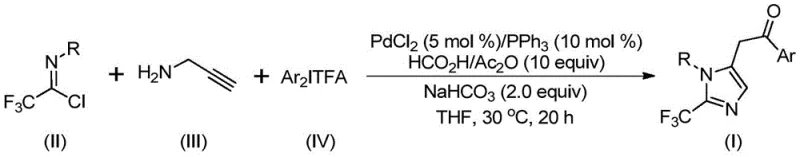
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups, due to their profound impact on drug pharmacokinetics. Patent CN111423381A introduces a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing critical gaps in current synthetic capabilities. This innovation leverages a transition metal palladium-catalyzed carbonylation tandem reaction, utilizing readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts. The significance of this technology lies in its ability to operate under exceptionally mild conditions, specifically at 30°C, while maintaining high reaction efficiency and broad substrate compatibility. For R&D directors and process chemists, this represents a paradigm shift away from hazardous reagents towards safer, more controllable synthetic pathways that are inherently designed for scalability and regulatory compliance in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functionalities has been fraught with significant challenges regarding safety and operational complexity. Traditional literature methods often rely on synthons like trifluorodiazoethane, which, while effective, pose severe safety hazards due to their explosive nature and instability, making them unsuitable for large-scale commercial manufacturing. Furthermore, alternative approaches using trifluoroethylimide acid halides have not been widely explored or optimized, leaving a gap in practical application despite their theoretical potential. Conventional routes frequently require harsh reaction conditions, specialized equipment for handling toxic gases like carbon monoxide, and complex purification protocols to remove heavy metal residues or unstable byproducts. These factors collectively drive up the cost of goods sold (COGS) and introduce substantial supply chain risks, particularly when aiming for the consistent quality required for active pharmaceutical ingredient (API) production.
The Novel Approach
The methodology disclosed in the patent offers a sophisticated solution by employing a palladium-catalyzed system that integrates carbonylation directly into the heterocycle formation process. By utilizing trifluoroethylimidoyl chloride and propargylamine as key building blocks, the reaction proceeds through a cascade mechanism that efficiently constructs the imidazole core while installing the trifluoromethyl group at the 2-position with high precision. A standout feature of this novel approach is the in situ generation of carbon monoxide from a formic acid and acetic anhydride mixture, which completely eliminates the need for external CO gas sources, thereby enhancing operational safety and simplifying reactor design. This strategy not only improves the atom economy of the process but also ensures excellent functional group tolerance, allowing for the synthesis of diverse derivatives essential for medicinal chemistry optimization campaigns without compromising yield or purity.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Reaction
The mechanistic pathway of this transformation is a testament to modern organometallic catalysis, involving a intricate sequence of bond formations and rearrangements driven by the palladium center. The reaction likely initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, yielding a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization, followed by palladium-catalyzed aminopalladation of the alkyne moiety to generate a vinyl-palladium intermediate. Further isomerization leads to an alkyl-palladium species, which is then subjected to carbonylation under the influence of carbon monoxide released from the formic acid/acetic anhydride additive, forming a crucial acyl-palladium intermediate. The cycle concludes with oxidative addition of the diaryliodonium salt to form a tetravalent palladium species, followed by reductive elimination to release the final 2-trifluoromethyl substituted imidazole product and regenerate the active catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal cyclizations. The stepwise nature of the palladium cycle allows for better kinetic control over side reactions, minimizing the formation of polymeric byproducts or regioisomers that often plague imidazole synthesis. The use of diaryliodonium salts as oxidants and aryl sources is particularly beneficial, as these reagents are generally stable and predictable in their reactivity profiles compared to aryl halides which might require higher temperatures or more aggressive ligands. Furthermore, the mild reaction temperature of 30°C significantly reduces the thermal degradation of sensitive functional groups on the aromatic rings, ensuring that the final impurity profile is clean and manageable, which is a critical parameter for downstream processing and regulatory filing of pharmaceutical intermediates.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
To implement this synthesis effectively, precise control over stoichiometry and reaction parameters is essential to maximize yield and minimize waste. The process begins by charging a reaction vessel with the palladium catalyst system, specifically palladium chloride and triphenylphosphine, along with sodium bicarbonate as the base. The carbon monoxide source, a mixture of formic acid and acetic anhydride, is added alongside the organic solvent, preferably tetrahydrofuran (THF) for optimal solubility and conversion rates. The key substrates, trifluoroethylimidoyl chloride, propargylamine, and the specific diaryliodonium salt corresponding to the desired aryl group, are then introduced. Detailed standardized synthesis steps follow below to ensure reproducibility and safety.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, and a mixture of acetic anhydride and formic acid in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter, mix with silica gel, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
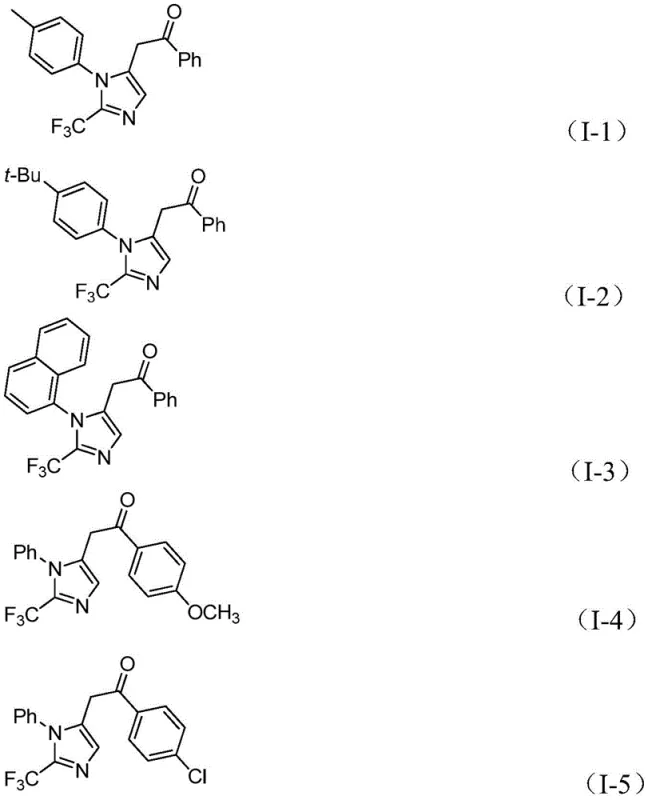
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The reliance on commercially available and inexpensive starting materials, such as aromatic amines for preparing the imidoyl chloride and standard propargylamine, drastically simplifies the sourcing strategy and reduces exposure to volatile raw material markets. By eliminating the need for specialized high-pressure equipment or hazardous gas handling infrastructure, the capital expenditure (CAPEX) required for implementation is significantly lowered, making it accessible for both pilot plants and full-scale commercial facilities. This accessibility translates directly into a more resilient supply chain capable of responding quickly to market demands for complex heterocyclic building blocks.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of cheap catalysts like palladium chloride and the avoidance of expensive, specialized trifluoromethylating agents. The in situ generation of carbon monoxide removes the cost and safety burden associated with purchasing and storing CO gas cylinders. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to lower operational expenditures (OPEX). The high atom efficiency and reduced need for extensive purification steps due to cleaner reaction profiles further drive down the overall cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved as the key reagents, including diaryliodonium salts and trifluoroethylimidoyl chlorides, can be synthesized from widely available commodity chemicals like aryl boronic acids and aromatic amines. This reduces dependency on single-source suppliers for exotic reagents. The robustness of the reaction across various substrates means that supply disruptions for one specific derivative can often be mitigated by adjusting the synthetic route slightly without changing the core process infrastructure, ensuring continuous availability of critical intermediates for downstream drug synthesis.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively at the gram level with clear pathways to kilogram and tonnage production. The use of THF as a solvent is compatible with standard recovery and recycling systems found in most chemical manufacturing sites. Additionally, the avoidance of explosive diazo compounds and toxic gas feeds aligns perfectly with increasingly stringent environmental, health, and safety (EHS) regulations, reducing the regulatory burden and permitting timelines associated with new process validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing a realistic view of the process capabilities and limitations for potential adopters.
Q: What are the safety advantages of this synthesis method compared to traditional routes?
A: This method avoids the use of hazardous trifluorodiazoethane, utilizing safer trifluoroethylimidoyl chloride instead, significantly reducing explosion risks associated with diazo compounds.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly states the method can be expanded to the gram level and potentially larger scales due to mild reaction conditions (30°C) and commercially available starting materials.
Q: What is the source of carbon monoxide in this carbonylation reaction?
A: Carbon monoxide is generated in situ from the decomposition of the formic acid and acetic anhydride mixture, eliminating the need for handling toxic CO gas cylinders.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the palladium-catalyzed carbonylation described in CN111423381A for developing next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-trifluoromethyl imidazole intermediate meets the exacting standards required by global pharmaceutical clients, minimizing risk in your drug development timeline.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the supply of high-quality fine chemical intermediates.
