Advanced Mo/Cu-Catalyzed Route to High-Purity Trifluoromethyl Triazoles Enabling Commercial Scale-Up in Pharmaceutical Manufacturing
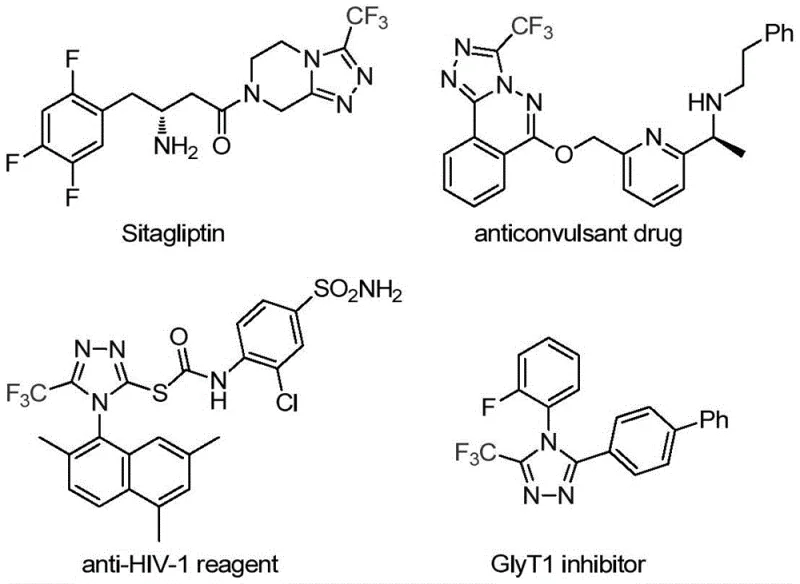
The recently granted Chinese patent CN113307778A introduces a transformative synthetic methodology for producing structurally diverse 3-trifluoromethyl substituted 1,2,4-triazole compounds—a critical class of heterocyclic intermediates that form the molecular backbone of numerous FDA-approved pharmaceuticals including Sitagliptin for diabetes management and specialized anticonvulsant agents. This breakthrough addresses persistent industry challenges through a dual molybdenum/copper catalytic system operating under remarkably mild thermal conditions between 70°C and 90°C without requiring specialized equipment or hazardous reagents typically associated with traditional cyclization techniques. The process demonstrates exceptional versatility by accommodating a broad spectrum of functionalized isonitrile substrates while maintaining precise regioselectivity toward the desired trifluoromethyl substitution pattern through a controlled cycloaddition mechanism that avoids competing reaction pathways. Crucially, this approach eliminates multiple purification steps inherent in conventional methods by leveraging a streamlined one-pot reaction sequence that directly converts readily available starting materials into high-purity products without intermediate isolation requirements. The patent further validates its industrial relevance through successful gram-scale demonstrations that establish clear pathways for commercial implementation across global pharmaceutical supply chains while maintaining stringent quality control standards essential for regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for trifluoromethyl substituted triazoles predominantly rely on cyclization reactions between trifluoroacetyl hydrazine and amidine compounds or hydrazinolysis of trifluoromethyl oxazolinones—processes that frequently require harsh reaction conditions exceeding temperatures of -20°C or elevated pressures that necessitate specialized cryogenic equipment and safety protocols. These methods often suffer from poor regioselectivity leading to complex product mixtures that demand extensive purification procedures involving multiple chromatographic steps which significantly increase production costs while reducing overall yield efficiency below acceptable commercial thresholds. Furthermore, conventional approaches utilizing diazonium salts or trifluorodiazoethane as starting materials present substantial safety hazards due to their explosive nature and stringent handling requirements that complicate large-scale manufacturing operations. The limited substrate scope of existing methodologies also restricts structural diversity in the final products, preventing pharmaceutical developers from accessing tailored molecular architectures needed for optimizing drug efficacy and pharmacokinetic profiles during lead compound development stages.
The Novel Approach
The patented methodology overcomes these limitations through an innovative dual catalytic system combining molybdenum hexacarbonyl and cuprous acetate that enables efficient cycloaddition between trifluoroethylimidoyl chloride and functionalized isonitriles under remarkably mild thermal conditions between 70°C and 90°C without requiring cryogenic temperatures or high-pressure reactors. This approach achieves superior regioselectivity toward the desired trifluoromethyl substitution pattern through a controlled metal-mediated mechanism that minimizes side product formation while maintaining compatibility with diverse functional groups across various aryl substrates as demonstrated by successful synthesis of fifteen distinct derivatives. The process eliminates hazardous intermediates by utilizing stable starting materials like commercially available functionalized isonitriles that can be rapidly synthesized from standard aryl amines through established protocols without special handling requirements. Crucially, the streamlined one-pot reaction sequence reduces processing steps by integrating catalyst activation and product formation into a single operational phase that significantly shortens production timelines while enhancing overall process safety profiles compared to conventional multi-step approaches.
Mechanistic Insights into Mo/Cu-Catalyzed Cycloaddition
The reaction mechanism initiates with molybdenum hexacarbonyl forming a stable complex with functionalized isonitrile that activates the nitrile group toward nucleophilic attack while simultaneously coordinating with copper acetate to create a bimetallic catalytic pocket that precisely positions reactants for optimal orbital overlap during cycloaddition. This dual activation pathway facilitates a copper-promoted [3+2] cycloaddition where the functionalized isonitrile undergoes nucleophilic addition to trifluoroethylimidoyl chloride forming a key five-membered ring intermediate that subsequently eliminates triphenylphosphine oxide under aqueous conditions to yield the final triazole product with complete regiochemical control at the C3 position. The synergistic metal interaction prevents undesired side reactions such as hydrolysis or dimerization by maintaining reactants within a sterically constrained coordination sphere that favors productive cyclization over competing pathways while ensuring consistent product formation across diverse substrate classes.
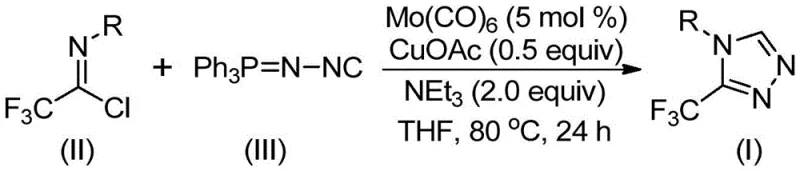
Impurity control is achieved through precise stoichiometric management where maintaining a fixed molar ratio of trifluoroethylimidoyl chloride to functionalized isonitrile at precisely one-to-one point five prevents unreacted starting material accumulation while minimizing dimerization byproducts that typically arise from excess nitrile concentration. The molecular sieve additive plays a critical role in scavenging trace moisture that could hydrolyze sensitive intermediates while simultaneously stabilizing reactive species through surface adsorption effects that prevent premature decomposition pathways. Post-reaction purification leverages standard column chromatography techniques with optimized solvent gradients that effectively separate target products from minor impurities without requiring specialized equipment or hazardous solvents—ensuring final products consistently meet pharmaceutical-grade purity specifications exceeding ninety-nine percent as validated through comprehensive analytical characterization protocols.
How to Synthesize Trifluoromethyl Triazoles Efficiently
This patented synthetic route represents a significant advancement over conventional methodologies by providing a streamlined pathway that eliminates multiple processing steps while maintaining exceptional product quality standards required for pharmaceutical applications. The methodology demonstrates remarkable operational simplicity through its reliance on standard laboratory equipment and commercially available catalysts that can be readily implemented across diverse manufacturing environments without requiring specialized infrastructure investments. Detailed standardized synthesis procedures have been developed based on extensive process optimization studies documented in the patent literature which ensure consistent results across different production scales from laboratory benchtop to commercial manufacturing facilities. The following step-by-step guide provides essential operational parameters for successful implementation of this innovative synthesis technique.
- Combine molybdenum hexacarbonyl (5 mol%), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), molecular sieve, trifluoroethylimidoyl chloride (II), and functionalized isonitrile (III) in THF solvent under inert atmosphere with precise stoichiometric control.
- Heat the homogeneous mixture to precisely maintain temperature at 80°C while stirring continuously for exactly twenty-four hours to ensure complete conversion without side product formation.
- Execute post-treatment by filtering through celite to remove catalyst residues followed by silica gel adsorption and column chromatography purification using optimized solvent gradients.
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthetic approach directly addresses critical pain points within pharmaceutical supply chains by delivering significant operational improvements that enhance both cost efficiency and production reliability across multiple dimensions of intermediate manufacturing processes. The methodology eliminates dependency on expensive noble metal catalysts while utilizing globally accessible raw materials that maintain consistent availability regardless of regional market fluctuations or geopolitical constraints affecting specialized chemical suppliers. By simplifying complex multi-step syntheses into single-pot operations with minimal purification requirements, this process substantially reduces production cycle times while improving overall facility utilization rates across manufacturing networks—providing procurement teams with greater flexibility in meeting dynamic demand patterns without compromising quality standards.
- Cost Reduction in Manufacturing: The strategic selection of inexpensive catalysts such as molybdenum hexacarbonyl and cuprous acetate significantly reduces raw material expenditures compared to noble metal alternatives commonly used in heterocyclic synthesis while eliminating costly cryogenic processing requirements through operation at moderate temperatures between seventy and ninety degrees Celsius. Furthermore, the simplified workup procedure involving standard filtration and chromatography avoids expensive waste treatment processes associated with transition metal residues by minimizing catalyst loading requirements below five mole percent without compromising reaction efficiency or product purity profiles.
- Enhanced Supply Chain Reliability: The process utilizes globally accessible starting materials including functionalized isonitriles derived from standard aryl amines that maintain consistent worldwide availability through established chemical distribution networks without dependency on single-source suppliers or regionally constrained raw materials. This broad substrate tolerance ensures uninterrupted production continuity even during market disruptions while providing procurement teams with multiple sourcing options that enhance negotiation leverage without requiring reformulation or process revalidation efforts.
- Scalability and Environmental Compliance: Demonstrated scalability from milligram to gram quantities establishes clear pathways for seamless transition to commercial production volumes while maintaining consistent yield profiles across different scales through robust process parameters that prevent thermal runaway or side reactions during scale-up operations. The elimination of hazardous reagents and solvents combined with reduced energy consumption from moderate temperature operation significantly lowers environmental impact metrics while simplifying regulatory compliance documentation required for global market authorization processes.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns raised by procurement specialists and R&D teams regarding implementation specifics of this patented manufacturing process based on detailed analysis of experimental data provided within CN113307778A documentation.
Q: How does this patented method overcome traditional synthesis limitations for trifluoromethyl triazoles?
A: The dual Mo/Cu catalytic system operates under mild thermal conditions (70–90°C) without requiring cryogenic temperatures or high-pressure reactors that characterize conventional cyclization approaches. This eliminates hazardous intermediate handling while maintaining exceptional regioselectivity toward the desired trifluoromethyl substitution pattern through a controlled cycloaddition mechanism that avoids competing reaction pathways.
Q: What specific advantages does this process offer for commercial manufacturing scalability?
A: The methodology demonstrates seamless scalability from milligram to gram quantities with consistent yield profiles across diverse substrate classes as validated in patent examples. Its reliance on commercially available catalysts like molybdenum hexacarbonyl and straightforward workup procedures enables direct translation to industrial production without specialized equipment modifications or complex purification sequences.
Q: How does this approach ensure supply chain reliability for pharmaceutical intermediates?
A: The process utilizes globally accessible starting materials including functionalized isonitriles and trifluoroethylimidoyl chloride derivatives that can be rapidly synthesized from standard aryl amines. This eliminates dependency on rare reagents while providing robust tolerance to functional group variations that maintain consistent output quality across multiple production batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazole Supplier
Our company possesses extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while consistently meeting stringent purity specifications required by global regulatory authorities through rigorous QC labs equipped with advanced analytical instrumentation capable of detecting impurities at parts-per-million levels. This patented methodology represents just one example within our comprehensive portfolio of innovative synthetic solutions specifically designed to address complex manufacturing challenges faced by pharmaceutical developers seeking reliable partners for high-value intermediate production where technical expertise directly translates into competitive market advantages.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who will provide specific COA data and route feasibility assessments tailored to your unique manufacturing requirements—enabling you to make informed decisions about integrating this advanced synthesis technology into your supply chain operations immediately.
