Advanced Carbonylation Strategy for Evodiamine: Scaling Complex Alkaloid Production
Advanced Carbonylation Strategy for Evodiamine: Scaling Complex Alkaloid Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to produce bioactive alkaloids, and the synthesis of evodiamine stands out as a prime candidate for process innovation. Patent CN107629051B discloses a groundbreaking three-step method that utilizes carbonylation reactions to construct the evodiamine skeleton, marking a significant departure from conventional multi-step syntheses. This technology leverages N,N-dimethylaniline as a primary feedstock, transforming it through oxidative carbonylation, aminolysis, and final cyclization to yield the target compound with high structural fidelity. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more atom-economical and operationally simple manufacturing protocols. The ability to generate such a complex tetracyclic structure through a concise sequence not only enhances potential yields but also drastically reduces the cumulative waste associated with longer synthetic routes. As we analyze the technical specifics, it becomes clear that this methodology offers a robust framework for the reliable supply of high-purity evodiamine intermediates.
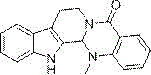
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of evodiamine has been hindered by reliance on N-methyl anthranilic acid as a starting material, which itself requires a preliminary methylation step from anthranilic acid. This traditional approach inherently introduces additional unit operations, increasing both the time and capital expenditure required for production. The methylation process often necessitates stringent control over stoichiometry to prevent over-alkylation or the formation of difficult-to-remove impurities, thereby complicating the purification workflow. Furthermore, the subsequent steps to build the indole-quinazoline fused ring system typically involve harsh reagents or extreme thermal conditions that can degrade sensitive functional groups. For supply chain managers, these inefficiencies translate into longer lead times and higher vulnerability to raw material price fluctuations, as the supply chain for specialized methylated precursors is often less robust than that for commodity chemicals. The cumulative effect of these drawbacks is a manufacturing process that struggles to meet the demands of modern green chemistry and cost-effective mass production.
The Novel Approach
In stark contrast, the method described in CN107629051B introduces a streamlined three-step sequence that bypasses the need for pre-methylated anthranilic acid derivatives entirely. By initiating the synthesis with N,N-dimethylaniline, a widely available and inexpensive commodity chemical, the process immediately lowers the barrier to entry for raw material sourcing. The core innovation lies in the first step: an oxidative carbonylation reaction that directly installs the carbonyl functionality while simultaneously constructing the isatoic anhydride motif. This is followed by a highly efficient aminolysis with tryptamine and a final acid-catalyzed cyclization. This logical progression minimizes the number of isolation and purification stages, which is critical for maintaining high overall throughput. The use of mild reaction conditions, particularly in the aminolysis step which proceeds at room temperature, further underscores the operational simplicity of this new route. For manufacturers, this translates to a significantly reduced footprint in terms of reactor occupancy time and energy consumption, facilitating a more agile response to market demand for this valuable therapeutic intermediate.
Mechanistic Insights into Pd-Catalyzed Oxidative Carbonylation
The heart of this synthetic innovation is the palladium-catalyzed oxidative carbonylation of N,N-dimethylaniline, a transformation that elegantly merges C-H activation with carbonyl insertion. In this mechanism, the palladium catalyst, potentially existing as Pd(II) species like palladium chloride or palladium acetate, coordinates with the nitrogen lone pair of the dimethylaniline substrate. Under an atmosphere of carbon monoxide and oxygen, the catalyst facilitates the insertion of a carbonyl group ortho to the amino group, driven by the re-oxidation of the metal center by the copper co-catalyst. This cycle effectively converts a simple aromatic amine into a reactive N-methyl isatoic anhydride, a versatile building block that primes the molecule for subsequent nucleophilic attack. The choice of oxidant, ranging from copper acetate to benzoquinone, plays a crucial role in regenerating the active palladium species, ensuring the catalytic turnover remains high throughout the reaction duration of 1 to 10 hours. Understanding this mechanistic nuance is vital for R&D teams aiming to optimize the process further, as fine-tuning the CO/O2 ratio (typically 10:1 to 100:1) can dramatically influence the selectivity and conversion rates.
Following the formation of the anhydride, the synthesis proceeds through a nucleophilic ring-opening aminolysis with tryptamine, leading to the formation of the key linear intermediate, N-(2-indolylethyl)-2-(methylamino) benzamide. This intermediate serves as the precise precursor for the final ring-closing event. The structural integrity of this molecule is paramount, as it contains the necessary spatial arrangement for the subsequent intramolecular condensation. The final cyclization, mediated by p-toluenesulfonic acid and triethyl orthoformate in refluxing toluene, acts as a dehydrative condensation that seals the quinazoline ring, completing the tetracyclic core of evodiamine. This step-wise construction allows for rigorous quality control at each stage, ensuring that impurities from the initial carbonylation do not propagate into the final API intermediate. The ability to isolate and characterize this specific amide intermediate provides a critical checkpoint for purity assurance.
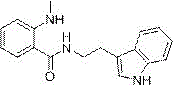
How to Synthesize Evodiamine Efficiently
To implement this synthesis effectively, operators must adhere to the specific parameters outlined in the patent examples, particularly regarding the catalyst loading and gas pressure. The process begins with the careful preparation of the reaction vessel under an inert atmosphere before introducing the CO/O2 mixture, ensuring safety and reaction consistency. Detailed standard operating procedures for the workup, including filtration to remove metal catalysts and column chromatography for purification, are essential for achieving the reported yields of up to 68% in the first step and 95% in the aminolysis.
- Perform oxidative carbonylation of N,N-dimethylaniline using a palladium catalyst and copper oxidant under CO/O2 atmosphere to yield N-methyl isatoic anhydride.
- Conduct aminolysis reaction between the resulting N-methyl isatoic anhydride and tryptamine in acetonitrile to form the key amide intermediate.
- Execute acid-catalyzed cyclization using p-toluenesulfonic acid and triethyl orthoformate in refluxing toluene to close the ring and form evodiamine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this carbonylation-based route offers profound advantages for procurement strategies and supply chain resilience. The shift away from complex, multi-step methylations towards a direct carbonylation using commodity gases (CO) and bulk chemicals (N,N-dimethylaniline) fundamentally alters the cost structure of evodiamine production. By eliminating the need for specialized, high-cost precursors, manufacturers can leverage economies of scale associated with petrochemical feedstocks. This reduction in raw material complexity directly correlates to a stabilization of supply costs, shielding the production budget from the volatility often seen in niche fine chemical markets. Furthermore, the simplified workflow reduces the requirement for extensive intermediate storage and handling, lowering overhead costs related to warehousing and logistics. For procurement managers, this means negotiating contracts based on more stable and predictable input costs, enhancing the overall financial viability of the project.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in the final steps and the use of earth-abundant copper salts as oxidants in the initial step significantly lowers the expense associated with reagent procurement. Unlike processes that require precious metals in stoichiometric amounts or difficult-to-remove heavy metals, this method utilizes catalytic amounts of palladium which can potentially be recovered, alongside inexpensive copper oxidants. The mild reaction conditions, particularly the room temperature aminolysis, result in substantial energy savings by removing the need for cryogenic cooling or extreme heating in those specific stages. Additionally, the high yield observed in the aminolysis step (up to 95%) minimizes material loss, ensuring that the maximum amount of valuable tryptamine is incorporated into the final product, thereby optimizing the cost-per-kilogram of the active intermediate.
- Enhanced Supply Chain Reliability: Sourcing N,N-dimethylaniline and tryptamine is significantly more straightforward than sourcing custom-synthesized methylated anthranilic acid derivatives, as these are established bulk chemicals with multiple global suppliers. This diversification of the supply base reduces the risk of single-source bottlenecks that can halt production lines. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent ratios, implies that the process is less sensitive to minor variations in utility quality or environmental conditions, leading to more consistent batch-to-batch output. For supply chain heads, this reliability translates into shorter lead times and the ability to maintain lower safety stock levels while still meeting delivery commitments to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The adherence to green chemistry principles is not merely a regulatory checkbox but a strategic asset for long-term scalability. The use of toluene and acetonitrile, while requiring proper management, is standard in the industry, and the avoidance of highly toxic reagents simplifies waste treatment protocols. The three-step nature of the synthesis reduces the total volume of solvent and reagents required per kilogram of product compared to longer linear sequences, thereby decreasing the facility's overall environmental footprint. This efficiency makes the process highly attractive for scale-up from pilot plant to commercial tonnage, as the engineering challenges associated with heat transfer and mixing are minimized by the mild thermal profile. Consequently, facilities can achieve commercial scale-up of complex pharmaceutical intermediates with reduced capital investment in specialized containment or scrubbing systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbonylation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the feasibility and benefits of the route.
Q: How does this carbonylation method improve upon traditional evodiamine synthesis?
A: Traditional methods often rely on the methylation of anthranilic acid, which involves multiple steps and lower overall efficiency. This novel three-step carbonylation route utilizes readily available N,N-dimethylaniline and constructs the core skeleton directly through oxidative carbonylation, significantly simplifying the synthetic pathway and reducing operational complexity.
Q: What are the critical reaction conditions for the oxidative carbonylation step?
A: The process employs a palladium catalyst system (such as PdCl2 or Pd(PPh3)2Cl2) alongside a copper oxidant. The reaction proceeds under a mixed gas atmosphere of carbon monoxide and oxygen (volume ratio 10:1 to 100:1) at moderate temperatures between 40°C and 150°C, ensuring mild conditions suitable for scale-up.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the method is designed with scalability in mind. It features simple operation processes, mild reaction conditions, and the use of common solvents like toluene and acetonitrile. Furthermore, it aligns with green chemistry principles by avoiding excessive hazardous reagents, making it highly viable for industrial production of high-purity evodiamine.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Evodiamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the carbonylation route described in CN107629051B for the production of high-value alkaloids. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions and gas-liquid transformations, maintaining stringent purity specifications through our rigorous QC labs. We understand that the consistency of evodiamine supply is critical for your downstream formulation processes, and our commitment to quality assurance guarantees that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this streamlined route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global market for traditional medicine components and novel therapeutics.
