Advanced Catalytic Strategy for Diacetylene Intermediates: Scaling Efficiently for Global Supply Chains
Advanced Catalytic Strategy for Diacetylene Intermediates: Scaling Efficiently for Global Supply Chains
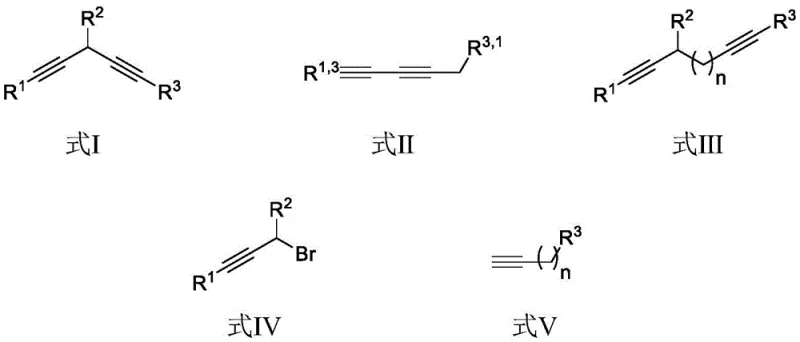
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more sustainable and efficient pathways to complex molecular scaffolds. A pivotal advancement in this domain is detailed in patent CN111662147A, which discloses a robust method for the preparation of various diynes and their derivatives. This technology addresses critical bottlenecks in the construction of conjugated alkyne systems, which are indispensable building blocks in modern medicinal chemistry and material science. By leveraging a monovalent copper catalytic system in conjunction with potassium phosphate bases, the process achieves high selectivity under remarkably mild conditions. For R&D directors and procurement specialists alike, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates. The ability to synthesize 1,3-diynes and 1,4-diynes with reduced metal waste and improved atom economy aligns perfectly with the industry's shift towards greener manufacturing protocols without compromising on yield or purity specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of asymmetric 1,3-diynes has been plagued by significant technical challenges that hinder efficient commercial scale-up. Traditional methodologies such as the Glaser-Hay coupling often suffer from unavoidable homocoupling side reactions, necessitating the use of a large excess of one alkyne substrate to drive the reaction towards the desired cross-coupled product. This not only inflates raw material costs but also complicates downstream purification processes, leading to substantial yield losses. Furthermore, the Cadiot-Chodkiewicz coupling, while effective for certain substrates, relies on the pre-functionalization of terminal alkynes into alkynyl bromides, which are notoriously unstable and hazardous to handle on a large scale. The requirement for stoichiometric amounts of copper salts in earlier iterations of these protocols further exacerbates environmental concerns, generating heavy metal waste streams that require costly treatment and disposal procedures. These legacy limitations create a fragile supply chain for complex diacetylene intermediates, often resulting in long lead times and inconsistent batch quality.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent data introduces a streamlined catalytic cycle that fundamentally reshapes the efficiency of diyne construction. By utilizing propargyl bromide compounds and terminal alkynes as direct substrates in the presence of a catalytic amount of monovalent copper, the process eliminates the need for stoichiometric metal reagents. The reaction proceeds smoothly in polar aprotic solvents like N,N-dimethylacetamide at a温和 temperature of 40°C, significantly reducing energy consumption compared to high-temperature alternatives. This methodology demonstrates exceptional versatility, capable of producing not only 1,4-diynes but also asymmetric 1,3-diynes and extended aryl 1,n-diynes with high fidelity. The strategic use of potassium phosphate as a mild base ensures compatibility with a wide range of functional groups, minimizing degradation of sensitive moieties often found in advanced pharmaceutical intermediates. This shift from stoichiometric to catalytic regimes marks a transformative step in cost reduction in fine chemical manufacturing.
Mechanistic Insights into Cu-Catalyzed Alkyne Coupling
The mechanistic elegance of this transformation lies in the precise activation of the terminal alkyne by the monovalent copper species. Under the optimized reaction conditions, the copper catalyst facilitates the formation of a copper-acetylide intermediate, which subsequently undergoes nucleophilic attack on the propargyl bromide electrophile. This pathway is carefully tuned to favor cross-coupling over homocoupling, a persistent issue in oxidative alkyne dimerizations. The choice of ligand-free copper bromide simplifies the catalytic system, removing the cost and complexity associated with expensive phosphine ligands or gold catalysts often required in alternative methods. Kinetic studies suggest that the mild basicity of potassium phosphate is sufficient to deprotonate the terminal alkyne without promoting unwanted elimination side reactions of the propargyl bromide. This delicate balance is crucial for maintaining high atom economy and ensuring that the final product profile remains clean, thereby reducing the burden on purification teams during process development.
From an impurity control perspective, the mild thermal conditions play a pivotal role in preserving the integrity of the conjugated diyne system. Conjugated alkynes can be prone to polymerization or decomposition under harsh acidic or basic conditions, particularly when exposed to elevated temperatures. By maintaining the reaction at 40°C, the process mitigates thermal stress on the product, preventing the formation of oligomeric byproducts that are difficult to separate. Furthermore, the inert nitrogen atmosphere protects the reactive copper-acetylide species from oxidation, ensuring consistent catalyst turnover throughout the 16-hour reaction window. This level of control over the reaction environment translates directly into a narrower impurity profile, which is a critical quality attribute for any reliable pharmaceutical intermediate supplier. The ability to consistently deliver material with minimal trace metals and organic impurities simplifies the regulatory filing process for downstream drug manufacturers.
How to Synthesize Diacetylenes Efficiently
Implementing this synthesis route requires careful attention to reagent quality and atmospheric control to maximize yield and reproducibility. The protocol begins with the precise weighing of the propargyl bromide derivative and the terminal alkyne, typically employing a slight molar excess of the alkyne to drive the reaction to completion without excessive waste. The reaction mixture is assembled in a sealed vessel under a nitrogen blanket to exclude moisture and oxygen, which can deactivate the copper catalyst. Following the reaction period, a standardized workup procedure involving aqueous quenching and organic extraction ensures the efficient recovery of the crude product. Detailed standard operating procedures for this specific transformation are essential for technology transfer teams aiming to replicate these results in a pilot plant setting.
- Combine propargyl bromide compound, terminal alkyne, potassium phosphate base, and CuBr catalyst in N,N-dimethylacetamide solvent.
- Maintain the reaction mixture under a nitrogen atmosphere at 40°C for approximately 16 hours to ensure complete conversion.
- Quench with saturated ammonium chloride, extract with ethyl acetate, dry over magnesium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic methodology offers tangible strategic advantages beyond mere technical feasibility. The transition from stoichiometric heavy metal reagents to a catalytic system drastically reduces the volume of hazardous waste generated per kilogram of product. This reduction in waste load directly correlates with lower disposal costs and simplified environmental compliance reporting, which are increasingly critical factors in the total cost of ownership for chemical ingredients. Moreover, the use of commercially available and stable starting materials, such as substituted phenylacetylenes and propargyl bromides, ensures a resilient supply chain that is less susceptible to raw material shortages. The robustness of the reaction conditions allows for flexible scheduling and batch sizing, enabling manufacturers to respond swiftly to fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts, such as gold, and the reduction of copper loading from stoichiometric to catalytic levels result in substantial raw material savings. Additionally, the mild reaction temperature of 40°C significantly lowers energy consumption compared to processes requiring reflux conditions or cryogenic cooling. The simplified workup procedure, which avoids complex chromatographic separations often needed to remove metal residues, further streamlines the production timeline. These cumulative efficiencies translate into a more competitive pricing structure for high-purity diacetylene intermediates, providing a clear economic advantage in cost-sensitive therapeutic areas.
- Enhanced Supply Chain Reliability: By utilizing reagents that are stable and readily sourced from global chemical suppliers, the risk of production delays due to raw material scarcity is minimized. The process tolerance for various substituents on the aromatic rings means that a single platform technology can be adapted to produce a wide library of analogs, reducing the need for multiple distinct synthetic routes. This versatility enhances supply security, as alternative substrates can be quickly substituted if specific precursors face logistical bottlenecks. Consequently, partners can rely on a consistent flow of materials, supporting continuous manufacturing campaigns and reducing the need for large safety stocks.
- Scalability and Environmental Compliance: The inherent safety of operating at low temperatures and atmospheric pressure makes this process highly amenable to scale-up from gram to multi-ton quantities. The reduced generation of heavy metal waste aligns with stringent global environmental regulations, facilitating smoother permitting processes for new manufacturing lines. The use of common solvents like N,N-dimethylacetamide allows for easy solvent recovery and recycling, further enhancing the sustainability profile of the operation. This commitment to green chemistry principles not only meets current compliance standards but also future-proofs the supply chain against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diacetylene synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view of the process capabilities. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect a balance between theoretical potential and practical operational realities observed during the development of this method.
Q: How does this method improve upon traditional Glaser-Hay coupling?
A: Unlike Glaser-Hay coupling which often suffers from homocoupling side reactions requiring excess alkyne, this patented method utilizes a specific propargyl bromide coupling strategy that minimizes self-coupling impurities and operates under milder conditions.
Q: What are the stability concerns regarding the alkynyl bromide substrates?
A: While traditional Cadiot-Chodkiewicz coupling requires unstable alkynyl bromides, this process optimizes the handling of propargyl bromide derivatives, allowing for efficient in-situ generation or stable storage protocols that enhance operational safety.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
A: Yes, the use of catalytic rather than stoichiometric copper, combined with mild temperatures (40°C) and common solvents like DMAc, significantly simplifies heat management and waste treatment, making it highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diacetylene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex molecules depends on a partner who understands both the chemistry and the supply chain dynamics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of diacetylene intermediate meets the exacting standards required for pharmaceutical applications. Our infrastructure is designed to handle the specific safety and handling requirements of alkyne chemistry, providing a secure and compliant environment for your most critical projects.
We invite you to engage with our technical procurement team to discuss how this innovative catalytic process can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this route offers compared to your current supply sources. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver high-quality intermediates with the reliability and transparency that defines our business relationships. Let us collaborate to optimize your supply chain and accelerate your path to market.
