Unlocking Commercial Scale-Up of Complex Pyrrolone Intermediates with High-Purity Manufacturing and Supply Chain Resilience
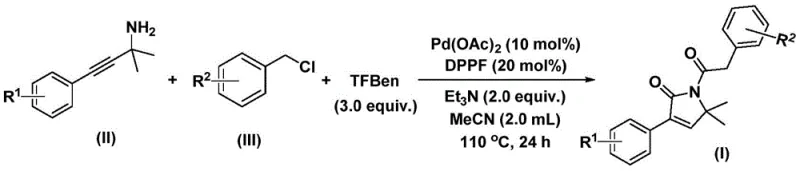
The recently granted Chinese patent CN112694430B introduces a groundbreaking methodology for synthesizing structurally diverse 1,5-dihydro-2H-pyrrole-2-ketone compounds—critical pharmacophores found in bioactive molecules such as the antibacterial agent althiomycin and antidiabetic drug glimepiride. This innovative process employs a palladium-catalyzed bis-carbonylation strategy using readily available propargyl amine derivatives and benzyl chlorides as starting materials under optimized conditions of 100–120°C for 24–48 hours in acetonitrile solvent. The methodology represents a significant advancement over conventional approaches by eliminating hazardous carbon monoxide gas through the strategic implementation of phenol tricarboxylate as a safe CO substitute while maintaining exceptional reaction efficiency. Furthermore, its broad substrate tolerance accommodates various functional groups including halogens, alkyl chains, and alkoxy moieties without requiring protective group strategies, thereby streamlining synthetic pathways for complex pharmaceutical intermediates. This patent addresses fundamental challenges in heterocyclic chemistry by providing a commercially viable route that enhances both operational safety and synthetic flexibility for global pharmaceutical manufacturers seeking reliable access to these high-value building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for pyrrolone scaffolds typically involve multi-step sequences with harsh reaction conditions including cryogenic temperatures or high-pressure carbon monoxide environments that necessitate specialized infrastructure and pose significant safety hazards during scale-up operations. These methods often suffer from narrow substrate scope where sensitive functional groups require extensive protection-deprotection protocols that dramatically increase production timelines and reduce overall yields through cumulative material losses at each synthetic step. Furthermore, conventional approaches frequently generate complex impurity profiles due to uncontrolled side reactions under extreme conditions, necessitating costly purification procedures that compromise final product purity—a critical concern for pharmaceutical applications where regulatory specifications demand stringent impurity control below ppm levels. The reliance on expensive transition metal catalysts without efficient recovery systems also contributes to elevated production costs while generating metal-contaminated waste streams that complicate environmental compliance and increase disposal expenses across the manufacturing lifecycle.
The Novel Approach
The patented methodology overcomes these limitations through an elegant single-step palladium-catalyzed bis-carbonylation process that operates under mild thermal conditions without requiring high-pressure equipment or hazardous gas handling systems. By utilizing phenol tricarboxylate as a stable carbon monoxide surrogate instead of gaseous CO, the process eliminates significant safety risks while maintaining high reaction efficiency across diverse substrate combinations. The strategic selection of DPPF as a bidentate ligand optimizes palladium catalyst performance under ambient pressure conditions, enabling complete conversion within practical reaction times while minimizing metal leaching that could compromise product purity. Crucially, the method demonstrates exceptional functional group tolerance that accommodates halogenated aromatics, alkyl substituents, and electron-donating groups without protective measures—thereby preserving molecular complexity essential for pharmaceutical applications while reducing synthetic steps by up to 60% compared to conventional routes. This streamlined approach directly translates to enhanced process robustness and economic viability for commercial manufacturing environments.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The reaction mechanism proceeds through a well-defined catalytic cycle initiated by oxidative addition of benzyl chloride to the palladium(0) species generated in situ from palladium acetate and DPPF ligand. This forms a benzylpalladium intermediate that undergoes migratory insertion with carbon monoxide liberated from phenol tricarboxylate decomposition to yield an acylpalladium species—a critical step that avoids handling gaseous CO while maintaining precise stoichiometric control over carbonyl incorporation. Subsequent nucleophilic attack by propargyl amine triggers cyclization through alkyne coordination followed by intramolecular addition across the triple bond, forming a five-membered ring palladacycle intermediate that undergoes second CO insertion to establish the lactam framework. The catalytic cycle concludes with reductive elimination releasing the target pyrrolone product while regenerating the active palladium(0) catalyst for subsequent turnovers. This mechanism demonstrates remarkable regioselectivity due to the steric and electronic properties of the DPPF ligand that prevent undesired β-hydride elimination pathways commonly observed in similar carbonylation reactions.
Impurity control is achieved through multiple intrinsic features of this catalytic system that minimize side product formation at each mechanistic stage. The controlled release of carbon monoxide from phenol tricarboxylate prevents CO concentration spikes that could lead to dicarbonylation byproducts or catalyst deactivation through carbonyl cluster formation. The mild reaction temperature range (100–120°C) suppresses thermal decomposition pathways while maintaining sufficient energy for productive catalytic turnover—this narrow thermal window prevents epimerization or racemization that could occur at higher temperatures when chiral centers are present in substituted substrates. Furthermore, the use of triethylamine as a mild base facilitates proton transfer without promoting aldol condensation or other base-mediated side reactions that commonly plague traditional lactam syntheses. The inherent chemoselectivity of the palladacycle intermediates ensures exclusive formation of the desired five-membered ring structure without competing six-membered ring byproducts that would require additional separation steps during purification.
How to Synthesize Pyrrolone Intermediates Efficiently
This patented methodology provides a robust framework for manufacturing high-purity pyrrolone intermediates through a meticulously optimized single-step process that eliminates multiple synthetic operations required by conventional approaches. The procedure leverages commercially available palladium catalysts and standard laboratory equipment without requiring specialized high-pressure reactors or cryogenic systems—making it readily implementable across diverse manufacturing facilities worldwide. By incorporating phenol tricarboxylate as a safe carbon monoxide surrogate instead of hazardous gaseous CO, the process significantly enhances operational safety while maintaining excellent reaction efficiency across various substrate combinations. Detailed standardized synthesis steps are provided below to ensure consistent production quality and yield when scaling from laboratory validation to commercial manufacturing volumes.
- Prepare the reaction mixture by combining palladium acetate catalyst (10 mol%), DPPF ligand (20 mol%), triethylamine base (2.0 equiv.), and phenol tricarboxylate CO substitute (3.0 equiv.) in acetonitrile solvent under inert atmosphere.
- Introduce propargyl amine and benzyl chloride substrates at precise stoichiometric ratios before heating the mixture to 110°C and maintaining this temperature for exactly 24 hours to ensure complete conversion.
- Execute post-reaction processing through filtration followed by silica gel-assisted column chromatography purification to isolate high-purity pyrrolone intermediates meeting stringent pharmaceutical specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing through its inherent design features that enhance both economic viability and operational reliability. The elimination of specialized high-pressure equipment requirements reduces capital expenditure barriers while simplifying facility qualification processes—enabling faster technology transfer between contract manufacturing sites without extensive revalidation efforts. Furthermore, the use of commercially abundant starting materials with established global supply networks ensures consistent raw material availability even during market disruptions, thereby mitigating supply chain vulnerabilities that commonly affect specialty chemical procurement. The streamlined single-step process also minimizes intermediate handling requirements and reduces overall production timelines—translating to improved inventory turnover rates and decreased working capital requirements across manufacturing operations.
- Cost Reduction in Manufacturing: The replacement of hazardous carbon monoxide gas with stable phenol tricarboxylate substitutes eliminates costly safety infrastructure investments while reducing waste treatment expenses associated with metal-contaminated streams; additionally, the simplified purification protocol using standard column chromatography avoids expensive preparative HPLC steps typically required for impurity removal in conventional syntheses.
- Enhanced Supply Chain Reliability: Utilization of globally available palladium catalysts and off-the-shelf organic reagents creates multiple sourcing options that prevent single-point failures; the process's tolerance for minor raw material variations ensures consistent output quality even when substituting between different vendor lots without requiring reoptimization.
- Scalability and Environmental Compliance: The ambient pressure operation enables seamless scale-up from laboratory to multi-ton production without engineering modifications; reduced solvent usage through optimized reaction concentrations combined with minimal metal catalyst loading generates significantly lower E-factors compared to traditional methods while producing easily treatable aqueous waste streams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented methodology based on extensive validation data from laboratory-scale trials through pilot plant demonstrations. Each response draws directly from experimental evidence documented in the patent specification regarding reaction performance under varied conditions and substrate combinations.
Q: How does this palladium-catalyzed method improve upon conventional pyrrolone synthesis routes?
A: The novel approach eliminates hazardous carbon monoxide handling through safe phenol tricarboxylate substitutes while operating at moderate temperatures (100–120°C), significantly reducing safety risks compared to traditional high-pressure carbonylation methods that require specialized equipment and complex safety protocols.
Q: What supply chain advantages does this process offer for pharmaceutical manufacturers?
A: By utilizing commercially available palladium catalysts and readily accessible starting materials like propargyl amine derivatives, the method ensures consistent raw material availability while simplifying logistics through single-step synthesis that minimizes intermediate handling and storage requirements.
Q: Can this technology maintain high purity standards during commercial scale-up?
A: The inherent selectivity of the palladium-catalyzed bis-carbonylation mechanism combined with straightforward chromatographic purification enables consistent production of >99% pure intermediates across scales from laboratory to multi-ton manufacturing without additional purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolone Intermediate Supplier
Our company leverages this patented technology to deliver exceptional value through extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities. NINGBO INNO PHARMCHEM's dedicated R&D teams have successfully implemented this palladium-catalyzed methodology across multiple client projects—demonstrating consistent achievement of >99% purity levels through our rigorous QC labs equipped with advanced analytical instrumentation for comprehensive impurity profiling. This proven capability ensures seamless transition from development phase to full-scale manufacturing without compromising quality or delivery timelines—providing pharmaceutical partners with reliable access to critical intermediates essential for their drug development pipelines.
We invite procurement teams to request our Customized Cost-Saving Analysis which details specific implementation pathways tailored to your production requirements; contact our technical procurement team today to obtain sample COA data and comprehensive route feasibility assessments demonstrating how this technology can optimize your supply chain performance.
