Advanced Cobalt-Catalyzed Synthesis of Isoindolinone Derivatives for Commercial Pharmaceutical Applications
Advanced Cobalt-Catalyzed Synthesis of Isoindolinone Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously seeks efficient, cost-effective, and environmentally benign methodologies for constructing complex heterocyclic scaffolds that serve as core structures in bioactive molecules. Patent CN110343087B introduces a groundbreaking approach to synthesizing isoindolinone derivatives through cobalt-catalyzed C-H bond activation and metal carbene migration insertion. This technology represents a significant leap forward in organic synthesis, addressing critical pain points related to catalyst cost, toxicity, and operational complexity that have historically plagued the production of nitrogen-containing heterocycles. By leveraging earth-abundant cobalt instead of precious metals, this method not only aligns with green chemistry principles but also offers a robust pathway for generating high-value pharmaceutical intermediates with exceptional atom economy and functional group compatibility.
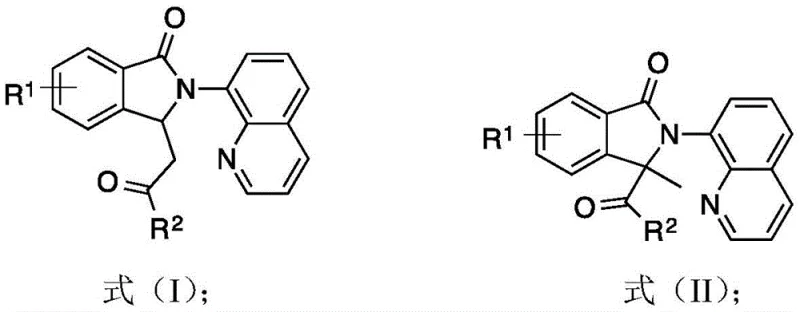
Isoindolinone motifs are pervasive in natural products and drug candidates, necessitating reliable synthetic routes that can withstand the rigors of process chemistry. The invention disclosed in this patent provides two distinct classes of isoindolinone derivatives, designated as Formula (I) and Formula (II), accessible through tunable reaction conditions. This dual-pathway capability allows chemists to selectively access different structural analogs from similar starting materials, thereby accelerating structure-activity relationship (SAR) studies during drug discovery. For procurement and supply chain leaders, the implications are profound: a shift towards base-metal catalysis reduces dependency on volatile precious metal markets, ensuring greater supply chain stability and predictable costing for long-term manufacturing contracts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic strategies for constructing the isoindolinone core often rely on classical cyclization reactions such as Aza-Nazarov cyclizations or Brønsted acid-catalyzed Friedel–Crafts reactions. These legacy methods frequently suffer from significant drawbacks, including the requirement for harsh reaction conditions, stoichiometric amounts of activating agents, and limited substrate scope due to poor functional group tolerance. Furthermore, many conventional routes involve multi-step sequences with low overall yields, generating substantial chemical waste and increasing the environmental footprint of the manufacturing process. The reliance on pre-functionalized starting materials also adds to the raw material costs and extends the lead time required to produce the final active pharmaceutical ingredient (API) intermediates, creating bottlenecks in the development timeline.
The Novel Approach
In stark contrast, the novel cobalt-catalyzed C-H activation methodology described in the patent offers a direct, atom-economical route to isoindolinone derivatives. By utilizing aromatic amides and alpha-carbonyl diazo compounds as readily available starting materials, this approach bypasses the need for pre-halogenated substrates, significantly streamlining the synthetic sequence. The ability to toggle between two distinct product classes (Formula I and Formula II) simply by modulating the oxidant and temperature provides unparalleled flexibility in molecular design. This transition metal-catalyzed strategy not only simplifies the operational workflow but also enhances the sustainability profile of the synthesis, making it an ideal candidate for modern, green manufacturing processes that prioritize efficiency and reduced waste generation.
Mechanistic Insights into Cobalt-Catalyzed C-H Activation and Carbene Insertion
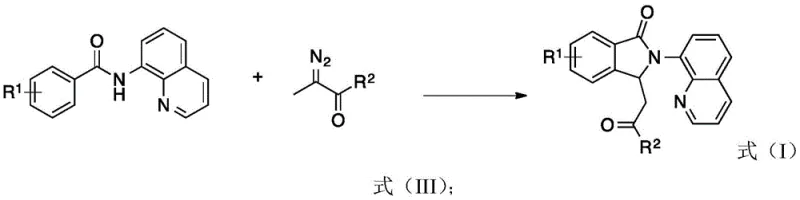
The mechanistic elegance of this transformation lies in the cooperative interplay between the cobalt catalyst and the directing group installed on the aromatic amide substrate. Initially, the cobalt species coordinates with the nitrogen atom of the 8-aminoquinoline moiety, facilitating the activation of the proximal ortho-C(sp2)–H bond through a concerted metalation-deprotonation (CMD) pathway or similar mechanism. This step generates a stable cyclometalated intermediate, which is pivotal for the subsequent regioselective functionalization. In the pathway leading to Formula (I) derivatives, the presence of oxidants like AgOAc and TEMPO promotes the conversion of the alpha-carbonyl diazo compound into a reactive enone intermediate. This electrophilic species then undergoes selective insertion into the carbon-metal bond, followed by reductive elimination to forge the new C-C bond and close the isoindolinone ring system with high fidelity.
Conversely, the formation of Formula (II) derivatives proceeds through a distinct mechanistic manifold driven by the use of tert-butyl hydroperoxide (TBHP) as the oxidant at lower temperatures. Under these conditions, the alpha-carbonyl diazo compound generates a metal-carbene intermediate upon interaction with the cobalt center. This highly reactive carbene species undergoes migratory insertion into the cobalt-carbon bond of the cyclometalated intermediate. The subsequent reductive elimination step releases the final product, featuring a quaternary carbon center at the 3-position of the isoindolinone ring. This divergence in mechanism highlights the sophistication of the catalytic system, allowing for precise control over the molecular architecture by merely adjusting the oxidative environment, a feature that is highly valued in the synthesis of complex pharmaceutical intermediates.
How to Synthesize Isoindolinone Derivatives Efficiently
The practical implementation of this synthesis involves dissolving the aromatic amide and alpha-carbonyl diazo compound in a solvent such as 1,2-dichloroethane, followed by the addition of the cobalt catalyst and the specific oxidant system tailored to the desired product type. The reaction mixture is then heated to the optimal temperature, ranging from 30°C for Type II derivatives to 130°C for Type I derivatives, and stirred for a period of 6 to 24 hours. Upon completion, the crude reaction mixture is cooled, filtered to remove inorganic salts, and the target product is isolated via standard column chromatography techniques. This straightforward workup procedure minimizes downstream processing costs and facilitates rapid purification, which is essential for maintaining high throughput in a commercial setting. The detailed standardized synthesis steps are outlined in the guide below.
- Prepare the reaction mixture by combining aromatic amide compounds and alpha-carbonyl diazo compounds in a suitable solvent such as 1,2-dichloroethane.
- Add the cobalt catalyst (Co(OAc)2 or Co(acac)2) and the appropriate oxidant system (AgOAc/TEMPO or TBHP) depending on the desired regioselectivity.
- Heat the reaction mixture to the specified temperature (30°C to 130°C) and maintain for 6 to 24 hours, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this cobalt-catalyzed technology translates into tangible strategic advantages regarding cost stability and operational resilience. The substitution of expensive noble metal catalysts with inexpensive cobalt salts drastically reduces the raw material cost basis, insulating the manufacturing process from the price volatility associated with rhodium, palladium, or iridium. Moreover, the ligand-free nature of the catalytic system eliminates the need for specialized, high-cost phosphine or N-heterocyclic carbene ligands, further simplifying the supply chain and reducing the inventory burden. The robustness of the reaction conditions ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed runs or difficult purifications.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of earth-abundant cobalt catalysts which are significantly cheaper than traditional precious metal alternatives. By eliminating the requirement for expensive ligands and reducing the number of synthetic steps through direct C-H functionalization, the overall cost of goods sold (COGS) is substantially lowered. Additionally, the high yields reported (ranging from 73% to 95%) maximize the utilization of starting materials, reducing waste disposal costs and improving the overall material efficiency of the production line.
- Enhanced Supply Chain Reliability: The starting materials, including aromatic amides and alpha-carbonyl diazo compounds, are commercially available and easy to synthesize, ensuring a stable and continuous supply of feedstocks. The simplicity of the reaction setup, which does not require inert atmosphere techniques or specialized high-pressure equipment, allows for execution in standard multipurpose reactors found in most chemical manufacturing facilities. This accessibility reduces the barrier to entry for contract manufacturing organizations (CMOs) and ensures that production capacity can be easily scaled up to meet market demand without significant capital expenditure.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the use of low-toxicity cobalt catalysts and the generation of minimal heavy metal waste align well with increasingly stringent global regulatory standards. The atom-economic nature of the C-H activation process means fewer byproducts are formed, simplifying effluent treatment and reducing the environmental footprint of the facility. The ability to run reactions at moderate temperatures (as low as 30°C for certain derivatives) also lowers energy consumption, contributing to a more sustainable manufacturing profile that appeals to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cobalt-catalyzed synthesis platform. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations for potential partners and licensees.
Q: What are the primary advantages of using cobalt catalysts over noble metals for isoindolinone synthesis?
A: Cobalt catalysts offer significant cost advantages due to their abundance and low toxicity compared to precious metals like palladium or rhodium. Furthermore, the cobalt-catalyzed process described in patent CN110343087B operates under ligand-free conditions, simplifying the reaction setup and reducing raw material costs while maintaining high yields and excellent functional group tolerance.
Q: How does the reaction condition affect the regioselectivity of the isoindolinone derivatives?
A: The patent demonstrates that regioselectivity can be precisely controlled by adjusting the oxidant and reaction temperature. Using AgOAc and TEMPO at 130°C favors the formation of Type I derivatives via enone intermediate insertion, whereas utilizing TBHP at a milder 30°C promotes metal carbene migration insertion to yield Type II derivatives, allowing for versatile synthetic planning.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly suitable for scale-up due to its use of inexpensive catalysts, simple post-treatment procedures involving filtration and chromatography, and robust yields ranging from 73% to 95%. The avoidance of sensitive ligands and the use of common solvents like 1,2-dichloroethane further enhance its feasibility for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoindolinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating drug discovery and development pipelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the cobalt-catalyzed synthesis of isoindolinones can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest quality standards required by global regulatory agencies.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective and versatile synthetic methodology for their next-generation therapeutic programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce time-to-market by integrating this cutting-edge cobalt-catalyzed technology into your manufacturing strategy today.
