Advancing Diacetylene Manufacturing: A Green Copper-Catalyzed Protocol for High-Purity Intermediates
The landscape of organic synthesis for conjugated systems is undergoing a significant transformation, driven by the urgent need for greener, more efficient methodologies in the production of fine chemical intermediates. Patent CN111662147A, published in September 2020, introduces a groundbreaking protocol for the preparation of various diynes and their derivatives, addressing long-standing challenges in the field of acetylene chemistry. This innovation leverages a monovalent copper catalytic system to couple propargyl bromide compounds with terminal alkynes, yielding structurally diverse 1,3-diynes and 1,4-diynes under remarkably mild conditions. For R&D directors and procurement specialists in the pharmaceutical and electronic materials sectors, this technology represents a pivotal shift away from harsh, stoichiometric metal processes toward a more sustainable and cost-effective manufacturing paradigm. The ability to synthesize these critical building blocks with high atom economy and reduced environmental impact positions this method as a cornerstone for next-generation intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of asymmetric 1,3-diynes and 1,4-diynes has been plagued by significant inefficiencies inherent in traditional coupling strategies such as the Glaser-Hay and Cadiot-Chodkiewicz reactions. The Glaser-Hay coupling, while classic, frequently suffers from competitive oxidative homocoupling of terminal alkynes, necessitating the use of a large excess of one substrate to drive the cross-coupling reaction forward, which drastically reduces atom economy and complicates downstream purification. Furthermore, the Cadiot-Chodkiewicz approach requires the pre-functionalization of one alkyne into an unstable alkynyl bromide, introducing safety hazards and stability issues during storage and handling. Earlier methods for 1,4-diyne synthesis often relied on stoichiometric amounts of organometallic reagents like Grignard or alkynyl lithium species, which demand stringent anhydrous conditions and cryogenic temperatures, thereby inflating operational costs and limiting scalability for commercial applications.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN111662147A utilizes a catalytic amount of monovalent copper, specifically copper bromide (CuBr), in conjunction with a mild inorganic base, potassium phosphate, to facilitate the coupling reaction. This novel approach eliminates the need for unstable alkynyl bromides by employing stable propargyl bromide precursors, which are commercially accessible and easier to handle. The reaction proceeds efficiently in polar aprotic solvents like N,N-dimethylacetamide (DMAc) at a moderate temperature of 40°C, significantly reducing energy consumption compared to high-temperature protocols. By shifting from stoichiometric metal usage to a catalytic regime (10-20 mol%), this process not only minimizes heavy metal waste but also simplifies the workup procedure, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations seeking to optimize their production lines.
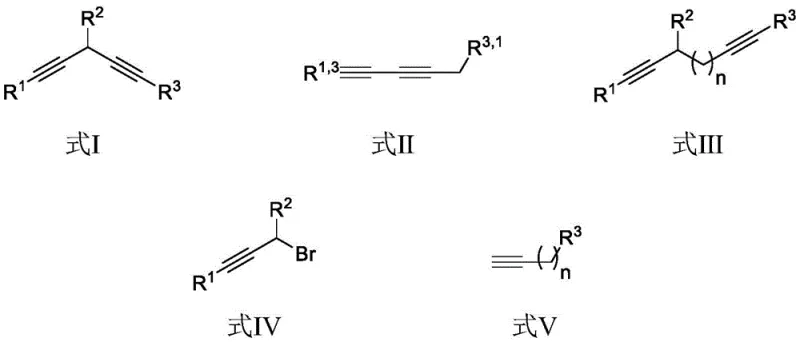
Mechanistic Insights into Cu-Catalyzed Cross-Coupling
The mechanistic pathway of this copper-catalyzed transformation involves the activation of the terminal alkyne by the monovalent copper species to form a reactive copper-acetylide intermediate in situ. This nucleophilic species then attacks the electrophilic propargyl bromide substrate, likely proceeding through an SN2 or SN2' displacement mechanism depending on the specific substitution pattern of the propargyl halide. The use of potassium phosphate as the base is critical, as comparative data within the patent demonstrates that weaker bases like potassium carbonate or dipotassium hydrogen phosphate result in significantly lower yields, highlighting the necessity for sufficient basicity to drive the deprotonation of the terminal alkyne without promoting decomposition of the sensitive propargyl bromide. Furthermore, the choice of solvent plays a pivotal role; DMAc provides superior solvation for the ionic intermediates compared to non-polar solvents like 1,2-dichloroethane, ensuring a homogeneous reaction environment that maximizes collision frequency between reactants.
From an impurity control perspective, this catalytic system offers distinct advantages by suppressing the formation of homocoupled byproducts that typically plague alkyne chemistry. The controlled generation of the copper-acetylide species prevents the accumulation of free radical intermediates that lead to Glaser-type dimers. Additionally, the mild reaction temperature of 40°C is optimal; experimental data shows that increasing the temperature to 60°C actually decreases the yield, suggesting that higher thermal energy promotes side reactions or catalyst degradation. This precise control over reaction parameters ensures a clean crude profile, reducing the burden on purification teams and enabling the production of high-purity OLED material or API precursors with minimal chromatographic effort. The robustness of the catalyst system allows for a broad substrate scope, accommodating both electron-rich and electron-deficient aryl groups as well as heterocycles like thiophene.
How to Synthesize Diacetylenes Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to strict procedural guidelines to replicate the high yields reported in the patent examples. The process begins with the precise weighing of the propargyl bromide derivative and the terminal alkyne substrate, maintaining a molar ratio where the alkyne is slightly in excess (1.2 to 1.5 equivalents) to ensure complete consumption of the halide. These reagents are combined with the copper bromide catalyst and potassium phosphate base in a reaction vessel equipped with magnetic stirring, followed by the addition of anhydrous N,N-dimethylacetamide. It is imperative to maintain an inert nitrogen atmosphere throughout the process to prevent oxidation of the copper catalyst and the alkyne substrates. The detailed standardized synthesis steps are provided in the guide below.
- Mix propargyl bromide compound, terminal alkyne, potassium phosphate base, and copper bromide catalyst in N,N-dimethylacetamide solvent.
- Seal the reaction vessel under a nitrogen inert atmosphere and stir at 40°C for 16 hours to ensure complete conversion.
- Quench with saturated ammonium chloride, extract with ethyl acetate, dry over magnesium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed protocol offers substantial strategic benefits that extend beyond mere technical feasibility. The transition from stoichiometric organometallic reagents to a catalytic system fundamentally alters the cost structure of diacetylene manufacturing by drastically reducing the consumption of expensive metal salts. Since the catalyst loading is kept low at 10-20 mol%, the raw material costs associated with metal procurement are significantly minimized, leading to direct cost reduction in fine chemical intermediates manufacturing. Moreover, the elimination of cryogenic conditions and the ability to run reactions at 40°C translates to lower energy expenditures for heating and cooling infrastructure, further enhancing the overall economic viability of the process for large-scale production runs.
- Cost Reduction in Manufacturing: The shift to a catalytic copper system removes the financial burden of purchasing and disposing of stoichiometric amounts of heavy metals, which are often costly and subject to strict environmental regulations. By utilizing inexpensive copper bromide and common inorganic bases, the process achieves a leaner cost profile without compromising on yield or quality. This efficiency allows manufacturers to offer more competitive pricing for complex diacetylene derivatives, making it a viable option for cost-sensitive projects in the generic pharmaceutical sector.
- Enhanced Supply Chain Reliability: The reliance on stable propargyl bromides and commercially available terminal alkynes mitigates the risk of supply chain disruptions often associated with specialized or unstable reagents like alkynyl lithium or Grignard reagents. These standard starting materials are widely sourced from multiple global vendors, ensuring continuity of supply even during market fluctuations. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, providing a buffer against supply chain volatility.
- Scalability and Environmental Compliance: The mild reaction conditions and reduced metal waste align perfectly with modern green chemistry principles, simplifying the regulatory approval process for new manufacturing sites. The absence of pyrophoric reagents enhances operational safety, reducing insurance and compliance costs. Furthermore, the simplified workup procedure involving standard aqueous extraction and silica gel chromatography facilitates easy scale-up from gram to kilogram quantities, supporting the commercial scale-up of complex polymer additives or drug intermediates without the need for specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diacetylene synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process optimization and application scope. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this method into existing production workflows.
Q: How does this copper-catalyzed method improve upon traditional Glaser-Hay coupling?
A: Unlike Glaser-Hay coupling which often suffers from homocoupling side reactions requiring excess reagents, this method utilizes stable propargyl bromides and catalytic copper to achieve high selectivity for cross-coupling without significant byproduct formation.
Q: What are the optimal reaction conditions for maximizing yield?
A: The patent data indicates that using N,N-dimethylacetamide (DMAc) as the solvent with potassium phosphate (K3PO4) as the base at 40°C provides superior yields compared to higher temperatures or alternative solvents like dichloroethane.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of mild temperatures (40°C), catalytic amounts of inexpensive copper salts, and readily available starting materials makes this protocol highly amenable to scale-up for industrial manufacturing of fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diacetylenes Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and material science innovation. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of diacetylene intermediate delivered meets the highest industry standards for pharmaceutical and electronic applications.
We invite you to collaborate with our technical procurement team to explore how this copper-catalyzed technology can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in high-quality chemical manufacturing.
