Advanced Lacosamide Manufacturing: A Safer, Scalable Synthetic Route for Global Supply Chains
The pharmaceutical industry is constantly seeking more efficient and safer pathways for the production of critical antiepileptic agents, and the patent CN112574058A represents a significant breakthrough in the synthesis of lacosamide. This novel methodology fundamentally restructures the traditional manufacturing process by utilizing glycine ethyl ester hydrochloride and benzophenone as primary starting materials, thereby circumventing the severe safety and environmental liabilities associated with legacy routes. By shifting away from hazardous methylating agents like dimethyl sulfate and toxic acylating reagents such as isopropyl chloroformate, this technology offers a robust framework for sustainable API production. The strategic design of this synthetic pathway not only enhances the overall atom economy but also streamlines the purification workflow, which is essential for maintaining high purity standards required by global regulatory bodies. For procurement and supply chain leaders, adopting this advanced route translates directly into reduced operational risk and a more stable supply of high-quality active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional industrial processes for lacosamide synthesis have long been plagued by the reliance on extremely hazardous reagents that pose significant challenges for both operational safety and environmental compliance. The classical route typically employs dimethyl sulfate for methylation, a substance known for its high toxicity and carcinogenic potential, which necessitates rigorous containment protocols and generates substantial amounts of acidic wastewater requiring costly neutralization. Furthermore, the use of isopropyl chloroformate for amide bond formation introduces additional risks related to thermal instability and the generation of corrosive byproducts. These legacy methods often result in lower atom economy and complex downstream processing requirements to remove trace impurities and residual toxic metals or reagents. Consequently, manufacturers face elevated production costs driven by waste treatment expenses and the need for specialized safety infrastructure, creating a bottleneck for scalable and cost-effective manufacturing in a competitive global market.
The Novel Approach
In stark contrast, the innovative route disclosed in patent CN112574058A offers a transformative solution by replacing these dangerous reagents with safer, more efficient alternatives that align with modern green chemistry principles. By initiating the synthesis with glycine ethyl ester hydrochloride and benzophenone, the process establishes a stable Schiff base intermediate that facilitates subsequent transformations without the need for toxic methylating agents. The substitution of dimethyl sulfate with X-methyl ether variants and the avoidance of isopropyl chloroformate drastically simplify the reaction profile, leading to cleaner reaction mixtures and higher crude yields. This approach not only mitigates the environmental burden associated with wastewater treatment but also enhances the overall safety profile of the manufacturing facility. For supply chain directors, this means a more resilient production capability that is less susceptible to regulatory shutdowns or safety incidents, ensuring consistent delivery of critical epilepsy medications to patients worldwide.
Mechanistic Insights into Benzophenone-Mediated Chiral Synthesis
The core of this technological advancement lies in the sophisticated manipulation of chiral intermediates through a carefully orchestrated sequence of condensation, substitution, and resolution steps. The process begins with the formation of a benzophenone imine derivative, which serves as a protective group for the amine functionality while activating the alpha-carbon for subsequent nucleophilic attack. This strategic protection-deprotection strategy allows for precise control over the stereochemistry of the molecule, which is critical for the biological activity of lacosamide. The methylation step is executed under mild conditions using sodium hydride and X-methyl ether, avoiding the harsh conditions typically required for traditional methylating agents. Following this, the aminolysis reaction with benzylamine proceeds with high selectivity, facilitated by sodium ethoxide catalysis, to construct the key carbon-nitrogen backbone of the target molecule. The final acetylation step is performed under controlled pH conditions to ensure the integrity of the chiral center is maintained throughout the synthesis.
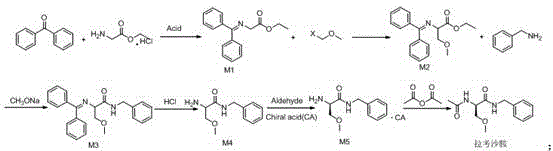
Impurity control is another critical aspect where this novel route demonstrates superior performance compared to conventional methods, particularly in the management of chiral purity and residual solvents. The use of L-tartaric acid for the resolution of intermediate M4 is a pivotal step that ensures the final product meets stringent enantiomeric excess specifications, with isomer content controlled to less than 0.01%. This high level of stereochemical control is achieved through the formation of a diastereomeric salt that can be easily purified by crystallization, effectively removing unwanted enantiomers and structural impurities. Furthermore, the avoidance of heavy metal catalysts and toxic reagents simplifies the impurity profile, reducing the burden on analytical quality control laboratories to detect and quantify trace contaminants. For R&D directors, this implies a more straightforward path to regulatory approval, as the impurity profile is cleaner and better understood, minimizing the risk of unexpected toxicological findings during clinical or commercial phases.
How to Synthesize Lacosamide Efficiently
The implementation of this synthesis route requires a precise understanding of reaction parameters and intermediate handling to maximize yield and purity at every stage of the process. The patent outlines a six-step sequence that transforms simple, commercially available starting materials into the high-value antiepileptic agent lacosamide with remarkable efficiency. Each step has been optimized to minimize side reactions and facilitate easy isolation of intermediates, often without the need for extensive chromatographic purification. The detailed standardized synthesis steps provided below offer a comprehensive guide for process chemists looking to replicate or scale this technology for commercial production. Adhering to these protocols ensures that the benefits of high atom economy and safety are fully realized in a manufacturing setting.
- React glycine ethyl ester hydrochloride with benzophenone to form intermediate M1.
- Methylate intermediate M1 using X-methyl ether to obtain intermediate M2.
- React intermediate M2 with benzylamine under sodium ethoxide catalysis to generate intermediate M3.
- Hydrolyze intermediate M3 with acid to produce intermediate M4.
- Resolve intermediate M4 using L-tartaric acid to obtain chiral intermediate M5.
- Acetylate intermediate M5 with acetic anhydride to finalize lacosamide production.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers substantial strategic advantages for procurement managers and supply chain heads looking to optimize their API sourcing strategies. The elimination of highly toxic and regulated reagents such as dimethyl sulfate significantly reduces the compliance burden and associated costs related to hazardous material handling, storage, and disposal. This simplification of the chemical inventory allows for more flexible manufacturing operations and reduces the risk of supply disruptions caused by regulatory restrictions on precursor chemicals. Additionally, the improved atom economy and higher yields inherent in this process translate directly into better raw material utilization, driving down the overall cost of goods sold without compromising on quality. For organizations focused on long-term sustainability goals, this route represents a significant step forward in reducing the environmental footprint of pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The removal of expensive and hazardous reagents like isopropyl chloroformate and dimethyl sulfate eliminates the need for specialized waste treatment protocols and costly safety infrastructure, leading to significant operational savings. By streamlining the synthesis to fewer steps with higher yields, the overall consumption of raw materials and solvents is drastically reduced, enhancing the economic viability of large-scale production. Furthermore, the simplified purification process reduces the time and resources required for downstream processing, allowing for faster batch turnover and increased manufacturing capacity. These cumulative efficiencies result in a more competitive cost structure that can be passed on to partners or reinvested into further R&D initiatives.
- Enhanced Supply Chain Reliability: The reliance on readily available and less regulated starting materials such as glycine ethyl ester hydrochloride and benzophenone ensures a more stable and resilient supply chain that is less vulnerable to geopolitical or regulatory shocks. Unlike traditional routes that depend on tightly controlled toxic precursors, this method allows for greater flexibility in sourcing and inventory management, reducing the risk of production delays. The robustness of the reaction conditions also means that the process is more forgiving of minor variations in raw material quality, further enhancing supply continuity. For supply chain heads, this translates into a more predictable delivery schedule and a reduced likelihood of stockouts for critical epilepsy medications.
- Scalability and Environmental Compliance: The inherent safety and simplicity of this synthetic route make it highly scalable from pilot plant operations to multi-ton commercial production without the need for major process re-engineering. The reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations, minimizing the risk of fines or operational shutdowns due to non-compliance. Additionally, the use of common solvents and standard reaction conditions facilitates technology transfer between manufacturing sites, enabling a distributed production model that can better serve global markets. This scalability ensures that the supply of lacosamide can be rapidly expanded to meet growing demand while maintaining the highest standards of environmental stewardship.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced lacosamide synthesis technology. These answers are derived directly from the patent data and technical specifications to provide accurate and actionable insights for decision-makers. Understanding these details is crucial for evaluating the feasibility of adopting this route for your specific manufacturing or sourcing needs. We encourage further discussion with our technical team to explore how this technology can be tailored to your specific requirements.
Q: How does this new route improve safety compared to traditional lacosamide synthesis?
A: This route eliminates the use of highly toxic dimethyl sulfate and isopropyl chloroformate, significantly reducing environmental hazards and operator risk.
Q: What is the chiral purity achievable with this resolution method?
A: The process utilizes L-tartaric acid for resolution, achieving high enantiomeric excess (ee) values, with final isomer content controlled to less than 0.01%.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the route features high atom economy and avoids complex purification steps, making it highly scalable for industrial manufacturing from 100 kgs to 100 MT.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lacosamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN112574058A to ensure the consistent supply of high-quality antiepileptic medications. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of lacosamide meets the highest international standards. We are committed to leveraging our technical expertise to optimize this novel route for maximum efficiency and cost-effectiveness, providing you with a competitive edge in the global pharmaceutical market.
We invite you to engage with our technical procurement team to discuss how we can support your specific lacosamide requirements through a Customized Cost-Saving Analysis. By partnering with us, you gain access to specific COA data and route feasibility assessments that will help you make informed decisions about your supply chain strategy. Our team is ready to collaborate with you to implement this safer, more efficient synthesis route, ensuring a stable and sustainable supply of this critical API for your patients. Contact us today to initiate a conversation about optimizing your lacosamide supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
