Advanced Asymmetric Double Conjugate Addition for High-Purity Chiral Ketone Manufacturing
The landscape of asymmetric synthesis is undergoing a significant transformation, driven by the urgent need for greener, more efficient, and metal-free methodologies in the production of complex organic molecules. Patent CN112811996B introduces a groundbreaking approach for the synthesis of optically active ketone compounds through an asymmetric double-conjugate addition reaction. This technology leverages the unique properties of chiral binaphthol or chiral tetrabenzocyclooctatetraene catalysts to facilitate the coupling of dienones with organoboronic acids. Unlike traditional methods that rely heavily on transition metals, this organocatalytic strategy offers a distinct advantage by ensuring the final product is free from toxic metal residues, a critical requirement for pharmaceutical intermediates. The invention addresses the long-standing challenge of achieving high diastereoselectivity and enantioselectivity in double addition reactions, providing a robust platform for generating chiral building blocks with exceptional purity.
Historically, the construction of C-C bonds via conjugate addition has been dominated by transition metal catalysis, which, while effective, presents substantial drawbacks for modern manufacturing. Conventional protocols often necessitate the use of expensive palladium, rhodium, or copper complexes, which not only inflate raw material costs but also introduce severe purification challenges. The removal of trace metal contaminants to meet stringent regulatory standards, such as ICH Q3D guidelines, requires additional processing steps like scavenging or recrystallization, which inevitably reduce overall yield and increase waste generation. Furthermore, previous attempts at organoboron conjugate additions, such as those reported by Sugiura et al. in 2014, often suffered from poor selectivity, predominantly yielding mono-addition products rather than the desired double-conjugate addition derivatives. This limitation restricted the utility of such reactions for synthesizing symmetrical or pseudo-symmetrical chiral ketones efficiently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on transition metal catalysts creates a bottleneck in the supply chain for high-purity fine chemicals. Beyond the economic burden of precious metals, the environmental footprint associated with mining and refining these elements is significant. In a production setting, the sensitivity of metal catalysts to air and moisture often demands rigorous exclusion techniques, utilizing gloveboxes or extensive Schlenk line operations, which complicates scale-up and increases operational expenditure. Additionally, the stability of organoboron reagents can be compromised under harsh metal-catalyzed conditions, leading to protodeboronation side reactions that lower atom economy. The inability to effectively control the stereochemistry at two distinct centers simultaneously in a double addition scenario has historically resulted in complex mixtures of diastereomers, necessitating difficult and costly chromatographic separations that are impractical for multi-kilogram production runs.
The Novel Approach
The methodology disclosed in CN112811996B circumvents these issues by employing a metal-free organocatalytic system that operates under remarkably mild conditions. By utilizing chiral binaphthol derivatives (such as Cat 5) or tetrabenzocyclooctatetraene phenols (such as Cat 9), the reaction achieves precise spatial control over the transition state. This novel approach enables the direct transformation of readily available dienones and stable organoboronic acids into complex chiral ketones in a single step. The reaction proceeds efficiently at room temperature (25°C) in common solvents like toluene, eliminating the need for energy-intensive heating or cooling cycles. As illustrated in the general reaction scheme below, the process tolerates a wide variety of functional groups, including halogens, ethers, and heterocycles, demonstrating remarkable versatility for diverse synthetic applications.
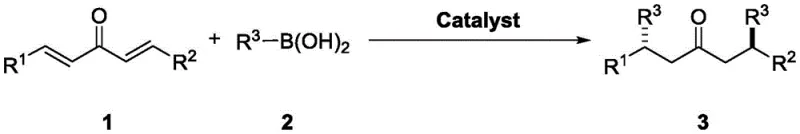
The core of this technological breakthrough lies in the sophisticated design of the chiral catalysts, which create a well-defined steric environment to guide the incoming nucleophile. The chiral binaphthol scaffold, depicted in the structural diagrams, features bulky substituents at the 3,3'-positions (such as bromine, iodine, or trifluoromethylphenyl groups) that restrict conformational freedom. This rigidity is crucial for inducing high enantioselectivity. During the catalytic cycle, the hydroxyl groups of the catalyst likely engage in hydrogen bonding with both the carbonyl oxygen of the dienone and the hydroxyl groups of the boronic acid. This dual activation mode aligns the reactants in a specific orientation that favors the formation of one enantiomer over the other. The result is a highly stereocontrolled process where the dl/meso ratio can exceed 95:5 and enantiomeric excess (ee) consistently surpasses 99%, as evidenced by the experimental data for substrates like 1a and 1b.
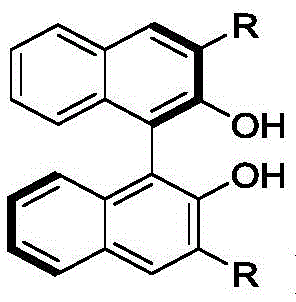
From an impurity control perspective, the absence of transition metals simplifies the impurity profile significantly. The primary byproducts are typically unreacted starting materials or minor mono-addition intermediates, which are easily separated via standard silica gel chromatography. The use of molecular sieves (4Å or 5Å) as additives plays a pivotal role in driving the equilibrium forward by sequestering water produced during the transmetallation or activation steps, thereby preventing the hydrolysis of the boronic acid and maintaining catalyst activity. This mechanistic feature ensures that the reaction proceeds to completion with high conversion rates, often exceeding 90% isolated yield. For R&D teams, this means a cleaner crude product that requires less downstream processing, directly translating to higher throughput and reduced solvent consumption during purification.
Mechanistic Insights into Chiral Binaphthol-Catalyzed Conjugate Addition
The exceptional performance of this system can be attributed to the cooperative activation mechanism facilitated by the chiral phosphoric acid-like environment of the binaphthol catalysts. Although technically phenols, in the presence of Lewis basic additives or under specific conditions, they mimic the behavior of chiral Brønsted acids. The catalyst activates the electrophilic dienone by coordinating with the carbonyl group, increasing its susceptibility to nucleophilic attack. Simultaneously, the organoboronic acid is activated, enhancing its nucleophilicity without the need for a separate base that might cause racemization. This concerted activation lowers the activation energy barrier for the C-C bond formation while maintaining a tight chiral pocket. The steric bulk of the catalyst substituents, such as the 3,5-bis(trifluoromethyl)phenyl groups in Cat 5, effectively blocks one face of the dienone, forcing the boronic acid to attack from the less hindered trajectory. This precise geometric control is what allows for the simultaneous establishment of two chiral centers with such high fidelity.
How to Synthesize Optically Active Ketones Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is straightforward and relies on standard organic synthesis techniques. The protocol typically involves charging a reaction vessel with the chiral catalyst and molecular sieves under an inert atmosphere, followed by the addition of the dienone and organoboronic acid substrates. Dry toluene is introduced as the solvent, and the mixture is stirred at ambient temperature. The reaction progress is monitored using thin-layer chromatography (TLC) until the starting dienone is fully consumed. Upon completion, the mixture is filtered to remove the molecular sieves and catalyst residues, and the solvent is evaporated under reduced pressure. The crude product is then purified via flash column chromatography to afford the target optically active ketone. A representative example of this procedure, converting dienone 1a and boronic acid 2a into product 3aa, is shown below, highlighting the simplicity and effectiveness of the workflow.
- Prepare the reaction vessel by adding molecular sieves (4Å or 5Å) and the chiral catalyst (e.g., Cat 5 or Cat 9) under an inert nitrogen atmosphere.
- Introduce the dienone substrate and the organoboronic acid reagent into the vessel, ensuring a molar ratio of approximately 1: 3 for optimal conversion.
- Add dry toluene as the solvent and stir the mixture at 25°C for 24 hours, monitoring progress via TLC until the starting material is fully consumed.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free catalytic technology offers compelling strategic benefits that extend beyond mere technical performance. The elimination of precious metal catalysts represents a direct and significant reduction in raw material costs, shielding the manufacturing process from the volatility of the global platinum and palladium markets. Furthermore, the use of organoboronic acids as coupling partners is advantageous because these reagents are commercially abundant, stable towards air and moisture, and generally inexpensive compared to organozinc or organotin alternatives. This stability simplifies logistics and storage requirements, reducing the risk of reagent degradation during transport and allowing for more flexible inventory management. The robustness of the reaction conditions also means that specialized equipment for handling pyrophoric or highly sensitive reagents is not required, lowering capital expenditure for new production lines.
- Cost Reduction in Manufacturing: The economic impact of removing transition metals from the synthesis cannot be overstated. By avoiding expensive metal salts and the associated ligands, the direct material cost per kilogram of product is drastically lowered. Moreover, the simplified purification process eliminates the need for specialized metal scavenging resins or multiple recrystallization steps designed to meet residual metal specifications. This streamlining of the downstream processing workflow reduces solvent usage, labor hours, and waste disposal costs. The high yields and selectivity reported in the patent mean that less starting material is wasted, improving the overall atom economy and further driving down the cost of goods sold (COGS) for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and stable reagents. Organoboronic acids are produced on a massive scale for Suzuki couplings, ensuring a secure and competitive supply base with multiple qualified vendors globally. The catalysts themselves, based on binaphthol scaffolds, can be synthesized from readily available chiral pool materials or purchased from established suppliers, mitigating the risk of single-source bottlenecks. The mild reaction conditions (25°C, atmospheric pressure) reduce the dependency on complex utility systems like cryogenic cooling or high-pressure steam, making the process easier to transfer between different manufacturing sites or contract manufacturing organizations (CMOs) without significant re-engineering.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is facilitated by its operational simplicity and safety profile. The absence of toxic heavy metals aligns perfectly with green chemistry principles and increasingly strict environmental regulations regarding heavy metal discharge in wastewater. This compliance reduces the regulatory burden and permitting time for new facilities. The reaction generates minimal hazardous waste, primarily consisting of organic solvents that can be recovered and recycled, and spent molecular sieves that are non-hazardous. This environmentally friendly profile enhances the sustainability credentials of the supply chain, a factor that is becoming increasingly important for downstream pharmaceutical customers aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric double conjugate addition technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on substrate compatibility, catalyst loading, and product quality. Understanding these nuances is essential for evaluating the feasibility of this route for specific target molecules in your pipeline.
Q: Does this synthesis method involve transition metal catalysts?
A: No, the method described in patent CN112811996B utilizes chiral organic small molecule catalysts, specifically binaphthol or tetrabenzocyclooctatetraene derivatives, eliminating the risk of heavy metal contamination in the final product.
Q: What kind of stereoselectivity can be achieved with this protocol?
A: The process demonstrates excellent stereocontrol, consistently achieving enantiomeric excess (ee) values greater than 99% and high diastereoselectivity (dl/meso ratios often exceeding 95:5) across a broad range of substrates.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, the reaction operates at mild temperatures (0-30°C) using common solvents like toluene and stable reagents like organoboronic acids, making it highly scalable and safe for industrial production without requiring extreme pressure or cryogenic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Ketones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the asymmetric double conjugate addition technology described in CN112811996B for the production of high-value chiral intermediates. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate this innovative academic research into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high enantioselectivity and yields observed in the lab are maintained during large-scale campaigns. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to evaluate the feasibility of this metal-free route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that compares this organocatalytic approach against your current metal-dependent methods. By leveraging our supply chain networks and process optimization skills, we can help you secure a reliable source of high-purity optically active ketones while significantly reducing your overall manufacturing costs. Please contact us to request specific COA data, route feasibility assessments, and a comprehensive proposal tailored to your volume requirements.
