Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Iodine-Promoted Cyclization for Global Pharma Supply Chains
Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Iodine-Promoted Cyclization for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust and economically viable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN114920707B, which discloses a highly efficient preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. These specific heterocyclic motifs are ubiquitous in modern medicinal chemistry, forming the core skeleton of potent bioactive inhibitors such as Factor IXa inhibitors and GlyT1 inhibitors, as well as anticonvulsant drugs. The introduction of a trifluoromethyl group is particularly strategic, as it significantly enhances the metabolic stability and lipophilicity of the parent molecule, thereby improving overall pharmacokinetics. This patented technology represents a paradigm shift by utilizing the common organic solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source, streamlining the synthesis of these high-value intermediates.
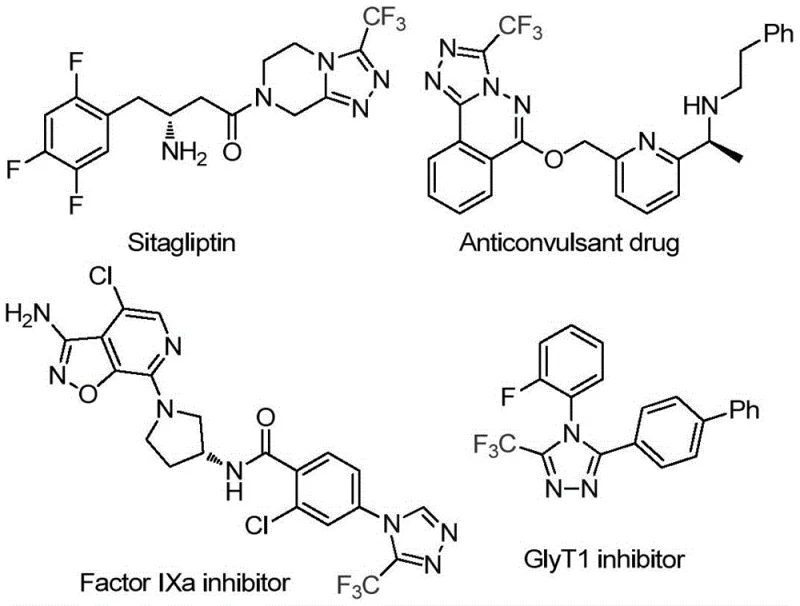
The structural versatility of these compounds is evident in their application across diverse therapeutic areas. As illustrated in the provided chemical structures, the 3-trifluoromethyl-1,2,4-triazole core is integral to the efficacy of complex drug molecules. For R&D directors focusing on pipeline optimization, having access to a reliable supply of these functionalized intermediates is crucial for accelerating lead optimization campaigns. The ability to rapidly access diverse analogues through a unified synthetic platform allows for more extensive structure-activity relationship (SAR) studies without being bottlenecked by complex or low-yielding synthetic routes. This technological advancement directly addresses the need for high-purity pharmaceutical intermediates that can be seamlessly integrated into downstream coupling reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of trifluoromethyl-substituted triazole rings has often relied on multi-step sequences involving harsh reaction conditions or expensive, specialized reagents. Conventional methodologies frequently necessitate the use of pre-functionalized hydrazines or nitriles that require stringent anhydrous and anaerobic environments to prevent side reactions or decomposition of sensitive intermediates. Furthermore, many existing protocols utilize stoichiometric amounts of costly transition metal catalysts or oxidants, which not only inflate the raw material costs but also introduce significant challenges in downstream purification due to heavy metal residue limits imposed by regulatory bodies. The logistical burden of maintaining inert atmospheres and sourcing exotic carbon donors often results in prolonged lead times and reduced overall process efficiency, making these traditional routes less attractive for large-scale commercial manufacturing where cost and throughput are paramount.
The Novel Approach
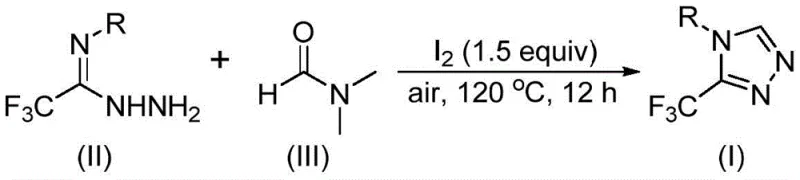
In stark contrast, the novel approach described in the patent leverages a tandem cyclization strategy promoted by molecular iodine, utilizing DMF as a dual-purpose solvent and reactant. This method elegantly bypasses the need for external carbon sources by activating the methyl or formyl groups inherent in the DMF molecule itself. The reaction proceeds smoothly under air atmosphere at moderate temperatures ranging from 110 to 130°C, eliminating the capital expenditure associated with specialized inert gas lines and glovebox operations. By employing readily available starting materials like trifluoroethyliminohydrazide and common solvents, this route drastically simplifies the operational complexity. The result is a streamlined process that offers high reaction efficiency and broad substrate tolerance, enabling the synthesis of various 4-substituted derivatives simply by modifying the initial hydrazide precursor, thus providing a versatile platform for generating chemical diversity.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation is a fascinating example of solvent participation in organic synthesis, offering deep insights for process chemists aiming to optimize reaction parameters. The reaction initiates with the activation of DMF by molecular iodine, which facilitates the cleavage of the solvent molecule to generate reactive species. Depending on whether the formyl or the N-methyl group of DMF acts as the carbon source, two distinct but convergent pathways are proposed. In the formyl pathway, a condensation occurs between the formyl group and the trifluoroethyliminohydrazide to form a hydrazone intermediate, followed by intramolecular cyclization and elimination of dimethylamine. Alternatively, the N-methyl pathway involves the formation of an amine salt, nucleophilic addition, and subsequent oxidative aromatization. Understanding these nuances allows for precise control over impurity profiles, ensuring that the final product meets the stringent purity specifications required for GMP manufacturing.
From an impurity control perspective, the simplicity of the reagent system contributes to a cleaner reaction profile. Since DMF is used in excess as the solvent, the kinetics favor the desired cyclization over potential oligomerization or decomposition pathways that might occur with limiting reagents. The use of molecular iodine as a mild promoter avoids the generation of toxic heavy metal waste streams, simplifying the environmental compliance aspect of the process. Post-reaction workup typically involves standard filtration and column chromatography, techniques that are well-established in industrial settings. This mechanistic robustness ensures that the process is not only scientifically sound but also practically viable for scaling up, providing a reliable foundation for producing high-purity OLED material precursors or pharmaceutical intermediates with consistent quality batch after batch.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis protocol requires careful attention to the stoichiometry of the iodine promoter and the thermal profile of the reaction to maximize yield. The patented procedure outlines a straightforward operational sequence where trifluoroethyliminohydrazide and molecular iodine are introduced into DMF, followed by heating under ambient air conditions. The flexibility of the reaction window, allowing for temperatures between 110°C and 130°C over a period of 10 to 15 hours, provides process engineers with the latitude to adjust parameters based on specific reactor capabilities and heat transfer characteristics. This adaptability is crucial for translating laboratory success into pilot plant operations, ensuring that the thermal energy input is sufficient to drive the cyclization to completion without degrading the sensitive trifluoromethyl moiety.
- Combine molecular iodine, trifluoroethyliminohydrazide, and organic solvent DMF in a reaction vessel under air atmosphere.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for 10 to 15 hours to ensure complete conversion.
- Upon completion, perform standard post-treatment including filtration, washing, drying, and column chromatography purification to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. The primary value proposition lies in the drastic simplification of the supply chain for raw materials. By utilizing DMF, a commodity chemical available in bulk quantities globally, the dependency on niche, high-cost specialty reagents is eliminated. This shift significantly mitigates supply risk, ensuring continuity of supply even during market fluctuations for exotic chemicals. Furthermore, the elimination of stringent anhydrous and anaerobic requirements reduces the operational overhead associated with specialized equipment maintenance and utility consumption, such as nitrogen or argon gas. These factors collectively contribute to a more resilient and cost-efficient manufacturing model that can better withstand global logistical disruptions.
- Cost Reduction in Manufacturing: The economic impact of this methodology is profound, primarily driven by the dual functionality of DMF which removes the need for purchasing separate carbon source reagents. By consolidating the solvent and reactant roles into a single, inexpensive material, the overall material cost per kilogram of product is substantially lowered. Additionally, the avoidance of expensive transition metal catalysts removes the necessity for costly metal scavenging steps and associated validation testing, further driving down the cost of goods sold (COGS). This lean approach to reagent selection allows for significant margin improvement, making the final intermediates more competitive in the global marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on universally available starting materials such as aromatic amines, trifluoroacetic acid, and hydrazine hydrate for the precursor synthesis ensures a robust upstream supply chain. These commodities are produced by multiple vendors worldwide, reducing the risk of single-source bottlenecks. The operational simplicity of running the reaction under air atmosphere means that production does not rely on complex infrastructure that could be prone to failure or require specialized technical staff. This reliability translates into shorter lead times for order fulfillment and greater flexibility in production scheduling, allowing suppliers to respond more agilely to fluctuating demand from downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat management and waste disposal, but this method is inherently designed for scalability. The use of a high-boiling solvent like DMF facilitates temperature control in large reactors, while the absence of heavy metals simplifies wastewater treatment protocols. The reaction generates minimal hazardous waste compared to traditional methods, aligning with increasingly strict environmental regulations and corporate sustainability goals. This environmental compatibility not only reduces disposal costs but also enhances the brand reputation of the manufacturer as a responsible partner, which is a critical factor for multinational corporations evaluating potential suppliers for their green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this method. Understanding these details helps stakeholders make informed decisions regarding process integration and resource allocation.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (C1 synthon), which significantly simplifies the reaction setup and reduces raw material costs compared to traditional methods requiring separate carbon donors.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, one of the key operational benefits of this patented method is that it proceeds efficiently under air atmosphere without the need for rigorous anhydrous or oxygen-free environments, greatly facilitating industrial scale-up.
Q: What types of substituents are tolerated on the aromatic ring?
A: The method demonstrates broad substrate scope, successfully accommodating various substituents including alkyl, alkoxy, alkylthio, halogens (fluorine, chlorine), and trifluoromethyl groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical instrumentation to verify identity and assay. By leveraging advanced synthetic methodologies like the iodine-promoted cyclization described herein, we can offer our partners a competitive edge through superior process economics and reliable supply continuity.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of scaling a new candidate, we are ready to provide comprehensive support. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in complex heterocyclic synthesis can accelerate your drug development timeline while optimizing your overall budget.
