Advancing Benzofluorenone Manufacturing: A Technical Analysis of Cu-Catalyzed Radical Cyclization
Introduction to Advanced Benzofluorenone Synthesis Technology
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex polycyclic aromatic frameworks, particularly benzofluorenones, which serve as critical scaffolds in drug discovery and material science. As detailed in patent CN110872218B, a novel copper-catalyzed approach has emerged that fundamentally shifts the paradigm for accessing these valuable structures. This technology addresses the longstanding challenges associated with synthesizing bioactive molecules such as cauliphine, an alkaloid exhibiting potent anti-myocardial ischemia activity, and intermediates for antibiotics like prekinamycin. 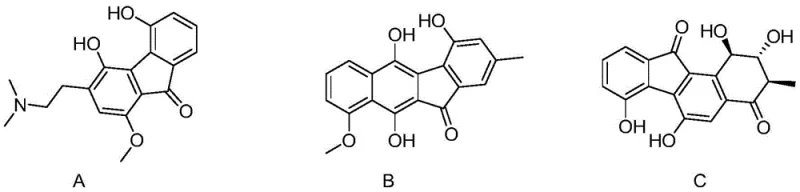 By leveraging a radical cycloaddition mechanism, this invention provides a safe, environmentally friendly, and highly efficient pathway that eliminates the need for hazardous oxidants, thereby offering a superior alternative for the reliable pharmaceutical intermediate supplier seeking to optimize their production portfolios.
By leveraging a radical cycloaddition mechanism, this invention provides a safe, environmentally friendly, and highly efficient pathway that eliminates the need for hazardous oxidants, thereby offering a superior alternative for the reliable pharmaceutical intermediate supplier seeking to optimize their production portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzofluorenone core has relied upon several classical synthetic strategies, each fraught with significant operational and environmental drawbacks that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Traditional routes often involve the direct oxidation of benzofluorene, which necessitates the use of stoichiometric amounts of toxic and severely polluting oxidants, creating substantial waste disposal burdens and safety risks for plant personnel. Alternatively, Friedel-Crafts acylation of aryl carboxylic acids typically demands harsh reaction conditions, including high temperatures and strong Lewis acids, which frequently result in poor functional group tolerance and difficult purification profiles. Furthermore, intramolecular Diels-Alder reactions of propynones, while conceptually elegant, often suffer from limited substrate scope and require rigorous exclusion of moisture and oxygen, complicating the manufacturing process and driving up overall production costs.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN110872218B introduces a transformative copper-catalyzed radical cyclization strategy that operates under remarkably mild conditions. This innovative route utilizes readily available precursors, specifically an alkynyl ketone derivative (Formula I) and an aryl diazonium tetrafluoroborate salt (Formula II), which undergo oxidative aromatization in the presence of a catalytic amount of copper iodide or copper bromide. 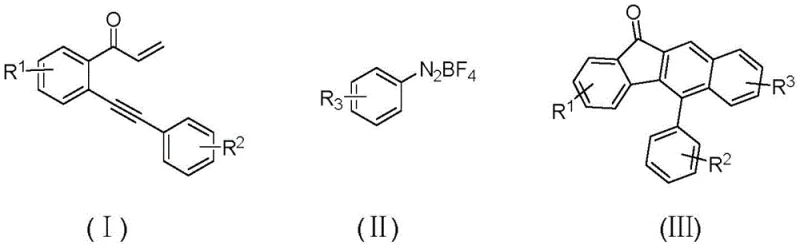 The reaction proceeds efficiently at temperatures between 70-90°C, preferably at 80°C, completing within a short timeframe of 1-3 hours. Crucially, this method employs non-toxic ethanol as the primary solvent, eliminating the need for chlorinated solvents or expensive polar aprotic media, and achieves high atom economy by avoiding the generation of waste gas, thus representing a significant leap forward in cost reduction in pharmaceutical intermediate manufacturing.
The reaction proceeds efficiently at temperatures between 70-90°C, preferably at 80°C, completing within a short timeframe of 1-3 hours. Crucially, this method employs non-toxic ethanol as the primary solvent, eliminating the need for chlorinated solvents or expensive polar aprotic media, and achieves high atom economy by avoiding the generation of waste gas, thus representing a significant leap forward in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Cu-Catalyzed Radical Cyclization
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the copper catalyst, which enables a free radical ring addition mode that is both selective and robust. The reaction initiates with the interaction between the copper catalyst and the diazonium salt, generating an aryl radical species in situ under thermal conditions without the need for external photo-irradiation or harsh chemical initiators. This aryl radical then attacks the electron-deficient alkyne moiety of the enone substrate, triggering a cascade of cyclization events that ultimately forge the rigid benzofluorenone skeleton. The use of cuprous iodide or cuprous bromide at loading levels as low as 0.05 to 0.15 equivalents ensures that the catalytic cycle is sustained efficiently, minimizing metal contamination in the final product and reducing the burden on downstream purification processes, which is a critical consideration for R&D directors focused on impurity control.
Furthermore, the mechanism exhibits exceptional tolerance towards a wide array of electronic and steric environments, allowing for the introduction of diverse substituents without compromising yield or selectivity. The patent data indicates that substituents such as halogens (fluorine, chlorine, bromine), alkyl groups (methyl, tert-butyl), and alkoxy groups can be accommodated at various positions on the aromatic rings (R1, R2, R3). This versatility is attributed to the radical nature of the transformation, which is less sensitive to electronic deactivation compared to ionic mechanisms like electrophilic aromatic substitution. Consequently, this method enables the rapid generation of diverse libraries of high-purity OLED material precursors or drug candidates, facilitating structure-activity relationship studies and accelerating the drug development timeline for partner organizations.
How to Synthesize Benzofluorenone Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and reproducibility. The process involves mixing the alkynyl ketone substrate, the diazonium salt coupling partner, and the copper catalyst in a solvent system, followed by controlled heating and a straightforward workup procedure. The simplicity of the protocol, which avoids inert atmosphere gloveboxes or specialized high-pressure reactors, makes it accessible for standard organic synthesis facilities. For detailed operational instructions, please refer to the standardized synthesis guide below which outlines the precise molar ratios and purification steps required to achieve the reported yields of 43% to 65% across various substrates.
- Mix compound (I), diazonium salt (II), and Cu catalyst (CuI/CuBr) in ethanol solvent.
- Heat the mixture to 70-90°C and react for 1-3 hours to facilitate radical cycloaddition.
- Cool to room temperature, adsorb on silica gel, and purify via column chromatography to isolate the target benzofluorenone (III).
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this copper-catalyzed methodology offers compelling advantages that directly address key pain points in the global supply chain for fine chemicals. The shift away from hazardous oxidants and harsh acidic conditions not only enhances workplace safety but also significantly reduces the regulatory compliance burden associated with waste management and environmental protection. By utilizing ethanol, a commodity chemical with a stable global supply, manufacturers can insulate themselves from the volatility of specialized solvent markets, ensuring consistent production continuity even during periods of raw material scarcity. This stability is paramount for supply chain heads who must guarantee uninterrupted delivery of critical intermediates to downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive and toxic reagents, which lowers both raw material costs and waste disposal fees. The use of catalytic amounts of inexpensive copper salts, rather than stoichiometric precious metals or oxidants, drastically reduces the cost of goods sold (COGS). Additionally, the mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for exotic corrosion-resistant alloys, leading to substantial capital expenditure savings and lower maintenance costs over the lifecycle of the production equipment.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically substituted chalcones or alkynyl ketones and aryl diazonium salts, are derived from abundant commodity feedstocks such as anilines and acetophenones. This broad base of raw material availability mitigates the risk of supply bottlenecks that often plague niche synthetic routes dependent on single-source suppliers. Furthermore, the robustness of the reaction against moisture and oxygen variations means that production schedules are less likely to be disrupted by minor fluctuations in plant conditions, ensuring a reliable benzofluorenone supplier can meet tight delivery windows consistently.
- Scalability and Environmental Compliance: The inherent safety profile of this method, characterized by low operation risk and the absence of waste gas generation, makes it ideally suited for scaling from kilogram to multi-ton quantities. The simplified workup procedure, involving silica gel adsorption and standard column chromatography or crystallization, facilitates easier technology transfer from R&D to commercial manufacturing scales. Moreover, the alignment with green chemistry principles through the use of non-toxic solvents and high atom economy supports corporate sustainability goals, enhancing the marketability of the final product to environmentally conscious pharmaceutical clients.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patented technology, we have compiled a set of frequently asked questions based on the specific data and embodiments provided in the patent documentation. These insights are designed to address common concerns regarding reaction scope, safety protocols, and scalability potential. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows or for procurement specialists assessing the long-term viability of this supply source.
Q: What are the primary advantages of this Cu-catalyzed method over traditional oxidation routes?
A: Unlike traditional methods that require toxic oxidants or harsh Friedel-Crafts conditions, this patent describes a radical cycloaddition pathway using mild temperatures (70-90°C) and non-toxic ethanol, significantly reducing environmental hazards and operational risks.
Q: What is the substrate scope for this benzofluorenone synthesis?
A: The method demonstrates excellent adaptability, tolerating various substituents including halogens (F, Cl, Br), alkyl groups (methyl, tert-butyl), and alkoxy groups on the aromatic rings, allowing for diverse functionalization of the core structure.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes inexpensive copper catalysts and common solvents like ethanol, operates at atmospheric pressure without specialized high-pressure equipment, and avoids hazardous waste gas generation, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofluorenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Cu-catalyzed synthesis route described in CN110872218B for producing high-value benzofluorenone derivatives. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzofluorenone intermediate adheres to the highest industry standards for pharmaceutical and electronic applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green chemistry approach can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply chain for your critical benzofluorenone needs.
