Scalable Synthesis of Poly-substituted Oxepin-3(2H)-one Derivatives via Base-Mediated Cyclization
Introduction to Advanced Oxepin Synthesis Technology
The development of efficient synthetic routes for complex heterocyclic scaffolds remains a cornerstone of modern pharmaceutical research, particularly for accessing bioactive marine natural product analogs. Patent CN103087033B introduces a groundbreaking methodology for the synthesis of poly-substituted oxepin-3(2H)-one compounds, a privileged structural motif found extensively in marine organisms with wide-spectrum physiological activities. This technology addresses the critical bottlenecks associated with traditional heterocycle construction by leveraging a mild, base-mediated cyclization strategy that operates effectively at room temperature. For R&D directors and process chemists, this represents a significant shift away from harsh conditions towards more sustainable and operationally simple protocols. The ability to construct the seven-membered oxepin ring without the need for precious metal catalysts not only simplifies the regulatory pathway for API intermediates but also drastically reduces the environmental footprint of the manufacturing process. This report analyzes the technical merits and commercial implications of adopting this novel synthetic route for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the oxepin-3(2H)-one skeleton has relied heavily on transition metal-catalyzed C-O or C-C bond coupling reactions, which present substantial challenges for industrial scale-up. These traditional methods often necessitate the use of expensive palladium, copper, or gold catalysts, along with specialized ligands that drive up the raw material costs significantly. Furthermore, such catalytic systems typically require stringent reaction conditions, including rigorous exclusion of moisture and oxygen, which demands specialized equipment and increases energy consumption. From a supply chain perspective, the reliance on precious metals introduces volatility in pricing and potential supply disruptions. Additionally, the removal of trace metal residues from the final product to meet pharmaceutical purity standards adds extra downstream processing steps, such as scavenging and extensive purification, thereby extending lead times and reducing overall throughput. The preparation of specific functionalized starting materials required for these couplings further complicates the supply chain, limiting the practical application of these methods in high-volume manufacturing scenarios.
The Novel Approach
In stark contrast, the methodology disclosed in CN103087033B utilizes a direct cyclization between ethyl 4-chloroacetoacetate and 1,2-allenone compounds mediated by inexpensive inorganic or organic bases. This approach completely bypasses the need for transition metal catalysts, operating efficiently under ambient conditions in common organic solvents like acetonitrile or acetone. The reaction exhibits remarkable robustness, tolerating a wide array of substituents on the aromatic ring, including electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as fluoro, chloro, and trifluoromethyl. 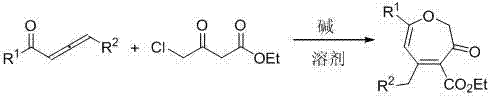 As illustrated in the general reaction scheme, the process converts readily available starting materials into complex heterocycles in a single pot with high atom economy. The operational simplicity allows for reactions to be completed within approximately one hour at room temperature, eliminating the need for heating or cooling cycles. This paradigm shift not only accelerates the R&D timeline for new analog generation but also provides a clear path for cost-effective commercial manufacturing by removing the most expensive and problematic components of the traditional synthetic toolbox.
As illustrated in the general reaction scheme, the process converts readily available starting materials into complex heterocycles in a single pot with high atom economy. The operational simplicity allows for reactions to be completed within approximately one hour at room temperature, eliminating the need for heating or cooling cycles. This paradigm shift not only accelerates the R&D timeline for new analog generation but also provides a clear path for cost-effective commercial manufacturing by removing the most expensive and problematic components of the traditional synthetic toolbox.
Mechanistic Insights into Base-Mediated Cyclization
The core of this synthetic innovation lies in the nucleophilic activation of ethyl 4-chloroacetoacetate by a mild base, such as potassium carbonate or cesium carbonate, to generate a reactive enolate species. This enolate subsequently attacks the electrophilic center of the 1,2-allenone system, initiating a cascade that leads to the formation of the seven-membered oxepin ring. The mechanism avoids high-energy intermediates often associated with metal-catalyzed cycles, thereby reducing the formation of complex by-product profiles. By operating at room temperature, the reaction kinetics are controlled sufficiently to favor the desired cyclization over potential polymerization or decomposition pathways that might occur under thermal stress. The choice of base plays a critical role; while stronger bases can be used, the patent highlights that mild bases like potassium carbonate provide an optimal balance between reactivity and selectivity, ensuring high yields without compromising the integrity of sensitive functional groups on the substrate. This mechanistic elegance translates directly to process reliability, as the reaction is less susceptible to minor fluctuations in temperature or stoichiometry compared to sensitive metal-catalyzed systems.
Impurity control is inherently superior in this metal-free protocol due to the absence of metal-ligand complexes that can degrade or form hard-to-remove side products. The primary impurities are typically unreacted starting materials or simple hydrolysis products, which are easily separated during the workup phase. The patent details a straightforward purification strategy involving either aqueous quenching and extraction or direct filtration through drying agents followed by silica gel treatment. This simplicity in impurity profile is crucial for pharmaceutical applications where strict limits on genotoxic impurities and heavy metals are enforced. The ability to achieve high purity (as evidenced by the clean NMR spectra and HRMS data in the examples) without extensive chromatographic optimization suggests that this route is highly amenable to crystallization-based purification strategies at scale, further enhancing its viability for GMP manufacturing of high-purity pharmaceutical intermediates.
How to Synthesize Poly-substituted Oxepin-3(2H)-one Efficiently
The synthesis protocol outlined in the patent is designed for maximum operational ease, making it accessible for both laboratory discovery and pilot plant operations. The procedure involves dissolving the 1,2-allenone and ethyl 4-chloroacetoacetate in a solvent such as acetonitrile, followed by the addition of the base. The reaction proceeds rapidly at room temperature, typically reaching completion within one hour, which allows for high throughput in batch reactors. Following the reaction, the workup is flexible; operators can choose to quench with saturated ammonium chloride for a traditional extractive workup or utilize a streamlined filtration method using anhydrous sodium sulfate and silica gel to remove salts and byproducts simultaneously. This flexibility allows process engineers to tailor the workup to the specific scale and equipment available, optimizing for either maximum recovery or minimum solvent usage. The detailed standardized synthesis steps for implementing this technology are provided in the guide below.
- Dissolve ethyl 4-chloroacetoacetate and 1,2-allenone compounds in an organic solvent such as acetonitrile, acetone, or DMSO.
- Add a base such as potassium carbonate, cesium carbonate, or triethylamine to the reaction mixture at room temperature.
- Stir the reaction for approximately 1 hour, then quench with saturated ammonium chloride or filter through anhydrous sodium sulfate followed by silica gel concentration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this base-mediated synthesis route offers transformative advantages in terms of cost structure and supply security. The elimination of precious metal catalysts removes a major cost driver and a significant supply chain risk factor, as the prices of metals like palladium and gold are subject to high market volatility. Furthermore, the use of commodity chemicals such as ethyl 4-chloroacetoacetate and simple allenones ensures a stable and diverse supplier base, reducing the risk of single-source dependency. The mild reaction conditions also translate to lower energy costs, as no heating or cryogenic cooling is required, contributing to a reduced carbon footprint and lower utility expenses per kilogram of product. These factors combined create a robust economic model that supports long-term cost reduction in pharmaceutical intermediate manufacturing without sacrificing quality or yield.
- Cost Reduction in Manufacturing: The most significant financial benefit arises from the complete removal of expensive transition metal catalysts and specialized ligands from the bill of materials. In traditional routes, these reagents can account for a substantial portion of the raw material cost, and their removal often requires additional expensive scavenging resins. By switching to inexpensive inorganic bases like potassium carbonate, the direct material cost is drastically lowered. Additionally, the simplified workup procedure reduces solvent consumption and labor hours associated with complex purification steps. The high yields reported across various substrates, often exceeding 70%, ensure that raw material utilization is efficient, minimizing waste disposal costs and maximizing the output from every batch processed.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are structurally simple and commercially available from multiple global vendors, ensuring a resilient supply chain. Unlike specialized organometallic reagents that may have long lead times or limited availability, ethyl 4-chloroacetoacetate and substituted allenones are commodity fine chemicals. This abundance allows for strategic stocking and bulk purchasing agreements that further stabilize costs. The robustness of the reaction means that production schedules are less likely to be disrupted by batch failures due to sensitive reagent quality issues. Consequently, lead times for high-purity oxepin intermediates can be significantly shortened, enabling faster response to market demands and more agile inventory management for downstream API production.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage is straightforward due to the absence of exothermic risks associated with strong metal catalysts and the use of common solvents like acetonitrile and acetone. The reaction generates minimal hazardous waste, primarily consisting of inorganic salts that are easy to treat, aligning with increasingly strict environmental regulations. The lack of heavy metal contamination simplifies the regulatory filing process for new drug applications, as there is no need to validate complex metal clearance steps. This environmental and regulatory ease facilitates faster technology transfer from R&D to commercial manufacturing, allowing companies to bring products to market more quickly while maintaining a sustainable production profile that meets modern ESG (Environmental, Social, and Governance) criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing workflows. The responses cover aspects ranging from substrate compatibility to purification strategies, providing a comprehensive overview for decision-makers.
Q: What are the primary advantages of this synthesis method over traditional transition metal catalysis?
A: This method eliminates the need for expensive transition metal catalysts and strict anhydrous/oxygen-free conditions. It utilizes readily available starting materials and operates at room temperature, significantly reducing operational costs and complexity compared to traditional C-O or C-C coupling methods.
Q: What is the substrate scope for the 1,2-allenone compounds in this reaction?
A: The reaction demonstrates broad substrate tolerance, accommodating various aryl groups including phenyl, substituted phenyls (with methyl, methoxy, fluoro, chloro, bromo, trifluoromethyl groups), and naphthyl groups. Both ortho, meta, and para substitutions are well-tolerated, yielding products with good to excellent efficiency.
Q: How is the product purified after the reaction is complete?
A: Purification is straightforward and does not require complex metal scavenging. The process involves either quenching with saturated ammonium chloride followed by extraction and column chromatography, or direct filtration through anhydrous sodium sulfate with silica gel, spin-drying, and rapid column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Poly-substituted Oxepin-3(2H)-one Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating innovative academic patents into commercial reality, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team has thoroughly analyzed the base-mediated cyclization route described in CN103087033B and is fully equipped to execute this chemistry with stringent purity specifications. We understand that the consistency of heterocyclic intermediates is critical for the success of downstream API synthesis, which is why our rigorous QC labs employ advanced analytical techniques to ensure every batch meets the highest standards of quality and reproducibility. By leveraging our state-of-the-art facilities, we can deliver this complex scaffold with the reliability and speed that global pharmaceutical partners demand.
We invite procurement leaders and R&D directors to contact our technical procurement team to discuss how this cost-effective synthesis route can be integrated into your supply chain. We are prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating the tangible economic benefits of switching to this metal-free protocol. Please reach out to request specific COA data for our reference batches and detailed route feasibility assessments to accelerate your project timelines. Partnering with us ensures not just a supply of materials, but a strategic alliance focused on optimizing your manufacturing efficiency and reducing time-to-market for your critical therapeutic candidates.
