Advanced Metal-Free Synthesis of Oxepin-3(2H)-one Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with economic efficiency. Patent CN103087033A introduces a groundbreaking method for the synthesis of poly-substituted oxepin-3(2H)-one compounds, a structural motif prevalent in marine natural products with significant biological activity. This technology represents a paradigm shift from traditional transition metal-catalyzed processes to a more sustainable, base-mediated approach. By leveraging simple starting materials like ethyl 4-chloroacetoacetate and 1,2-allenone compounds, this method achieves high yields under remarkably mild conditions. For R&D directors and procurement managers, this patent offers a viable pathway to produce high-purity pharmaceutical intermediates without the burden of heavy metal residues or complex reaction setups. The implications for supply chain stability and cost reduction in fine chemical manufacturing are profound, as it removes dependencies on scarce catalytic resources.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the oxepin-3(2H)-one skeleton has relied heavily on transition metal-catalyzed C-O or C-C bond coupling reactions. These conventional methodologies often necessitate the use of expensive and sometimes toxic precious metal catalysts, which significantly inflate the raw material costs for large-scale production. Furthermore, these reactions typically require stringent operational conditions, such as anhydrous and oxygen-free environments, which demand specialized equipment and increase energy consumption. The purification process is also notoriously difficult, as removing trace metal residues to meet pharmaceutical purity standards often requires additional processing steps like chelation or extensive chromatography. These factors collectively create bottlenecks in commercial scale-up of complex pharmaceutical intermediates, limiting the ability of manufacturers to respond quickly to market demand while maintaining cost competitiveness.
The Novel Approach
In stark contrast, the novel approach detailed in CN103087033A utilizes a simple base-catalyzed cyclization strategy that operates efficiently at room temperature. This method eliminates the need for expensive metal catalysts and special reagents, relying instead on readily available inorganic bases such as potassium carbonate or cesium carbonate. The reaction conditions are exceptionally mild, requiring no strict anhydrous or oxygen-free protocols, which simplifies the operational workflow and reduces safety risks associated with hazardous reagents. As illustrated in the general reaction scheme below, the process involves the direct coupling of ethyl 4-chloroacetoacetate with 1,2-allenone compounds, leading to the formation of the target oxepin structure with high atom economy.
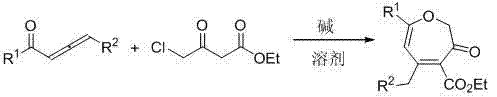
This streamlined process not only accelerates the reaction timeline but also simplifies the downstream purification, as the absence of metal catalysts means fewer impurities to remove. For a reliable pharmaceutical intermediates supplier, adopting this technology translates to a more agile and cost-effective production capability.
Mechanistic Insights into Base-Catalyzed Cyclization
The core of this synthetic breakthrough lies in the efficient nucleophilic substitution and subsequent cyclization mechanism facilitated by the base. The reaction initiates with the deprotonation of the active methylene group in ethyl 4-chloroacetoacetate by the base, generating a nucleophilic enolate species. This enolate then attacks the electrophilic center of the 1,2-allenone compound, triggering a cascade of bond formations that construct the seven-membered oxepin ring. The mild basic conditions ensure that the reaction proceeds selectively without promoting side reactions that could lead to complex impurity profiles. This mechanistic simplicity is crucial for R&D teams focused on impurity control, as it minimizes the formation of by-products that are difficult to separate. The robustness of this mechanism across various substrates demonstrates its versatility for synthesizing diverse derivatives.
Furthermore, the tolerance of this method to various functional groups on the aryl ring of the allenone substrate is a significant advantage for medicinal chemistry applications. For instance, substrates containing electron-withdrawing or electron-donating groups, such as halogens or alkoxy groups, participate effectively in the reaction. The specific example shown below highlights the synthesis of a bromo-substituted derivative, achieving an impressive yield of 80% under standard conditions. This high efficiency underscores the method's potential for producing high-purity oxepin-3(2H)-one derivatives with diverse substitution patterns, catering to the specific needs of drug discovery programs.
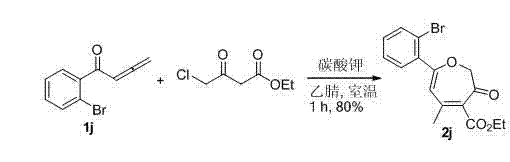
The ability to maintain high yields even with sterically hindered or electronically diverse substrates ensures that the process is scalable and reliable. For supply chain heads, this means reducing lead time for high-purity fine chemicals, as the process is less prone to failure or variability compared to sensitive metal-catalyzed alternatives.
How to Synthesize Poly-substituted Oxepin-3(2H)-one Efficiently
Implementing this synthesis route in a production environment requires adherence to specific operational parameters to maximize yield and purity. The process begins with the dissolution of the starting materials in a suitable organic solvent, followed by the controlled addition of the base catalyst. The reaction is monitored to ensure completion within the typical one-hour timeframe, after which standard workup procedures are employed. The detailed standardized synthesis steps see the guide below.
- Dissolve ethyl 4-chloroacetoacetate and 1,2-allenone compounds in an organic solvent such as acetonitrile or acetone.
- Add a base catalyst like potassium carbonate or cesium carbonate to the reaction mixture at room temperature.
- Stir the reaction for approximately 1 hour, then quench and purify the product using standard extraction and column chromatography techniques.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology addresses several critical pain points in the chemical supply chain, particularly regarding cost and reliability. The elimination of precious metal catalysts directly impacts the bill of materials, leading to substantial cost savings in raw material procurement. Additionally, the mild reaction conditions reduce energy consumption and equipment wear, further enhancing the economic viability of the process. For procurement managers, this translates to a more predictable cost structure and reduced exposure to the volatility of precious metal markets. The simplicity of the operation also means that production can be scaled up with lower capital expenditure on specialized reactor systems.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete removal of expensive transition metal catalysts. Traditional methods often rely on palladium or rhodium complexes, which are not only costly to purchase but also require expensive ligands and rigorous recovery processes to meet regulatory standards. By switching to inexpensive inorganic bases like potassium carbonate, the direct material cost is drastically simplified. Furthermore, the simplified purification process reduces the consumption of solvents and chromatography media, which are significant operational expenses in fine chemical manufacturing. This qualitative shift in the cost structure allows for more competitive pricing without compromising on the quality of the final pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability of specialized reagents and catalysts. This novel method utilizes starting materials that are commodity chemicals, readily available from multiple global suppliers. Ethyl 4-chloroacetoacetate and various allenone precursors are produced in large volumes for other industrial applications, ensuring a stable and resilient supply base. This reduces the risk of production delays caused by single-source dependencies or geopolitical disruptions affecting rare metal supplies. For supply chain heads, this means a more robust procurement strategy that can withstand market fluctuations and ensure consistent delivery schedules to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces challenges related to heat management and waste disposal. The room temperature operation of this synthesis significantly reduces the energy load required for heating or cooling, making it easier to scale from kilogram to ton quantities without complex engineering controls. Moreover, the absence of heavy metals simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations. The reduced generation of hazardous waste lowers the environmental compliance burden and associated costs. This makes the process not only economically attractive but also sustainable, appealing to partners who prioritize green chemistry principles in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. They are derived from the specific advantages and operational details outlined in the patent data, providing clarity for stakeholders evaluating this route for potential adoption. Understanding these details is essential for making informed decisions about integrating this method into existing production workflows.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN103087033A utilizes inexpensive inorganic bases such as potassium carbonate, eliminating the need for costly transition metal catalysts and simplifying the purification process.
Q: What are the typical reaction conditions for this oxepin synthesis?
A: The reaction proceeds under mild conditions, specifically at room temperature, and typically completes within 1 hour, making it highly energy-efficient and suitable for large-scale production.
Q: Is the starting material readily available for commercial manufacturing?
A: Yes, the starting materials, including ethyl 4-chloroacetoacetate and various 1,2-allenone compounds, are simple to prepare and commercially accessible, ensuring a stable supply chain for continuous production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxepin-3(2H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this metal-free synthesis are fully realized at an industrial level. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of oxepin-3(2H)-one derivatives meets the highest industry standards. Our infrastructure is designed to handle complex synthetic routes with precision, providing our partners with a dependable source of high-quality intermediates.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate the potential of this technology for your projects with confidence. Together, we can drive efficiency and innovation in the production of valuable pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
