Advanced Synthesis of Isoindolinone-Substituted Dipeptides via MOF-Cu Catalysis for Commercial Scale-Up
Introduction to Next-Generation Dipeptide Synthesis
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more sustainable and efficient synthetic routes for complex bioactive scaffolds. A pivotal advancement in this domain is documented in patent CN110372774B, which discloses a novel methodology for the preparation of isoindolinone-substituted α-acyloxyamide dipeptide derivatives. These compounds represent a critical class of structural motifs found in numerous natural alkaloids and pharmaceutical active molecules, serving as essential building blocks for drug discovery and agrochemical formulation. The patent introduces a groundbreaking approach utilizing a Metal-Organic Framework copper catalyst (MOF-Cu) to facilitate a three-component Ugi reaction. This technology addresses long-standing challenges in peptide synthesis by enabling a one-pot transformation under exceptionally mild conditions. By leveraging this innovative catalytic system, manufacturers can achieve high selectivity and yield while significantly simplifying the downstream processing workflow. The strategic implementation of this chemistry positions supply chains to deliver high-purity pharmaceutical intermediates with greater reliability and reduced environmental impact.
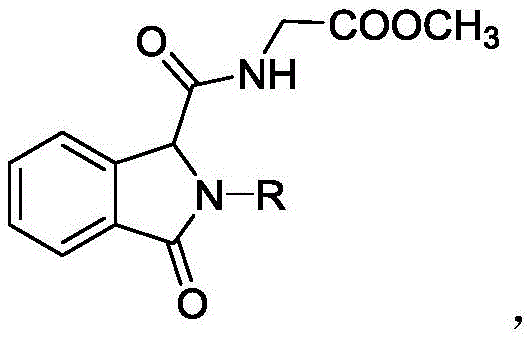
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted chain dipeptide derivatives has been fraught with significant operational hurdles that impede efficient commercial production. Traditional methodologies often rely on multi-step synthetic sequences that require harsh reaction conditions, such as elevated temperatures, strong acidic or basic environments, and the use of toxic organic solvents. These rigorous conditions not only increase energy consumption but also pose safety risks and generate substantial hazardous waste, complicating regulatory compliance and waste management protocols. Furthermore, conventional routes frequently suffer from poor atom economy and low overall yields due to the necessity of isolating unstable intermediates and performing extensive purification between steps. The reliance on homogeneous catalysts in many legacy processes introduces another layer of complexity, as removing trace metal residues from the final product to meet stringent pharmaceutical purity specifications often requires costly and time-consuming chelation or chromatography steps. These cumulative inefficiencies result in prolonged lead times and inflated production costs, making it difficult for procurement teams to secure cost reduction in pharmaceutical intermediates manufacturing without compromising on quality standards.
The Novel Approach
In stark contrast to these legacy limitations, the technology described in CN110372774B presents a paradigm shift through the application of a heterogeneous MOF-Cu catalyst in a three-component Ugi reaction. This novel approach consolidates what was previously a multi-step sequence into a single, streamlined pot operation conducted at room temperature. The use of a robust metal-organic framework support ensures that the catalytic active sites remain accessible while preventing metal leaching, thereby maintaining the structural integrity of the catalyst throughout the reaction cycle. This heterogeneity allows for the catalyst to be simply filtered off upon reaction completion, eliminating the need for complex metal scavenging procedures and drastically reducing the burden on downstream purification. The reaction proceeds efficiently in common green solvents such as methanol, ethanol, or even water, further enhancing the environmental profile of the process. By integrating these features, the new method offers a direct pathway to commercial scale-up of complex polymer additives and bioactive intermediates, providing a competitive edge through operational simplicity and superior resource utilization.
Mechanistic Insights into MOF-Cu Catalyzed Ugi-3CR
The core of this technological breakthrough lies in the unique properties of the MOF-Cu catalyst, which is constructed from divalent copper ions coordinated with terphenyltetracarboxylic acid ligands. This specific architecture creates a highly porous and stable framework that acts as a solid support for the catalytic centers involved in the Ugi three-component reaction (Ugi-3CR). Mechanistically, the reaction initiates with the condensation of an aldehyde component, specifically methyl 2-formylbenzoate, with an amine substrate to form an imine intermediate in situ. The MOF-Cu catalyst facilitates this condensation and subsequently activates the isonitrile component, promoting its nucleophilic attack on the iminium ion. The presence of the carboxylic acid moiety within the aldehyde substrate then triggers an acyl transfer, leading to the formation of the characteristic α-acyloxyamide backbone and the subsequent cyclization to form the isoindolinone ring system. The copper nodes within the MOF structure likely act as Lewis acid sites, coordinating with the carbonyl oxygen atoms to lower the activation energy of the rate-determining steps, thereby accelerating the reaction kinetics without the need for external heating.
From an impurity control perspective, the heterogeneous nature of the MOF-Cu catalyst provides a distinct advantage in managing the杂质 profile of the final product. Unlike homogeneous catalysts that dissolve completely and can become entrapped within the product matrix, the solid MOF particles remain discrete and can be physically separated via simple filtration. This physical separation mechanism ensures that the final crude product is virtually free of heavy metal contaminants, a critical requirement for reliable agrochemical intermediate supplier certifications and pharmaceutical grade materials. Furthermore, the patent data indicates that the catalyst retains its structural fidelity and catalytic efficiency over multiple recycling runs, suggesting that the active sites are not poisoned by reaction byproducts. This stability minimizes the generation of metal-containing waste streams and ensures consistent batch-to-batch reproducibility, which is paramount for maintaining strict quality control standards in large-scale manufacturing environments.
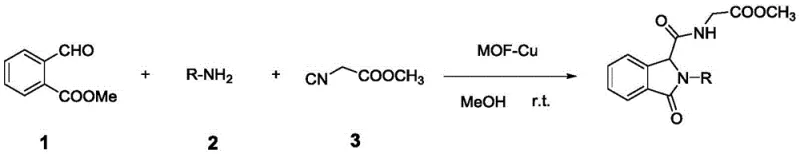
How to Synthesize Isoindolinone Derivatives Efficiently
The practical execution of this synthesis route is designed for maximum operational ease, making it highly attractive for process chemists looking to implement robust manufacturing protocols. The procedure begins with the preparation of the key aldehyde precursor, methyl 2-formylbenzoate, which is synthesized via a straightforward esterification of 2-formylbenzoic acid. Once this precursor is secured, the core transformation involves dissolving the aldehyde and the chosen amine in a protic solvent such as methanol or ethanol at ambient temperature. Following complete dissolution, the solid MOF-Cu catalyst is introduced to the mixture, followed by the addition of the isonitrile component, which serves as the final coupling partner. The reaction mixture is then stirred for a period ranging from 12 to 24 hours, during which time the multicomponent assembly occurs spontaneously without the need for external energy input. Upon confirmation of reaction completion via thin-layer chromatography, the solid catalyst is removed by filtration, the solvent is evaporated under reduced pressure, and the crude residue is purified by column chromatography to yield the target dipeptide derivative in high purity. For detailed standard operating procedures and specific parameter optimization, please refer to the guide below.
- Preparation of Methyl 2-formylbenzoate by reacting 2-formylbenzoic acid with ethanol and concentrated sulfuric acid at 10-90°C.
- Dissolving methyl 2-formylbenzoate and an amine compound in methanol, ethanol, or water at room temperature.
- Adding the MOF-Cu catalyst and an isonitrile compound, stirring for 12-24 hours, filtering the catalyst, and purifying the residue via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this MOF-Cu catalyzed synthesis route translates into tangible strategic benefits that directly impact the bottom line and operational resilience. The primary advantage lies in the drastic simplification of the manufacturing process, which eliminates the need for expensive heating infrastructure and complex multi-stage purification trains. By operating at room temperature, the process significantly lowers energy utility costs and reduces the thermal load on production facilities, contributing to a leaner and more cost-effective operational model. Additionally, the ability to recover and reuse the heterogeneous catalyst multiple times without significant loss of activity means that the effective cost of the catalytic system is amortized over many batches, leading to substantial savings in raw material expenditures. This efficiency is compounded by the use of commodity-grade solvents and readily available starting materials, which mitigates supply risk and ensures a stable flow of inputs for continuous production schedules.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the removal of expensive homogeneous metal catalysts from the process flow result in a significantly streamlined cost structure. The simplified workup procedure, which relies on filtration rather than complex extraction or chelation, reduces labor hours and solvent consumption, driving down the overall cost of goods sold. Furthermore, the high yields reported in the patent examples indicate excellent material throughput, minimizing waste disposal costs and maximizing the value derived from each kilogram of raw material input.
- Enhanced Supply Chain Reliability: The reliance on simple, commercially available starting materials such as methyl 2-formylbenzoate, various amines, and isonitriles ensures that the supply chain is not vulnerable to bottlenecks associated with exotic or proprietary reagents. The robustness of the reaction conditions, which tolerate ambient temperatures and common solvents, allows for flexible manufacturing scheduling and reduces the risk of batch failures due to equipment malfunction or thermal runaway. This reliability is crucial for maintaining consistent inventory levels and meeting the just-in-time delivery demands of downstream pharmaceutical and agrochemical clients.
- Scalability and Environmental Compliance: The one-pot nature of the Ugi reaction combined with the heterogeneous catalyst system makes this process inherently scalable from laboratory benchtop to industrial reactor volumes. The absence of toxic heavy metal residues in the final product simplifies regulatory filing and reduces the burden of environmental compliance testing. Moreover, the potential for using greener solvents like ethanol or water aligns with global sustainability initiatives, enhancing the corporate social responsibility profile of the manufacturing operation and facilitating easier permitting for facility expansions.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of this technology for their specific applications, we have compiled a set of frequently asked questions based on the detailed technical disclosures within the patent literature. These inquiries address critical aspects regarding catalyst performance, process scalability, and the biological relevance of the resulting compounds. Understanding these nuances is essential for R&D teams planning to integrate this synthetic route into their pipeline for developing new bioactive agents or optimizing existing manufacturing processes. The answers provided below are derived directly from the experimental data and embodiments described in the source documentation, ensuring accuracy and technical validity for your assessment.
Q: What are the advantages of using MOF-Cu catalyst over traditional homogeneous catalysts?
A: The MOF-Cu catalyst offers significant advantages including heterogeneity which allows for easy filtration and recovery, structural stability allowing for multiple reuse cycles without loss of catalytic activity, and operation under mild room temperature conditions which reduces energy costs.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the method is highly suitable for industrial scale-up. It utilizes a one-pot three-component reaction with readily available starting materials, operates at room temperature, and involves a simple workup procedure involving filtration and solvent removal, minimizing complex purification steps.
Q: What is the biological activity profile of the synthesized compounds?
A: The synthesized isoindolinone-substituted α-acyloxyamide dipeptide derivatives exhibit inhibitory activity against various fungal strains, including Penicillium digitatum, Penicillium italicum, and Rhizoctonia solani, indicating potential applications in agrochemical development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoindolinone Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the MOF-Cu mediated Ugi reaction in reshaping the landscape of fine chemical production. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial realities. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of isoindolinone-substituted dipeptide derivatives we produce adheres to the highest international quality standards. We are committed to leveraging our technical expertise to optimize this specific synthetic route, ensuring maximum efficiency and minimal environmental footprint for our global clientele.
We invite forward-thinking organizations to collaborate with us to unlock the full commercial potential of these valuable intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out to us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our capabilities can enhance your supply chain security and drive your product development timelines forward with confidence and precision.
