Advanced Photocatalytic Synthesis of Beta-Aminocarbonyl Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to construct complex molecular scaffolds. A significant breakthrough in this domain is detailed in patent CN114957037A, which discloses a novel method for synthesizing beta-aminocarbonyl compounds through photo-induced homolytic decarboxylation of oxalyl oxime esters followed by addition to activated olefins. This technology represents a paradigm shift from traditional synthetic routes, offering a green chemistry approach that leverages visible light energy to drive chemical transformations. The beta-aminocarbonyl motif is a critical structural element found in numerous high-value bioactive molecules, including anticancer agents like paclitaxel and antidiabetic drugs such as sitagliptin. By enabling the direct construction of this skeleton under mild conditions, this invention addresses long-standing challenges in process chemistry, providing a reliable foundation for the development of next-generation pharmaceutical intermediates.
Historically, the synthesis of beta-aminocarbonyl backbones has relied heavily on multi-step sequences or transition metal-catalyzed aminocarbonylation reactions. Conventional methods often necessitate the use of carbon monoxide (CO) gas as the carbonyl source, which poses severe safety risks due to its high toxicity and potential for leakage in large-scale reactors. Furthermore, these traditional processes frequently require harsh reaction conditions, including elevated temperatures and pressures, along with expensive noble metal catalysts that can leave trace impurities difficult to remove from the final active pharmaceutical ingredient (API). The complexity of purification and the stringent safety protocols required for handling toxic gases significantly inflate the operational costs and extend the lead time for high-purity pharmaceutical intermediates. These limitations have created a pressing demand for alternative methodologies that can deliver the same structural complexity without the associated hazards and economic burdens.
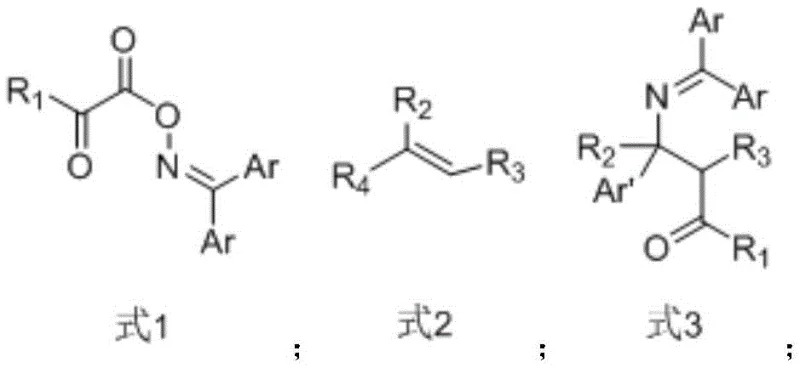
The novel approach described in the patent data overcomes these limitations by utilizing a visible light-induced radical mechanism. Instead of relying on toxic CO gas, this method employs oxalyl oxime esters as safe and stable precursors that undergo homolytic cleavage to generate the necessary carbonyl and amino radicals in situ. The reaction proceeds efficiently at room temperature using simple blue LED irradiation, eliminating the need for energy-intensive heating or high-pressure equipment. This transition from thermal/transition-metal catalysis to photocatalysis not only enhances the safety profile of the manufacturing process but also improves functional group tolerance. The mild conditions allow for the preservation of sensitive moieties on the substrate, thereby expanding the scope of accessible chemical space and enabling the synthesis of diverse derivatives that were previously difficult to obtain. This technological leap positions the method as a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Visible-Light Induced Homolytic Decarboxylation
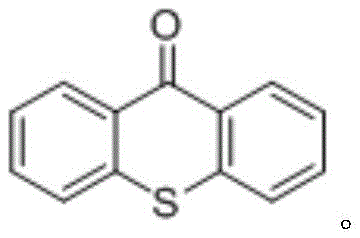
The core of this innovation lies in the precise manipulation of radical species through photoredox catalysis. Upon irradiation with visible light, the photocatalyst, preferably 9-thioxanthone, absorbs photon energy and enters an excited state. This excited species facilitates the homolytic cleavage of the weak N-O bond in the oxalyl oxime ester substrate. This cleavage event generates an imino radical and an acyloxy radical simultaneously. The acyloxy radical rapidly undergoes decarboxylation to release carbon dioxide and form a carbon-centered radical, which serves as the carbonyl source equivalent. This carbon radical then adds selectively to the activated olefin, followed by radical recombination or hydrogen atom transfer to finalize the beta-aminocarbonyl structure. The use of 9-thioxanthone is particularly advantageous as it is an organic photocatalyst that avoids the contamination issues associated with heavy metal complexes like iridium or ruthenium, ensuring a cleaner impurity profile for the final product.
Controlling the impurity profile is critical for any process intended for pharmaceutical applications. In this photo-induced system, the selectivity is governed by the kinetics of the radical addition steps. The difference in reaction rates between the oxygen-centered and nitrogen-centered radicals ensures that the addition occurs at the specific position on the olefin to yield the desired beta-aminocarbonyl motif rather than side products. The patent data indicates that the process tolerates a wide range of substituents on both the oxime ester and the olefin, including electron-withdrawing groups like halogens and nitro groups, as well as electron-donating alkyl and alkoxy groups. This broad substrate compatibility is evidenced by the successful synthesis of various derivatives with yields reaching up to 73%. The ability to maintain high selectivity across diverse electronic environments minimizes the formation of regioisomers and by-products, simplifying the downstream purification process and enhancing the overall mass balance of the synthesis.
How to Synthesize Beta-Aminocarbonyl Compounds Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility. The general protocol involves dissolving the oxalyl oxime ester and the activated olefin in a dry organic solvent, typically dichloromethane, although ethyl acetate and dimethyl sulfoxide are also viable options. A catalytic amount of 9-thioxanthone (1-5 mol%) is added to the mixture, which is then degassed with argon to prevent oxygen quenching of the radical species. The reaction vessel is placed under a 40W blue LED light source at a distance of approximately 1.5 cm and stirred at room temperature for 8 to 12 hours. Monitoring via thin-layer chromatography (TLC) is recommended to determine the endpoint. Once complete, the solvent is removed under reduced pressure, and the crude residue is purified via silica gel column chromatography using a petroleum ether and ethyl acetate gradient. Detailed standardized synthesis steps are provided in the guide below.
- Dissolve the oxalyl oxime ester and activated olefin in a suitable solvent such as dichloromethane, adding a photocatalyst like 9-thioxanthone.
- Purge the reaction mixture with inert gas (argon) and irradiate with a blue LED light source (40W) at room temperature for 8-12 hours.
- Monitor reaction progress via TLC, then concentrate under reduced pressure and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photo-induced synthesis method offers tangible strategic benefits beyond mere chemical novelty. The elimination of toxic carbon monoxide gas removes a significant regulatory and safety hurdle, simplifying facility compliance and reducing insurance liabilities. Furthermore, the shift from expensive transition metal catalysts to organic photocatalysts like 9-thioxanthone drastically lowers the raw material costs and eliminates the need for costly metal scavenging steps during purification. This simplification of the workflow translates directly into substantial cost savings in pharmaceutical intermediate manufacturing. The mild reaction conditions also mean that standard glass-lined reactors equipped with LED arrays can be used, avoiding the capital expenditure required for high-pressure autoclaves. These factors combined create a more resilient and cost-efficient supply chain for critical drug building blocks.
- Cost Reduction in Manufacturing: The replacement of noble metal catalysts with organic photocatalysts significantly reduces the bill of materials. Additionally, the avoidance of high-pressure equipment and toxic gas handling infrastructure lowers both capital and operational expenditures. The simplified workup procedure, which does not require complex metal removal resins, further decreases processing time and waste disposal costs, leading to a more economically viable production model for high-volume intermediates.
- Enhanced Supply Chain Reliability: By utilizing stable solid reagents like oxalyl oxime esters instead of gaseous carbon monoxide, the logistics of raw material storage and transport become much safer and more straightforward. The robustness of the reaction conditions allows for greater flexibility in scheduling and batch planning, reducing the risk of production delays caused by equipment failure or safety incidents. This reliability ensures a consistent supply of high-purity pharmaceutical intermediates to meet the demanding timelines of drug development projects.
- Scalability and Environmental Compliance: The use of visible light as the energy source aligns perfectly with green chemistry principles, minimizing the carbon footprint of the synthesis. The reaction generates carbon dioxide as the only stoichiometric by-product, which is easily managed. The scalability of LED technology allows for easy translation from laboratory benchtop to pilot plant and commercial scale without the engineering challenges associated with scaling thermal exotherms. This environmental compliance facilitates smoother regulatory approvals and enhances the sustainability profile of the final pharmaceutical product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this photo-induced synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity on process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of this photo-induced method over traditional transition metal catalysis?
A: This method eliminates the need for toxic carbon monoxide gas and expensive transition metal catalysts. It operates under mild visible light irradiation at room temperature, significantly improving safety profiles and reducing equipment costs associated with high-pressure or high-temperature reactors.
Q: What is the typical yield range for this synthesis protocol?
A: According to patent data CN114957037A, the process demonstrates robust efficiency with isolated yields ranging from 44% to 73%, depending on the specific substrate electronics and steric hindrance of the activated olefins used.
Q: Is this process scalable for industrial pharmaceutical intermediate production?
A: Yes, the protocol utilizes standard LED light sources and common organic solvents like dichloromethane. The mild reaction conditions (room temperature, atmospheric pressure) and absence of hazardous gases make it highly suitable for scale-up in commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Aminocarbonyl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photo-induced decarboxylation technology for the synthesis of complex pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our facilities are equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We are committed to delivering high-quality beta-aminocarbonyl compounds that serve as vital building blocks for the next generation of therapeutic agents.
We invite you to collaborate with us to leverage this advanced synthetic methodology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this greener route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a competitive advantage through superior chemistry and reliable supply chain execution.
