Advanced Iron-Catalyzed Synthesis of Phosphonimides for Commercial Scale-Up
Advanced Iron-Catalyzed Synthesis of Phosphonimides for Commercial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for safer, more sustainable, and cost-effective synthetic routes. A significant breakthrough in this domain is documented in Chinese Patent CN111978349B, which details a novel methodology for synthesizing phosphonimide compounds. These compounds are critical intermediates in the production of complex pharmaceutical agents and agrochemicals, serving as versatile building blocks for nitrogen-phosphorus double bond structures. The patented approach distinguishes itself by replacing hazardous azide precursors with stable N-hydrocarbyloxyamide derivatives, utilizing inexpensive iron catalysts to drive the reaction under mild conditions. This technological leap not only enhances operational safety but also streamlines the production workflow, making it an ideal candidate for reliable phosphonimide supplier networks aiming to optimize their manufacturing portfolios.
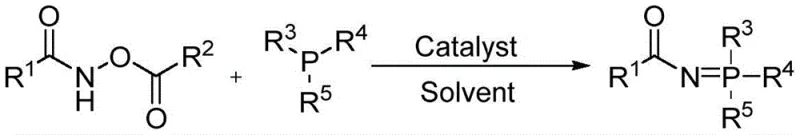
For R&D directors and process chemists, the implications of this patent are profound. The ability to conduct these transformations at room temperature in an air atmosphere removes the stringent requirement for inert gas shielding and specialized low-temperature equipment. Furthermore, the use of earth-abundant iron catalysts, such as ferrous chloride, offers a distinct advantage over precious metal systems, aligning perfectly with modern green chemistry principles. This report delves deep into the mechanistic advantages, commercial viability, and scalability of this innovative process, providing a comprehensive roadmap for integrating this technology into high-volume commercial production lines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phosphonimides has relied heavily on the classic Staudinger reaction or modifications thereof, which typically involve the reaction of azide compounds with tertiary phosphines. As illustrated in prior art such as the Katritzky method shown below, these conventional pathways necessitate the use of acyl azides as key starting materials or intermediates.
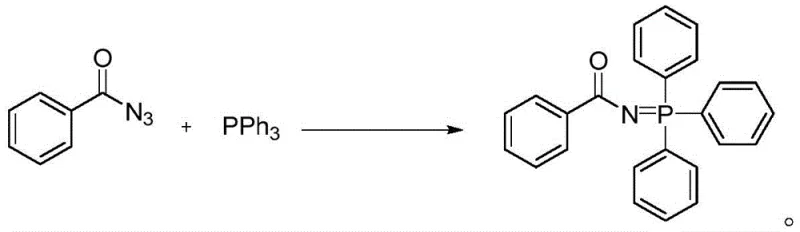
The reliance on azide chemistry presents severe limitations for industrial scale-up. Acyl azides are inherently unstable and possess high explosive potential, posing significant safety risks during storage, transportation, and handling, especially in large quantities. Additionally, the generation of azide intermediates often requires harsh reaction conditions, including the use of toxic reagents like sodium azide or thionyl chloride, which complicates waste disposal and environmental compliance. The instability of the resulting phosphonimide intermediates in some classical routes further necessitates immediate downstream processing, limiting flexibility in manufacturing scheduling and increasing the risk of batch failure due to decomposition.
The Novel Approach
In stark contrast, the method disclosed in CN111978349B introduces a paradigm shift by utilizing N-hydrocarbyloxyamide derivatives as the nitrogen source. This innovative strategy completely bypasses the formation of explosive azide species, thereby fundamentally altering the safety profile of the synthesis. The reaction proceeds smoothly between the N-hydrocarbyloxyamide derivative and a tertiary phosphine, such as triphenylphosphine, in the presence of a catalytic amount of an iron salt. This new route operates under exceptionally mild conditions, specifically at room temperature ranging from 0 to 35°C and under a simple air atmosphere, eliminating the need for energy-intensive heating or cooling systems. The simplicity of the operation, combined with the high stability of the starting materials, allows for a much more robust and forgiving process window, which is critical for maintaining consistent quality in commercial manufacturing environments.
Mechanistic Insights into Iron-Catalyzed Phosphonimide Formation
The core of this technological advancement lies in the activation of the N-O bond within the N-hydrocarbyloxyamide derivative by the iron catalyst. While the precise mechanistic cycle may involve complex coordination chemistry, the overarching principle involves the iron species facilitating the nucleophilic attack of the tertiary phosphine on the carbonyl or adjacent nitrogen center, leading to the cleavage of the weak N-O bond and the subsequent formation of the robust N=P double bond. The choice of catalyst is pivotal; the patent highlights that inexpensive iron salts like ferrous chloride (FeCl2), ferrous acetate, or ferric chloride are highly effective. This contrasts sharply with older methods that might require stoichiometric amounts of phosphine or more expensive transition metals. The catalytic nature of the iron species ensures that only minute quantities (0.01 to 0.1 molar equivalents) are needed to drive the reaction to completion, significantly reducing the metal load in the final product and simplifying purification protocols.
From an impurity control perspective, this mechanism offers superior selectivity. Traditional azide routes often suffer from side reactions involving the decomposition of the azide group, leading to difficult-to-remove nitrogenous byproducts. In the iron-catalyzed pathway, the byproduct is typically a carboxylic acid derivative resulting from the cleavage of the acyloxy group, which is generally easier to separate from the target phosphonimide via standard aqueous workup or crystallization. The broad substrate scope demonstrated in the patent, accommodating electron-withdrawing and electron-donating groups on the aromatic rings as well as various aliphatic chains, suggests that the catalytic system is highly tolerant of steric and electronic variations. This robustness ensures that the process can be applied to a wide library of analogues without requiring extensive re-optimization for each new derivative, a key factor for contract development and manufacturing organizations (CDMOs).
How to Synthesize N-Acylphosphonimides Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and solvent choices to maximize yield and purity. The general protocol involves charging a reaction vessel with the N-hydrocarbyloxyamide derivative, the tertiary phosphine, and the selected iron catalyst in an appropriate organic solvent. The patent specifies that solvents such as dichloroethane, methanol, ethanol, acetonitrile, or tetrahydrofuran are suitable, with dichloroethane often providing optimal results. The molar ratio of the amide derivative to the phosphine is typically maintained between 1:1 and 1:2, while the catalyst loading is kept low, around 0.01 to 0.1 equivalents. Detailed standardized synthesis steps see the guide below.
- Combine N-hydrocarbyloxyamide derivative, tertiary phosphine, and an iron-based catalyst in a reaction vessel with an organic solvent.
- Stir the mixture at room temperature (0-35°C) under an air atmosphere for 0.5 to 12 hours.
- Upon completion, perform standard post-treatment workup to isolate the high-purity phosphonimide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iron-catalyzed methodology represents a strategic opportunity to reduce costs and mitigate supply risks. The elimination of hazardous azide reagents removes the need for specialized storage facilities and expensive safety protocols, directly translating to lower overhead costs. Furthermore, the use of commodity chemicals like triphenylphosphine and iron salts ensures a stable and predictable supply chain, unaffected by the volatility often seen with specialty azide reagents. The ability to run reactions at room temperature also drastically reduces energy consumption compared to processes requiring reflux or cryogenic conditions, contributing to both cost reduction in pharmaceutical intermediate manufacturing and a smaller carbon footprint.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted. By replacing expensive and dangerous azide precursors with stable N-hydrocarbyloxyamides, raw material costs are stabilized. The use of cheap iron catalysts instead of precious metals like palladium or rhodium further drives down the bill of materials. Additionally, the simplified workup procedure, which avoids complex quenching of reactive azides, reduces labor time and solvent usage. The high yields reported, often exceeding 80-90% even on gram scales, minimize waste and maximize the throughput of the reactor, ensuring that every kilogram of input material generates maximum value.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount in the pharmaceutical industry. The starting materials for this reaction, including substituted benzoyl derivatives and triphenylphosphine, are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failures related to temperature control or inert gas supply. This reliability allows for more accurate lead time predictions and ensures that high-purity phosphonimides can be delivered consistently to meet tight project deadlines.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this method is designed for scalability. The exothermic nature of the reaction is manageable at room temperature, reducing the risk of thermal runaway in large reactors. From an environmental standpoint, the avoidance of azides eliminates the generation of hydrazoic acid and other toxic nitrogenous wastes, simplifying effluent treatment. The use of iron, a non-toxic metal, aligns with increasingly stringent regulatory requirements regarding heavy metal residues in active pharmaceutical ingredients (APIs), facilitating smoother regulatory filings and approvals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: Why is this iron-catalyzed method safer than traditional Staudinger reactions?
A: Traditional methods often rely on explosive acyl azides as intermediates. This patented process utilizes stable N-hydrocarbyloxyamide derivatives, eliminating the safety hazards associated with handling and storing high-energy azide compounds.
Q: What are the optimal reaction conditions for scaling this synthesis?
A: The process operates efficiently at room temperature (0-35°C) under ambient air atmosphere, removing the need for expensive inert gas protection or cryogenic cooling, which significantly simplifies large-scale manufacturing.
Q: Can this method accommodate diverse substrate structures?
A: Yes, the protocol demonstrates excellent substrate tolerance, successfully converting derivatives with various substituents including alkyl, aryl, halogen, and heterocyclic groups into the corresponding phosphonimides with high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acylphosphonimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iron-catalyzed synthesis of phosphonimides described in CN111978349B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, ensuring stringent purity specifications and rigorous QC labs verify every batch. We understand that moving from bench-scale discovery to commercial supply requires a partner who can navigate the complexities of process optimization, safety management, and regulatory compliance with equal expertise.
We invite you to collaborate with us to leverage this advanced technology for your next project. Whether you require custom synthesis of novel phosphonimide derivatives or scale-up of existing routes, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific needs. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage through superior chemical manufacturing solutions.
