Advanced Palladium-Catalyzed Bis-Carbonylation for Scalable Pyrrol-2-One Production
Introduction to Next-Generation Pyrrol-2-One Synthesis
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a privileged scaffold in medicinal chemistry, serving as the core backbone for numerous bioactive natural products and pharmaceutical agents. As illustrated in the provided structural analysis, this heterocyclic system is integral to the efficacy of compounds such as Althiomycin, a potent antibacterial agent, Glimepiride, a widely prescribed hypoglycemic drug, and Isomalyngamide A, which demonstrates significant anticancer potential. 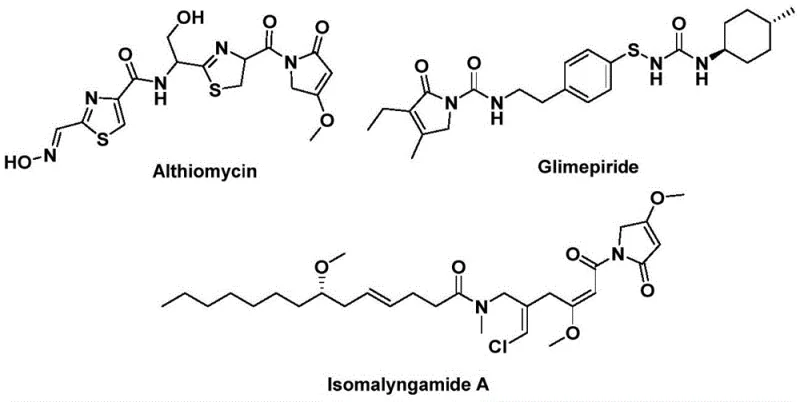 . Recognizing the critical importance of this pharmacophore, recent advancements detailed in patent CN112694430B have introduced a transformative preparation method that addresses long-standing synthetic challenges. This novel approach leverages a palladium-catalyzed bis-carbonylation strategy, utilizing readily available propargylamines and benzyl chlorides to construct the five-membered lactam ring with exceptional efficiency. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate supplier partnerships, this technology offers a robust pathway to access high-value scaffolds that were previously difficult to manufacture at scale.
. Recognizing the critical importance of this pharmacophore, recent advancements detailed in patent CN112694430B have introduced a transformative preparation method that addresses long-standing synthetic challenges. This novel approach leverages a palladium-catalyzed bis-carbonylation strategy, utilizing readily available propargylamines and benzyl chlorides to construct the five-membered lactam ring with exceptional efficiency. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate supplier partnerships, this technology offers a robust pathway to access high-value scaffolds that were previously difficult to manufacture at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of the 1,5-dihydro-2H-pyrrol-2-one skeleton has relied on multi-step sequences that often suffer from poor atom economy and harsh reaction conditions. Classical routes frequently involve the use of toxic gaseous carbon monoxide under high pressure, posing significant safety risks and requiring specialized equipment that increases capital expenditure for manufacturing facilities. Furthermore, conventional cyclization strategies often exhibit limited substrate scope, failing to tolerate sensitive functional groups such as halogens or trifluoromethyl moieties which are increasingly common in modern drug design. These limitations result in lower overall yields, complex purification protocols, and extended production timelines, creating bottlenecks in the supply chain for cost reduction in pharmaceutical intermediates manufacturing. The reliance on unstable reagents and the generation of substantial chemical waste further complicate environmental compliance, making traditional methods less attractive for sustainable commercial production.
The Novel Approach
In stark contrast, the methodology disclosed in the patent introduces a streamlined, one-pot synthesis that dramatically simplifies the operational complexity while enhancing safety profiles. By employing 1,3,5-tricarboxylic acid phenol ester as a solid carbon monoxide substitute, the process eliminates the hazards associated with handling gaseous CO, thereby facilitating safer scale-up operations. The reaction utilizes a palladium catalyst system comprising Pd(OAc)2 and DPPF ligand in the presence of triethylamine, enabling the direct coupling of propargylamines and benzyl chlorides. This innovative route not only achieves high conversion rates under relatively mild thermal conditions (100-120°C) but also demonstrates remarkable versatility across a wide range of substrates. The ability to synthesize the target lactam in a single step from cheap and commercially available starting materials represents a paradigm shift, offering substantial cost savings and improved throughput for industrial applications.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation
The success of this synthetic transformation lies in the intricate catalytic cycle driven by the palladium complex, which orchestrates two distinct carbonylation events followed by cyclization. The mechanism initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride, generating a reactive benzyl-palladium intermediate. Subsequently, carbon monoxide, released in situ from the thermal decomposition of the phenol ester surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the propargylamine, leading to the formation of a five-membered ring palladium complex. 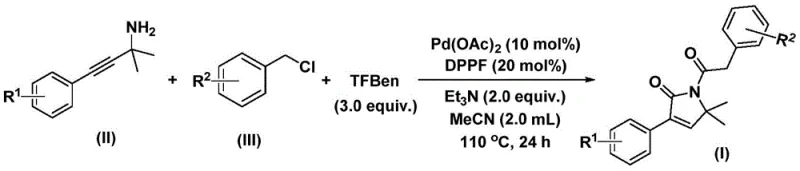 . A second insertion of carbon monoxide occurs, expanding the coordination sphere to a six-membered ring palladium intermediate, which finally undergoes reductive elimination to release the desired 1,5-dihydro-2H-pyrrole-2-ketone product and regenerate the active catalyst. This dual-carbonylation pathway is highly efficient, ensuring that the carbonyl groups are precisely installed to form the lactam functionality without requiring external pressurized gas sources.
. A second insertion of carbon monoxide occurs, expanding the coordination sphere to a six-membered ring palladium intermediate, which finally undergoes reductive elimination to release the desired 1,5-dihydro-2H-pyrrole-2-ketone product and regenerate the active catalyst. This dual-carbonylation pathway is highly efficient, ensuring that the carbonyl groups are precisely installed to form the lactam functionality without requiring external pressurized gas sources.
From an impurity control perspective, the choice of ligands and the specific reaction conditions play a pivotal role in minimizing side reactions. The use of DPPF as a bidentate ligand stabilizes the palladium center, preventing premature catalyst deactivation or the formation of homocoupling byproducts. Furthermore, the controlled release of carbon monoxide from the solid surrogate ensures a steady concentration of CO in the reaction medium, which suppresses competing pathways such as simple amidation or alkyne polymerization. The compatibility of the system with various electronic environments on the aromatic rings—ranging from electron-donating methoxy groups to strong electron-withdrawing trifluoromethyl groups—indicates a robust catalytic cycle that maintains high selectivity. This mechanistic stability is crucial for producing high-purity OLED material or pharmaceutical intermediates where trace impurities can compromise downstream biological activity or material performance.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-Ketone Efficiently
The practical implementation of this synthesis is designed for ease of execution in standard laboratory and pilot plant settings. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, and the two primary organic substrates in acetonitrile solvent. The mixture is then heated to promote the cascade reaction, after which standard workup procedures involving filtration and silica gel chromatography yield the pure product.
- Combine palladium acetate, DPPF ligand, triethylamine, 1,3,5-tricarboxylic acid phenol ester (CO source), propargylamine, and benzyl chloride in acetonitrile.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 1,5-dihydro-2H-pyrrole-2-ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits regarding cost structure and supply reliability. The shift from hazardous gaseous reagents to stable solid surrogates significantly reduces the regulatory burden and safety infrastructure costs associated with manufacturing. Moreover, the use of commodity chemicals like benzyl chlorides and propargylamines as starting materials ensures a stable and diversified supply base, mitigating the risk of raw material shortages that often plague specialty chemical production. The simplified one-pot nature of the reaction reduces the number of unit operations required, leading to lower energy consumption and reduced solvent usage, which aligns with green chemistry principles and lowers waste disposal costs.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the use of inexpensive, commercially available catalysts and ligands drastically lower the capital and operational expenditures. By consolidating multiple synthetic steps into a single transformation, the process minimizes labor hours and intermediate isolation losses, resulting in a more economical production profile. The high reaction efficiency observed across diverse substrates means that less raw material is wasted on failed batches or low-yielding steps, directly improving the cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Since the key starting materials are bulk chemicals with established global supply chains, manufacturers are not dependent on niche suppliers for exotic reagents. This accessibility ensures consistent production schedules and shorter lead times for high-purity pharmaceutical intermediates. The robustness of the reaction conditions allows for flexible manufacturing planning, as the process is less sensitive to minor variations in temperature or stoichiometry compared to traditional high-pressure carbonylations, thereby guaranteeing continuous supply continuity for downstream clients.
- Scalability and Environmental Compliance: The avoidance of toxic carbon monoxide gas simplifies the environmental permitting process and reduces the need for specialized scrubbing systems. The reaction generates minimal hazardous waste, and the use of acetonitrile, a common recyclable solvent, facilitates solvent recovery programs. This environmental friendliness supports corporate sustainability goals and ensures compliance with increasingly stringent international environmental regulations, making the commercial scale-up of complex pharmaceutical intermediates smoother and more socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. Understanding these details helps partners evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the carbon monoxide source in this synthesis?
A: The process utilizes 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid, safe carbon monoxide substitute, eliminating the need for handling hazardous CO gas cylinders.
Q: Does this method tolerate electron-withdrawing groups?
A: Yes, the catalytic system exhibits excellent substrate compatibility, successfully accommodating substituents such as fluorine, chlorine, bromine, and trifluoromethyl groups on the aromatic rings.
Q: What are the typical reaction conditions?
A: The reaction typically proceeds in acetonitrile at 110°C for 24 hours using Pd(OAc)2 and DPPF as the catalytic system with triethylamine as the base.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale innovation to industrial reality is seamless. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in palladium-catalyzed transformations allows us to optimize this specific bis-carbonylation route for maximum yield and minimal impurity formation, providing our clients with a competitive edge in the marketplace.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this novel synthetic route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on the foundation of cutting-edge chemistry and reliable manufacturing excellence.
