Advanced Imidazoline Salt Catalysis for High-Purity Alpha-Hydroxyketone Manufacturing
Advanced Imidazoline Salt Catalysis for High-Purity Alpha-Hydroxyketone Manufacturing
The landscape of fine chemical synthesis is constantly evolving, driven by the need for safer, more efficient, and scalable processes. A pivotal advancement in this domain is detailed in patent CN103347846A, which discloses a revolutionary method for producing alpha-hydroxyketone compounds. These versatile intermediates are critical building blocks in the pharmaceutical and agrochemical industries, serving as precursors for a wide array of bioactive molecules. The core innovation lies in the utilization of specific imidazoline salts acting as organocatalysts, which facilitate the coupling of aldehyde compounds under remarkably mild conditions. This technological breakthrough addresses long-standing challenges associated with traditional benzoin condensation and related coupling reactions, offering a pathway to higher purity and improved process economics. For R&D directors and procurement specialists alike, understanding the nuances of this catalytic system is essential for optimizing supply chains and reducing the total cost of ownership for complex chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-hydroxyketones via aldehyde coupling has relied on catalysts that present significant operational and safety hurdles. Prior art, such as the methods described in WO2008/19927, often necessitated the in-situ generation of catalysts using hazardous reagents like bis(trimethylsilyl)amide potassium salts. This approach not only introduces severe safety risks due to the moisture sensitivity and reactivity of silyl reagents but also complicates the purification process, leading to potential metal contamination and lower overall yields. Furthermore, the catalysts derived from 1,3-bis(2,6-diisopropylphenyl)imidazoline chlorides often failed to deliver satisfactory yields, limiting their industrial viability. The reliance on such cumbersome preparatory steps creates bottlenecks in manufacturing, increases waste generation, and elevates the barrier to entry for reliable pharmaceutical intermediate supplier networks seeking consistent quality.
The Novel Approach
In stark contrast, the methodology outlined in the present invention utilizes pre-formed imidazoline salts represented by Formula (1), which are stable, easy to handle, and highly effective. These catalysts feature aryl groups with electron-withdrawing substituents at the nitrogen positions, a structural modification that dramatically enhances their catalytic activity and stability. By eliminating the need for dangerous silyl reagents and complex in-situ activation, the new process streamlines the workflow, allowing for direct addition of the catalyst to the reaction mixture. This shift represents a paradigm change in cost reduction in fine chemical manufacturing, as it reduces raw material costs, minimizes safety infrastructure requirements, and simplifies the downstream processing needed to isolate the final product. The ability to operate under mild conditions further underscores the robustness of this novel approach.
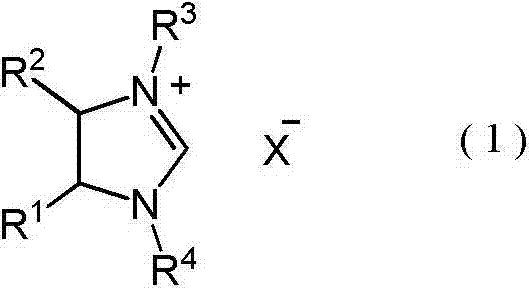
Mechanistic Insights into Imidazoline Salt-Catalyzed Coupling
The efficacy of this process is rooted in the unique electronic properties of the imidazoline salt catalyst. The presence of electron-withdrawing groups, such as halogens (chlorine, bromine) or nitro groups, on the N-aryl rings (R3 and R4 in Formula 1) modulates the electrophilicity of the carbene intermediate generated in situ. This electronic tuning facilitates the nucleophilic attack on the aldehyde carbonyl carbon, initiating the coupling sequence with greater precision than traditional thiazolium or triazolium salts. The reaction proceeds through a well-defined catalytic cycle where the base, typically an organic amine like DBU, deprotonates the imidazoline salt to generate the active carbene species. This species then reacts with the aldehyde substrate to form a Breslow intermediate, which subsequently attacks a second aldehyde molecule to forge the new carbon-carbon bond. The structural integrity of the catalyst ensures that side reactions are minimized, leading to a cleaner impurity profile.
Impurity control is a critical concern for any reliable agrochemical intermediate supplier or pharma partner. In this system, the specific substitution pattern on the catalyst prevents the formation of common by-products associated with uncontrolled aldol condensations or polymerizations. The patent data indicates that the reaction can be conducted in the presence of carbon dioxide, which appears to play a stabilizing role, possibly by interacting with the basic components or the intermediate enaminol species. This subtle interaction helps maintain the selectivity of the cross-coupling reaction, ensuring that the desired alpha-hydroxyketone is formed preferentially over homocoupling products when different aldehydes are used. Such mechanistic control is vital for achieving the high-purity specifications required for regulatory compliance in drug substance manufacturing.
How to Synthesize Alpha-Hydroxyketones Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize the benefits of the novel catalyst. The process generally involves charging the aldehyde substrates, the specific imidazoline salt catalyst, and a solvent into a reactor, followed by the addition of a base. The reaction is typically conducted at moderate temperatures, ranging from 40°C to 60°C, which balances reaction rate with energy efficiency. Detailed standard operating procedures regarding stoichiometry, mixing rates, and work-up protocols are essential for reproducibility. The following guide outlines the generalized steps derived from the patent examples to assist technical teams in evaluating this technology for their specific applications.
- Prepare the reaction vessel by charging aldehyde compounds, the specific imidazoline salt catalyst (Formula 1), and a compatible organic solvent.
- Introduce an organic base such as DBU to the mixture and maintain stirring under a controlled carbon dioxide atmosphere to stabilize the catalytic cycle.
- Heat the reaction mixture to temperatures between 40°C and 60°C, monitor conversion via GC/HPLC, and isolate the product through distillation or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this new catalytic technology offers tangible strategic benefits beyond mere chemical curiosity. The elimination of hazardous silyl reagents and the use of stable, shelf-stable catalyst salts directly translate to reduced logistics costs and simplified storage requirements. This stability ensures a more reliable supply of the catalyst itself, mitigating the risk of production stoppages due to reagent degradation. Furthermore, the high yields reported in the patent examples, such as the 88% yield achieved in Example 23 for 4-(methylthio)-2-oxo-1-butanol, suggest a substantial increase in atom economy. Higher yields mean less raw material waste and lower disposal costs, contributing to a greener and more cost-effective manufacturing footprint. These factors combined create a compelling business case for adopting this method in large-scale production environments.
- Cost Reduction in Manufacturing: The removal of expensive and hazardous activation reagents like bis(trimethylsilyl)amide potassium salts significantly lowers the bill of materials. Additionally, the simplified work-up procedure, which avoids complex metal scavenging steps often required with transition metal catalysts, reduces downstream processing time and consumable costs. The ability to achieve high conversion rates with low catalyst loading further drives down the unit cost of the final alpha-hydroxyketone product, enhancing overall margin potential for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The catalysts described are robust organic salts that do not require special inert atmosphere storage to the same extent as sensitive organometallics. This durability simplifies the supply chain, allowing for broader sourcing options and reduced lead times for high-purity intermediates. The use of readily available aldehyde starting materials, including formaldehyde polymers and simple aliphatic aldehydes, ensures that the upstream supply base remains stable and resilient against market fluctuations, securing continuous production capability.
- Scalability and Environmental Compliance: The reaction conditions are mild and operate at near-atmospheric pressure, making the process inherently safer and easier to scale from kilogram to multi-ton quantities. The avoidance of heavy metals aligns with stringent environmental regulations and corporate sustainability goals, reducing the burden of wastewater treatment and hazardous waste disposal. This environmental compatibility facilitates smoother regulatory approvals and supports the long-term viability of the manufacturing site as a sustainable partner in the global chemical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this imidazoline salt-catalyzed process. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this technology into existing production lines or new product development pipelines.
Q: What are the safety advantages of this new imidazoline salt catalyst compared to prior art?
A: Unlike previous methods requiring hazardous silyl reagents and potassium salts for catalyst generation, this process utilizes pre-formed, stable imidazoline salts, significantly reducing operational risks and simplifying handling protocols.
Q: Can this catalytic system handle cross-coupling reactions between different aldehydes?
A: Yes, the patent data demonstrates successful cross-coupling reactions between aliphatic aldehydes and formaldehyde or aromatic aldehydes, yielding specific alpha-hydroxyketone structures with high selectivity.
Q: How does the carbon dioxide atmosphere impact the reaction efficiency?
A: The presence of carbon dioxide during the stirring step has been shown to enhance the selectivity and yield of the alpha-hydroxyketone product, likely by stabilizing intermediate species within the catalytic cycle.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxyketone Supplier
The technical potential of the imidazoline salt-catalyzed synthesis of alpha-hydroxyketones is immense, offering a clear path to superior product quality and process efficiency. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch meets the exacting standards required by the global pharmaceutical industry. We understand the critical nature of these intermediates in the synthesis of life-saving medicines and are dedicated to providing a supply chain that is both robust and responsive to your evolving needs.
We invite you to explore how this advanced catalytic technology can optimize your manufacturing operations. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us collaborate to drive innovation and efficiency in your supply chain, ensuring you have a competitive edge in the marketplace through superior chemical solutions.
