Advanced Asymmetric Synthesis of Optically Active Ketones for Commercial Pharmaceutical Manufacturing
The landscape of asymmetric synthesis is undergoing a transformative shift driven by the urgent need for greener, more efficient, and cost-effective methodologies in the production of high-value pharmaceutical intermediates. A groundbreaking development in this field is detailed in Chinese Patent CN110054553B, which discloses a highly efficient method for synthesizing optically active ketone compounds through the asymmetric conjugate addition of organoboronic acids to alpha,beta-unsaturated ketones. This technology represents a significant leap forward by utilizing novel chiral tetrabenzocyclooctatetraene compounds as organocatalysts, effectively bypassing the traditional reliance on scarce and toxic transition metals. The core innovation lies in the ability to achieve exceptional yields and enantioselectivity under remarkably mild conditions, typically at room temperature, using stable and commercially accessible boronic acid reagents. For R&D directors and process chemists, this patent offers a robust pathway to construct critical C-C bonds with high stereocontrol, addressing the persistent challenges of impurity profiles and metal residue limits in modern drug substance manufacturing.
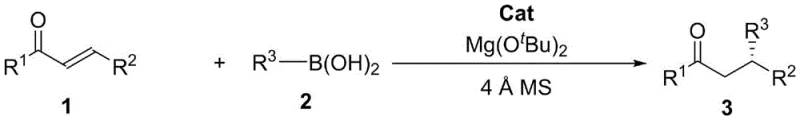
Furthermore, the versatility of this synthetic route is demonstrated by its broad substrate scope, accommodating various substituted phenyl groups, heterocycles like thienyl and furyl, and even naphthyl derivatives, making it a powerful tool for the diverse library synthesis required in early-stage drug discovery. The integration of molecular sieves and magnesium tert-butoxide additives further enhances the reaction efficiency by managing water content and activating the boron species, ensuring consistent performance across different batches. As a reliable pharmaceutical intermediate supplier, understanding and adopting such cutting-edge methodologies is crucial for maintaining a competitive edge in the global market, where purity and regulatory compliance are paramount. This report delves deep into the mechanistic nuances and commercial implications of this technology, providing a comprehensive analysis for stakeholders aiming to optimize their supply chains for complex chiral building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral ketones via conjugate addition has heavily relied on transition metal catalysis, particularly using rhodium, palladium, or copper complexes, which present substantial drawbacks for large-scale industrial applications. These traditional methods often necessitate the use of highly sensitive and unstable organometallic reagents, such as organozinc or Grignard reagents, which require stringent anhydrous conditions and cryogenic temperatures, thereby escalating energy consumption and operational complexity. Moreover, the inevitable presence of heavy metal residues in the final product poses a severe regulatory hurdle, forcing manufacturers to implement expensive and time-consuming purification protocols to meet the strict ppm limits mandated by health authorities like the FDA and EMA. The catalysts themselves are frequently air-sensitive and difficult to recover, leading to high material costs and significant waste generation, which contradicts the principles of green chemistry that are increasingly driving the pharmaceutical industry. Additionally, the limited functional group tolerance of many metal-catalyzed systems restricts their applicability to complex molecules containing sensitive moieties, often requiring tedious protection and deprotection strategies that further reduce overall process efficiency.
The Novel Approach
In stark contrast, the methodology outlined in Patent CN110054553B introduces a transition-metal-free catalytic system that leverages the unique structural properties of chiral tetrabenzocyclooctatetraene derivatives to drive the asymmetric conjugate addition with remarkable precision. This novel approach utilizes stable and non-toxic organoboronic acids as nucleophiles, which are not only inexpensive and readily available but also exhibit superior functional group compatibility, allowing for direct reactions with substrates containing esters, halides, and nitro groups without interference. The reaction proceeds smoothly at ambient temperatures (0 to 25 degrees Celsius) in common organic solvents like methyl tert-butyl ether (MTBE), eliminating the need for energy-intensive heating or cooling cycles and simplifying the engineering requirements for reactor setups. By employing a small organic molecule catalyst rather than a metal complex, the process inherently avoids heavy metal contamination, drastically simplifying the downstream purification workflow and reducing the environmental footprint of the manufacturing process. This paradigm shift from metal-based to organocatalytic systems represents a strategic advancement for cost reduction in API manufacturing, offering a cleaner, safer, and more economically viable route to high-purity chiral ketones.
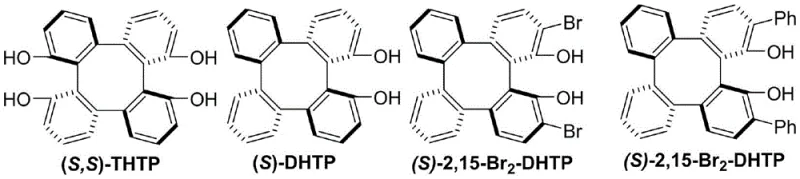
Mechanistic Insights into Chiral Tetrabenzocyclooctatetraene Catalyzed Conjugate Addition
The exceptional enantioselectivity observed in this transformation is fundamentally rooted in the sophisticated three-dimensional architecture of the chiral tetrabenzocyclooctatetraene catalyst, which creates a highly confined chiral pocket around the reactive center. The catalyst, specifically derivatives like (S,S)-THTP or (S)-2,15-Br2-DHTP, interacts with the magnesium tert-butoxide additive to form a transient chiral Lewis acid-base complex that activates the alpha,beta-unsaturated ketone substrate. This activation lowers the LUMO energy of the enone system, facilitating the nucleophilic attack by the organoboronic acid, which is simultaneously activated through coordination with the basic additive. The rigid backbone of the tetrabenzocyclooctatetraene scaffold ensures that the approach of the boronic acid is sterically directed to one specific face of the double bond, effectively discriminating between the two possible enantiomeric pathways and resulting in high optical purity. Detailed studies suggest that the hydroxyl groups on the catalyst play a critical role in hydrogen bonding networks that stabilize the transition state, further enhancing the stereochemical outcome of the reaction. This precise control over the reaction trajectory allows for the synthesis of products with enantiomeric excess (ee) values consistently exceeding 90%, and in optimized cases reaching up to 97%, which is critical for the biological activity of the resulting pharmaceutical intermediates.
From an impurity control perspective, the mechanism inherently minimizes side reactions such as 1,2-addition or homocoupling of the boronic acid, which are common pitfalls in traditional metal-catalyzed processes. The mild reaction conditions prevent thermal degradation of sensitive functional groups, ensuring a cleaner crude reaction profile that simplifies isolation. The use of 4 Angstrom molecular sieves acts as a crucial scavenger for trace water, which could otherwise hydrolyze the boronic acid or deactivate the magnesium additive, thus maintaining the integrity of the catalytic cycle throughout the reaction duration. This robustness against moisture and air, combined with the stability of the organocatalyst, translates to a process that is not only chemically elegant but also practically resilient, capable of delivering consistent quality across multiple batches. For process chemists, understanding these mechanistic underpinnings is vital for troubleshooting and optimizing the reaction parameters during scale-up, ensuring that the high selectivity observed on the milligram scale is faithfully reproduced in kilogram and ton quantities.
How to Synthesize Optically Active Ketones Efficiently
The practical implementation of this synthesis route is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment, requiring no specialized high-pressure or cryogenic apparatus. The general procedure involves charging a reaction vessel with the alpha,beta-unsaturated ketone substrate, the organoboronic acid coupling partner, the chiral catalyst, and the magnesium tert-butoxide additive under an inert atmosphere to prevent oxidation. Methyl tert-butyl ether (MTBE) is identified as the optimal solvent due to its ability to dissolve the reactants while maintaining the stability of the catalytic species, although other solvents like toluene or dichloromethane can also be employed depending on substrate solubility. The reaction mixture is then stirred at room temperature for a period ranging from 24 to 48 hours, with progress monitored by thin-layer chromatography (TLC) to ensure complete conversion of the starting material. Upon completion, the reaction is quenched with a small amount of water, and the product is isolated via standard extraction and purification techniques, typically flash column chromatography, yielding the target optically active ketone in high purity.
- Prepare the reaction vessel by adding molecular sieves, chiral catalyst Cat 1, magnesium tert-butoxide, alpha-beta-unsaturated ketone, and organoboronic acid under an inert nitrogen atmosphere.
- Add anhydrous methyl tert-butyl ether (MTBE) as the solvent and stir the mixture at 25 degrees Celsius for 24 to 48 hours until TLC analysis confirms complete consumption of the starting material.
- Quench the reaction with water, remove the solvent under reduced pressure, and purify the crude product via flash silica gel column chromatography to isolate the target optically active ketone.
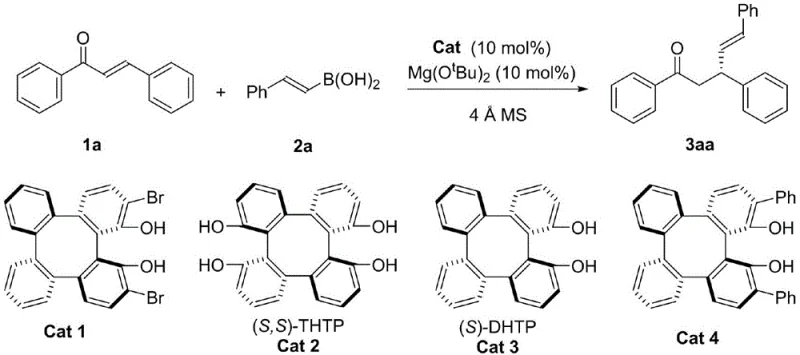
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers compelling strategic benefits that directly impact the bottom line and operational resilience of the manufacturing organization. The elimination of precious transition metals from the catalytic cycle removes a major source of cost volatility and supply risk, as the prices of metals like rhodium and palladium are subject to significant market fluctuations and geopolitical instability. By switching to an organocatalytic system based on abundant elements like carbon, hydrogen, and oxygen, companies can secure a more stable and predictable cost structure for their raw materials, insulating their production budgets from external shocks. Furthermore, the simplified downstream processing required to remove metal residues translates into significant savings in both time and resources, as expensive scavenger resins and additional purification steps are rendered unnecessary. This streamlining of the workflow not only reduces the direct cost of goods sold but also shortens the overall manufacturing cycle time, enabling faster time-to-market for new drug candidates and improving the responsiveness of the supply chain to customer demand.
- Cost Reduction in Manufacturing: The transition to a metal-free catalytic system fundamentally alters the cost dynamics of producing chiral ketones by removing the need for expensive heavy metal catalysts and the associated purification infrastructure. Without the requirement for rigorous metal scavenging processes, manufacturers can significantly reduce the consumption of specialized resins and solvents, leading to a leaner and more cost-effective production process. Additionally, the ability to run reactions at ambient temperature eliminates the energy costs associated with heating or cooling large reactors, contributing to lower utility bills and a reduced carbon footprint. The high yields and selectivity reported in the patent mean that less raw material is wasted on byproducts, maximizing the atom economy and further driving down the unit cost of the final API intermediate. These cumulative efficiencies create a robust economic case for adopting this technology, allowing companies to offer more competitive pricing to their clients while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on stable and commercially available organoboronic acids as key reagents ensures a secure and diversified supply base, mitigating the risks associated with single-source dependencies on exotic metal salts. Organoboronic acids are produced on a massive scale for Suzuki couplings and other transformations, meaning they are readily accessible from multiple global suppliers, which enhances bargaining power and reduces lead times for raw material procurement. The robustness of the reaction conditions, which tolerate a wide range of functional groups and do not require ultra-dry or oxygen-free environments beyond standard inert gas purging, makes the process less susceptible to disruptions caused by minor variations in reagent quality or environmental conditions. This operational flexibility allows for greater agility in production planning, enabling manufacturers to ramp up output quickly in response to surges in demand without compromising on product quality or consistency.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving basic quenching and chromatography, makes this process highly amenable to scale-up from gram to multi-ton scales without encountering the engineering bottlenecks typical of heterogeneous metal catalysis. The absence of toxic heavy metals simplifies waste management and disposal, aligning with increasingly stringent environmental regulations and corporate sustainability goals. This 'green' credential is becoming a key differentiator in the pharmaceutical industry, where customers are actively seeking suppliers who can demonstrate a commitment to environmentally responsible manufacturing practices. The potential for catalyst recovery and reuse, as hinted at in the patent disclosure, further enhances the sustainability profile of the process, offering a pathway to near-zero waste production. By adopting this technology, companies can future-proof their operations against tightening regulatory landscapes while appealing to eco-conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology, drawing directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of the method for potential adopters and partners in the fine chemical sector. Understanding these details is essential for making informed decisions about integrating this novel catalytic system into existing production pipelines.
Q: What are the primary advantages of this metal-free catalytic system over traditional transition metal methods?
A: This method eliminates the need for expensive and toxic transition metals like rhodium or palladium, thereby removing the costly and complex heavy metal removal steps required for pharmaceutical grade compliance, while utilizing stable and readily available organoboronic acids.
Q: How does the novel chiral catalyst influence enantioselectivity in this conjugate addition?
A: The chiral tetrabenzocyclooctatetraene catalyst creates a highly defined steric environment that directs the approach of the organoboronic acid to the alpha-beta-unsaturated ketone, achieving excellent enantiomeric excess (ee) values up to 97% under mild conditions.
Q: Is this synthesis protocol scalable for industrial production of API intermediates?
A: Yes, the protocol utilizes simple operational conditions such as ambient temperature stirring and standard workup procedures like filtration and chromatography, making it highly suitable for scale-up from laboratory to multi-ton commercial manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Ketones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the asymmetric conjugate addition technology described in Patent CN110054553B and are fully equipped to leverage it for the benefit of our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity and yields achieved in the lab are seamlessly translated to industrial manufacturing. Our state-of-the-art facilities are designed to handle sensitive organocatalytic reactions with precision, featuring rigorous QC labs that enforce stringent purity specifications to guarantee that every batch of optically active ketones meets the highest international standards. We understand that in the pharmaceutical industry, consistency is key, and our dedicated process development team works tirelessly to optimize every parameter of the synthesis to deliver a product that is not only pure but also cost-effective and reliably supplied.
We invite you to collaborate with us to explore how this advanced synthetic route can enhance your drug development pipeline and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating exactly how switching to this metal-free methodology can improve your margins. Please contact us today to request specific COA data for our chiral ketone portfolio and to discuss route feasibility assessments for your target molecules. Let NINGBO INNO PHARMCHEM be your trusted partner in navigating the complexities of modern asymmetric synthesis, delivering quality and value at every step of your supply chain.
