Advanced Ruthenium-Catalyzed Oxidation Technology for Scalable 1,2-Diketone Manufacturing
The landscape of organic synthesis for 1,2-diketones is undergoing a significant transformation driven by the urgent need for greener, more efficient manufacturing protocols. Patent CN102070383A introduces a groundbreaking methodology that leverages Ruthenium-based catalytic systems to achieve the direct oxidation of olefins into valuable 1,2-diketone scaffolds. This technology represents a pivotal shift away from hazardous stoichiometric oxidants towards a catalytic paradigm that utilizes environmentally benign oxidants such as hydrogen peroxide and tert-butyl hydroperoxide. For R&D directors and process chemists, this patent offers a robust solution that combines high selectivity with operational simplicity, addressing the critical pain points of waste generation and harsh reaction conditions often associated with traditional diketone synthesis. The ability to conduct these transformations under air atmosphere at moderate temperatures further underscores the practical utility of this invention for industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 1,2-diketones has relied heavily on methods that are increasingly untenable in modern regulatory and economic environments. Traditional approaches often involve the oxidation of alkynes, which necessitates the use of expensive and difficult-to-prepare starting materials, thereby inflating raw material costs and limiting substrate availability. Alternatively, the oxidation of alpha-hydroxy ketones is frequently employed, yet the precursor synthesis itself can be cumbersome and low-yielding. Perhaps most critically, classical oxidation protocols utilizing selenium dioxide or potassium permanganate suffer from severe drawbacks, including poor functional group tolerance and the generation of stoichiometric amounts of toxic inorganic salts. These legacy methods create substantial downstream processing burdens, requiring complex purification steps to remove heavy metal residues, which directly conflicts with the principles of green chemistry and imposes significant environmental compliance costs on manufacturers.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated Ruthenium-iodide catalytic system that fundamentally redefines the efficiency of olefin oxidation. By employing commercially available Ruthenium complexes such as dichlorobis(4-methylisopropylphenyl)ruthenium(II) or triruthenium dodecacarbonyl in conjunction with iodide co-catalysts, the reaction proceeds with exceptional selectivity. This method eliminates the need for toxic heavy metal oxidants, replacing them with cleaner alternatives like hydrogen peroxide or p-benzoquinone. The reaction conditions are remarkably mild, typically operating above 25°C and completing within 1 to 24 hours, which significantly reduces energy consumption compared to high-temperature processes. Furthermore, the broad substrate scope allows for the conversion of diaryl, aryl-alkyl, and even dialkyl olefins, providing a versatile platform for synthesizing diverse diketone intermediates required in pharmaceutical and agrochemical pipelines.
Mechanistic Insights into Ru-Catalyzed Olefin Oxidation
The efficacy of this transformation lies in the synergistic interaction between the Ruthenium center and the iodide co-catalyst, which facilitates a unique oxidative cycle. The Ruthenium species acts as the primary activation site for the oxidant, generating high-valent metal-oxo intermediates capable of attacking the electron-rich double bond of the olefin substrate. The presence of the iodide co-catalyst, such as tetrabutylammonium iodide or potassium iodide, is crucial as it likely participates in the regeneration of the active catalytic species and may assist in the oxygen transfer step, thereby enhancing the overall turnover number. This cooperative catalysis ensures that the reaction proceeds with high atom economy, minimizing the formation of over-oxidized byproducts or cleavage products that often plague non-selective oxidation methods. The mechanistic robustness allows the system to tolerate a wide array of electronic environments on the aromatic rings, from electron-donating methoxy groups to electron-withdrawing nitro and trifluoromethyl substituents.
From an impurity control perspective, this catalytic system offers distinct advantages by avoiding the formation of intractable inorganic sludge. Traditional methods using permanganate or chromate generate massive amounts of metal oxide waste that can trap organic products and complicate isolation. In the Ru-catalyzed process, the byproducts are primarily water or tert-butanol, which are easily separated during the aqueous workup. The high chemoselectivity ensures that sensitive functional groups such as esters, aldehydes, and halides remain intact, preserving the structural integrity of complex molecules. This precision is vital for multi-step syntheses where protecting group strategies add unnecessary cost and time. The ability to achieve high purity directly from the reaction mixture through simple extraction and chromatography demonstrates the superior profile of this technology for producing high-specification intermediates.
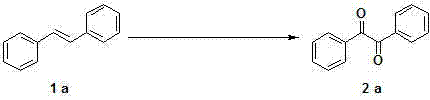
How to Synthesize 1,2-Diketones Efficiently
The implementation of this synthesis route is designed for operational ease, making it highly attractive for both laboratory scale-up and commercial production. The process begins with the precise charging of the Ruthenium catalyst and iodide promoter into a standard reaction vessel, followed by the addition of the olefin substrate and a biphasic or mixed solvent system comprising acetonitrile, toluene, and water. The oxidant is then introduced, and the mixture is heated under air, eliminating the need for inert gas purging which simplifies the engineering requirements. Detailed standardized synthesis steps see the guide below.
- Charge a reaction vessel with a Ruthenium catalyst (e.g., [Ru(cymene)Cl2]2, 1 mol%) and an iodide co-catalyst (e.g., KI, 30 mol%).
- Add the olefin substrate and a suitable solvent system, such as a mixture of acetonitrile, toluene, and water.
- Introduce the oxidant (e.g., hydrogen peroxide or tert-butyl hydroperoxide) and heat the mixture between 25°C and 100°C under air atmosphere until completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this Ru-catalyzed technology translates into tangible strategic benefits regarding cost structure and supply reliability. The shift from stoichiometric toxic oxidants to catalytic amounts of Ruthenium significantly alters the cost equation by reducing the volume of hazardous waste requiring disposal. Since the catalyst loading is as low as 1 mol%, the consumption of precious metal is minimized, mitigating the impact of volatile metal prices on the final product cost. Additionally, the use of commodity chemicals like hydrogen peroxide and common solvents ensures a stable supply chain that is not dependent on niche or regulated reagents. This stability is crucial for maintaining continuous production schedules and avoiding delays associated with the procurement of specialized oxidants.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the drastic reduction in waste treatment costs and the elimination of expensive stoichiometric reagents. By avoiding the use of selenium or osmium-based oxidants, manufacturers save substantially on hazardous waste disposal fees and regulatory compliance expenditures. The low catalyst loading further contributes to cost efficiency, as the expensive Ruthenium component is used sparingly while maintaining high turnover. Moreover, the simplified workup procedure, which often requires only extraction and silica gel adsorption, reduces solvent consumption and labor hours compared to the complex purification needed for traditional methods.
- Enhanced Supply Chain Reliability: The reliance on readily available, bulk-grade chemicals enhances the resilience of the supply chain. Reagents such as acetonitrile, toluene, and hydrogen peroxide are produced globally in massive quantities, ensuring that production is never halted due to raw material shortages. The robustness of the reaction conditions, which tolerate air and moisture to a significant extent, also reduces the risk of batch failures due to minor deviations in operational parameters. This reliability allows for more accurate forecasting and inventory management, ensuring that downstream customers receive their intermediates on time without unexpected disruptions.
- Scalability and Environmental Compliance: Scaling this process from grams to tons is facilitated by the mild reaction conditions and the absence of exothermic hazards associated with strong oxidants. The green nature of the chemistry aligns perfectly with increasingly stringent environmental regulations, future-proofing the manufacturing process against tighter emission standards. The reduction in toxic byproducts means that effluent treatment is simpler and less costly, supporting sustainable manufacturing goals. This environmental compliance is not just a regulatory necessity but a competitive advantage when supplying to multinational corporations with strict sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented oxidation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities.
Q: What are the primary advantages of this Ru-catalyzed method over traditional oxidation techniques?
A: Unlike traditional methods using toxic selenium dioxide or stoichiometric potassium permanganate which generate heavy metal waste, this Ru-catalyzed process utilizes greener oxidants like hydrogen peroxide and operates under mild conditions with high atom economy.
Q: Does this catalytic system tolerate sensitive functional groups on the olefin substrate?
A: Yes, the system exhibits excellent functional group compatibility, successfully oxidizing substrates containing halogens, esters, ethers, and heterocycles without significant degradation or side reactions.
Q: Is the catalyst loading economically viable for large-scale production?
A: The process is highly efficient, requiring only 1 mol% of the Ruthenium catalyst and 30 mol% of the iodide co-catalyst, which significantly reduces raw material costs compared to stoichiometric oxidation methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Diketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation pharmaceuticals and fine chemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are fully equipped to implement advanced catalytic technologies like the Ru-mediated oxidation described in CN102070383A, leveraging our rigorous QC labs to guarantee stringent purity specifications for every batch. Our commitment to quality ensures that the 1,2-diketones supplied meet the exacting standards required for API synthesis and complex molecule construction.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through innovative chemistry. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can accelerate your development timelines and enhance your product competitiveness in the global market.
