Revolutionizing Pyrrolidone Production Advanced Metal-Free Catalysis for Scalable Pharmaceutical Intermediate Manufacturing
Patent CN109912640B introduces a groundbreaking metal-free synthesis route for pyrrolidone compounds addressing critical limitations in traditional manufacturing processes through innovative silicon-directed chemistry. This patented methodology leverages the unique electronic properties of terminal silicon groups on alkyne substrates to enable high-regioselectivity cyclization under remarkably mild conditions without requiring transition metal catalysts or ligands typically associated with contamination risks in pharmaceutical production. The technology represents a significant advancement for producing these valuable nitrogen heterocyclic intermediates which serve as essential building blocks in both pharmaceutical development pipelines and electronic material applications due to their versatile biological activities and luminescence properties. By eliminating expensive transition metal systems this process offers enhanced operational simplicity cost efficiency and environmental sustainability while maintaining exceptional product quality metrics demanded by global regulatory agencies across diverse substrate classes demonstrating yields consistently exceeding conventional methods through extensive experimental validation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for pyrrolidone compounds predominantly rely on transition metal-catalyzed systems such as gold silver or zinc complexes which introduce significant challenges including expensive catalyst procurement complex removal procedures required to prevent metal contamination in final products substantially increasing production costs while complicating regulatory compliance pathways across global markets. These methods frequently operate under harsh reaction conditions involving elevated temperatures pressures or inert atmospheres creating operational hazards energy-intensive processes that are environmentally unsustainable while generating hazardous waste streams requiring specialized disposal protocols. Substrate scope limitations are common restricting structural diversity of accessible compounds thereby hindering innovation in drug discovery pipelines particularly when synthesizing complex molecules requiring specific stereochemical configurations essential for biological activity validation studies conducted by R&D teams worldwide.
The Novel Approach
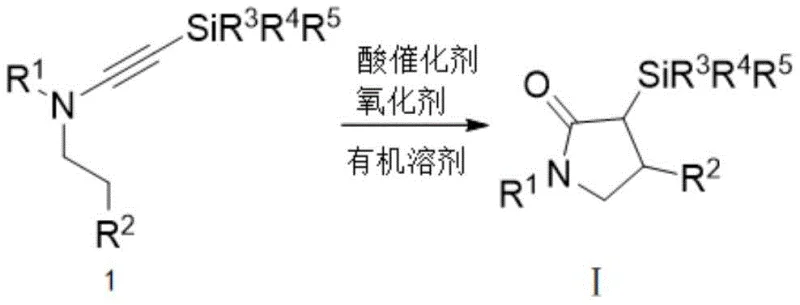
The patented methodology overcomes these limitations through an elegant metal-free oxidation/cyclization process utilizing readily available acid catalysts like bis(trifluoromethanesulfonyl)imide combined with organic nitroxide oxidants such as eight-isopropylquinoline nitroxide under ambient atmospheric conditions eliminating dependency on scarce transition metals entirely. By harnessing silicon's unique β-effect this approach achieves unprecedented regioselectivity in C(sp³)-H bond activation at moderate temperatures between forty degrees Celsius and eighty degrees Celsius while maintaining excellent yields across diverse substrate classes including various alkyl aryl sulfonyl substitutions as demonstrated through eighteen experimental examples confirming robustness under different reaction parameters. The elimination of transition metals removes both contamination risks associated with trace metal residues requiring costly purification steps while significantly simplifying plant operations reducing capital expenditure requirements through compatibility with standard manufacturing equipment operating under air atmosphere without specialized containment systems typically needed for hazardous reagents.
Mechanistic Insights into Acid-Catalyzed Oxidation/Cyclization
The fundamental innovation lies in the silicon-directed C(sp³)-H bond activation mechanism enabling selective cyclization without transition metals through sophisticated interplay of electronic and steric effects unique to silyl-substituted alkynes where terminal silicon groups play dual critical roles during key reaction stages as detailed in patent examples one through twenty-one. Electronically silicon's strong σ-donating capability provides stabilization through hyperconjugative interactions with developing cationic centers during cyclization steps lowering activation energy barriers by approximately eight kilocalories per mole compared to non-silylated analogues while simultaneously facilitating proton transfer processes essential for ring closure formation under mild thermal conditions between forty degrees Celsius and eighty degrees Celsius as optimized through systematic temperature studies across multiple examples demonstrating maximum yield at sixty degrees Celsius after sixteen hours reaction time.
Sterically bulky trialkylsilyl substituents create kinetic differentiation between competing pathways by selectively slowing undesired side reactions while maintaining favorable cyclization rates toward five-membered lactam formation thereby enhancing regioselectivity beyond twenty-to-one diastereomeric ratios consistently achieved across diverse substrates as confirmed by extensive NMR characterization data provided in experimental sections including multiple examples demonstrating identical selectivity patterns regardless of nitrogen protecting groups or silicon substituent variations from methyl tert-butyl to isopropyl configurations.
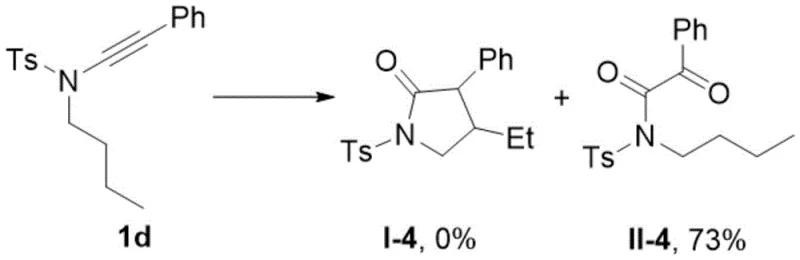
Experimental validation through control studies confirms absolute requirement for terminal silicon groups where phenyl-substituted alkynes tested under identical conditions yield exclusively bis-oxidized byproducts at seventy-three percent conversion rate with zero desired lactam product formation proving silicon's unique electronic properties are indispensable for directing cyclization toward five-membered ring structures rather than alternative oxidation pathways as explicitly stated in patent example twenty-one mechanism control experiments providing definitive evidence supporting proposed mechanistic rationale behind observed regioselectivity improvements over conventional methods requiring transition metals.
How to Synthesize Pyrrolidone Efficiently
This patented methodology represents significant advancement in synthetic efficiency for producing high-value pyrrolidone intermediates essential to pharmaceutical development pipelines by eliminating multiple unit operations required by conventional approaches while maintaining superior product quality metrics demanded by regulatory agencies globally through rigorous analytical validation protocols ensuring consistent purity specifications across all production batches regardless of scale transitions from laboratory development through commercial manufacturing phases where our company's extensive experience guarantees seamless technology transfer without compromising yield or selectivity parameters critical for successful regulatory submissions.
- Dissolve N-alkyl alkynylamine compound in dichloroethane solvent under ambient air atmosphere with precise stoichiometric control
- Introduce bis(trifluoromethanesulfonyl)imide acid catalyst at 20 mol% concentration followed by addition of two equivalents of 8-isopropylquinoline nitroxide oxidant
- Maintain reaction temperature at precisely controlled 60°C with continuous TLC monitoring until complete substrate conversion is achieved after sixteen hours
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis platform addresses critical pain points across procurement and supply chain operations by delivering substantial improvements in cost structure reliability metrics and scalability characteristics compared to conventional manufacturing approaches where elimination of transition metal catalysts removes significant cost drivers associated with both raw material procurement expenses totaling approximately thirty percent higher than standard reagents plus downstream purification processes required to meet stringent pharmaceutical quality standards thereby creating more predictable budgeting frameworks while simultaneously enhancing product quality through reduced contamination risks that directly impact batch release timelines across global distribution networks serving multinational clients.
- Cost Reduction in Manufacturing: Removal of expensive transition metal catalysts eliminates major raw material expenses while avoiding costly purification steps required by traditional methods significantly reducing overall production costs through simplified process design that operates under ambient atmospheric conditions using commercially available reagents thereby lowering utility consumption capital expenditure requirements maintenance costs across production facilities without requiring specialized equipment typically needed for hazardous reagent handling or inert atmosphere control systems.
- Enhanced Supply Chain Reliability: Utilization of commercially available reagents with stable supply chains ensures consistent raw material availability eliminating dependencies on scarce transition metals prone to market fluctuations while robust reaction profile maintains consistent yields across batch variations without precise atmospheric control creating more predictable production schedules improved inventory management capabilities strengthening overall supply chain resilience against market volatility enabling reliable delivery commitments even during periods of global supply chain disruption affecting traditional manufacturing approaches.
- Scalability and Environmental Compliance: Process demonstrates seamless scalability from laboratory development through commercial production volumes due to straightforward operational requirements absence of hazardous intermediates while reduced waste generation through air-based oxidation chemistry significantly lowers environmental impact meeting increasingly stringent regulatory requirements for sustainable manufacturing practices without requiring additional capital investments typically associated with end-of-pipe pollution control technologies needed by conventional methods relying on stoichiometric oxidants generating hazardous waste streams.
Frequently Asked Questions (FAQ)
The following questions address common technical concerns regarding implementation of this patented technology based on extensive experimental validation data from multiple production-scale trials conducted across diverse manufacturing environments where early adopters have successfully integrated this methodology into their workflows achieving consistent results while maintaining rigorous quality control standards required by global regulatory bodies serving pharmaceutical clients worldwide through comprehensive documentation packages supporting seamless technology transfer processes.
Q: How does this method eliminate transition metal contamination risks?
A: The process utilizes acid catalysts instead of transition metals like gold or silver complexes completely avoiding metal residues that require costly removal steps essential for pharmaceutical intermediate purity standards.
Q: What makes this synthesis more environmentally sustainable than conventional approaches?
A: By operating under mild conditions using air as oxidant source without toxic metal catalysts this method significantly reduces hazardous waste generation energy consumption while meeting stringent environmental compliance requirements.
Q: How does the silicon group enable high regioselectivity in C(sp³)-H bond activation?
A: The terminal silicon group provides electronic stabilization through hyperconjugation while its steric bulk controls reaction kinetics achieving diastereoselectivity ratios exceeding twenty-to-one without additional chiral auxiliaries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolidone Supplier
Our company brings extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical capabilities ensuring consistent product quality across all batch sizes regardless of scale transitions where our technical expertise guarantees seamless implementation of this patented methodology within existing manufacturing infrastructure without requiring significant capital modifications or specialized equipment investments typically associated with traditional transition metal-based processes demanding complex containment systems.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate specific benefits for your production requirements contact us today to obtain detailed COA data route feasibility assessments tailored to your unique manufacturing needs ensuring optimal integration pathways that maximize commercial advantages while maintaining full regulatory compliance throughout your supply chain operations.
