing Acid Catalysis
The core innovation lies in the precisely orchestrated sequence where compound (2) undergoes formylation via orthoformate ester activation under non-reducing acid catalysis followed by intramolecular cyclization facilitated by organic bases such as pyridine or triethylamine which collectively enable efficient construction of evodiamines characteristic indoloquinoline skeleton without requiring transition metal catalysts or harsh reaction conditions. The non-reducing nature of acetic acid prevents unwanted reduction side reactions that commonly occur with stronger acids while maintaining sufficient proton activity to activate orthoformate esters toward electrophilic attack on the indole nitrogen thereby generating key iminium intermediates essential for ring closure. This mechanism avoids common pitfalls associated with traditional synthetic routes including over-reduction byproducts and catalyst contamination issues that necessitate expensive metal removal protocols thus directly contributing to enhanced product purity profiles critical for pharmaceutical applications where even trace impurities can trigger regulatory rejection during quality control assessments.
Impurity control is achieved through strategic reaction design where reflux conditions during the organic base treatment phase promote selective crystallization that inherently excludes soluble impurities while maintaining molecular conformation favorable for high-yield conversion—this self-purifying characteristic eliminates dedicated chromatography steps that traditionally account for significant cost drivers in pharmaceutical intermediate manufacturing. The process demonstrates exceptional tolerance to minor variations in starting material quality due to its integrated reaction-purification design which maintains consistent product specifications even when using unpurified intermediates thus providing substantial operational flexibility that enhances supply chain resilience against raw material fluctuations common in global chemical markets.
How to Synthesize Evodiamine Efficiently
This section outlines the operational framework for implementing CN110092786Bs patented methodology which transforms complex natural product extraction into a streamlined synthetic pathway suitable for commercial manufacturing environments where consistent quality and process reliability are paramount concerns across global pharmaceutical supply chains. The methodology leverages readily available starting materials through a precisely controlled sequence that minimizes unit operations while maximizing yield stability—detailed standardized synthesis procedures including exact temperature profiles solvent specifications and catalyst loading parameters are provided below to ensure seamless technology transfer from laboratory development to full-scale production facilities.
- Condense tryptamine with N-methylisatoic anhydride in an appropriate solvent at elevated temperature to form the key intermediate compound without requiring purification.
- React the intermediate with trimethyl orthoformate under non-reducing acid catalysis at controlled temperature to facilitate formylation.
- Heat the reaction mixture with an organic base such as pyridine under reflux conditions to induce cyclization and precipitate high-purity evodiamine crystals.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis pathway delivers transformative value across procurement and supply chain functions by addressing fundamental pain points inherent in traditional manufacturing approaches through strategic process simplification that directly impacts cost structures delivery reliability and environmental compliance metrics essential for modern pharmaceutical operations where regulatory pressures continue to intensify globally.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes expensive metal removal protocols while integrated crystallization design reduces solvent consumption by avoiding multiple chromatography steps—this strategic simplification significantly lowers raw material costs and operational expenses without compromising product quality thereby delivering substantial cost savings through reduced processing complexity rather than incremental efficiency gains.
- Enhanced Supply Chain Reliability: Utilizing globally available starting materials like tryptamine and N-methylisatoic anhydride creates multiple sourcing options that mitigate single-supplier dependency risks while the robust reaction profile maintains consistent output quality across different production scales—this inherent flexibility enables reliable delivery schedules even during market volatility periods by eliminating bottlenecks associated with specialized equipment requirements common in traditional synthesis routes.
- Scalability and Environmental Compliance: The process demonstrates exceptional scalability from laboratory batches directly to multi-ton annual production volumes due to its minimal unit operations design which also reduces waste generation through solvent recycling opportunities—this inherent environmental compatibility aligns with global sustainability initiatives while providing regulatory advantages through simplified waste stream management that avoids hazardous byproducts associated with conventional metal-catalyzed reactions.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations derived directly from patent specifications regarding implementation challenges quality assurance parameters and scalability potential—these responses provide essential insights for procurement teams evaluating supplier capabilities against evolving regulatory requirements within pharmaceutical manufacturing ecosystems.
Q: What are the primary limitations of conventional evodiamine synthesis methods?
A: Traditional approaches suffer from complex multi-step processes requiring extensive purification stages that hinder high-purity isolation and result in low overall yields due to unstable intermediates.
Q: How does this patented method improve product purity and yield?
A: The process eliminates intermediate purification through direct reaction sequences and leverages reflux-induced crystallization to produce high-purity evodiamine with significantly enhanced yield stability.
Q: What scalability advantages does this synthesis route offer for commercial manufacturing?
A: The simplified reaction pathway with minimal unit operations enables seamless scale-up from laboratory batches to multi-ton annual production while maintaining stringent quality specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Evodiamine Supplier
NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art facilities equipped with rigorous QC labs that ensure consistent compliance with global regulatory standards including ICH guidelines—our technical expertise transforms complex synthetic challenges like this patented evodiamine process into reliable manufacturing solutions that deliver both scientific excellence and commercial viability across international markets where quality consistency is non-negotiable.
Leverage our specialized knowledge by requesting a Customized Cost-Saving Analysis tailored to your specific production requirements—contact our technical procurement team today to obtain detailed COA data and comprehensive route feasibility assessments that will optimize your supply chain performance while ensuring seamless integration into your existing manufacturing infrastructure.
