Advanced Metal-Free Oxidative Coupling for Commercial Scale-Up of Complex Triarylamine Skeletons
Advanced Metal-Free Oxidative Coupling for Commercial Scale-Up of Complex Triarylamine Skeletons
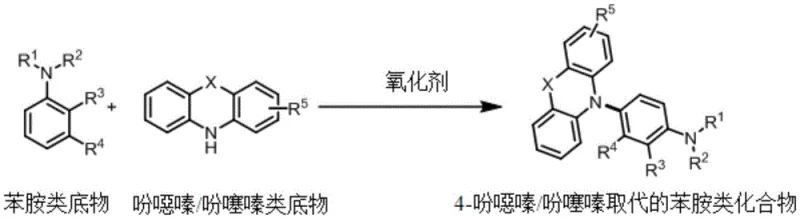
The landscape of organic photoelectric material synthesis is undergoing a significant transformation driven by the need for more sustainable and cost-effective manufacturing processes. Patent CN112266365A introduces a groundbreaking methodology for introducing phenoxazine and phenothiazine functional groups directly into the para-position of aniline derivatives. This innovation represents a paradigm shift from traditional transition metal-catalyzed cross-coupling reactions to a more atom-economical, dehydrogenative C(sp2)-H/N-H cross-coupling strategy. By leveraging inexpensive oxidants such as potassium persulfate under ambient air conditions, this technology eliminates the reliance on precious metal catalysts and pre-functionalized halogenated precursors. For R&D directors and procurement managers in the OLED and electrochromic industries, this development offers a compelling pathway to streamline supply chains and reduce the environmental footprint of producing high-value triarylamine intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of triarylamine skeletons, which are critical precursors for organic light-emitting diode (OLED) materials and electrochromic devices, has relied heavily on the Buchwald-Hartwig amination reaction. This conventional approach necessitates the use of halogenated aromatic hydrocarbons and diarylamines as starting materials, requiring a multi-step synthesis that begins with the energy-intensive and waste-generating halogenation of aromatic rings. Furthermore, the reaction mandates the addition of expensive noble metal catalysts like palladium or copper, along with specialized phosphine ligands and strong bases to facilitate the coupling. These stringent requirements not only inflate the raw material costs but also introduce significant challenges in downstream processing, particularly regarding the removal of toxic heavy metal residues to meet the rigorous purity specifications demanded by the electronic materials industry.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a direct oxidative cross-coupling strategy that bypasses the need for pre-functionalization entirely. By mixing an aniline substrate, a phenoxazine or phenothiazine substrate, and a cheap oxidant like potassium persulfate in a polar solvent such as acetonitrile, the reaction proceeds smoothly under an air atmosphere at room temperature. This method achieves the formation of the C-N bond through the activation of inert C(sp2)-H bonds and N-H bonds, resulting in high atom utilization and minimal waste generation. The simplicity of the protocol, which often completes within one hour without the need for inert gas protection, drastically simplifies the operational complexity and equipment requirements, making it highly attractive for large-scale industrial adoption.
Mechanistic Insights into Persulfate-Mediated Oxidative Coupling
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the persulfate oxidant. Under the reaction conditions, the persulfate anion undergoes homolytic cleavage or single-electron transfer to generate highly reactive sulfate radical anions. These radicals are capable of abstracting a hydrogen atom from the N-H bond of the phenothiazine or phenoxazine substrate, generating a nitrogen-centered radical species. Simultaneously, the electron-rich aniline derivative undergoes oxidation to form a radical cation. The subsequent coupling of these reactive intermediates, followed by deprotonation and rearomatization, leads to the formation of the desired C-N bond. This radical-mediated mechanism avoids the high energy barriers associated with traditional nucleophilic substitution, allowing the reaction to proceed under exceptionally mild thermal conditions while maintaining high selectivity for the para-position of the aniline ring.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed routes. The absence of transition metals eliminates the risk of metal-catalyzed side reactions such as homocoupling of the aryl halides or deboronation, which are common pitfalls in Suzuki or Buchwald-Hartwig chemistries. Furthermore, the use of air as the terminal oxidant source ensures that any radical byproducts are efficiently quenched or converted into benign inorganic salts like potassium sulfate, which are easily removed during the aqueous workup. The mild reaction temperature of 25°C further suppresses thermal degradation pathways, ensuring that sensitive functional groups on the substrate, such as nitriles or halogens, remain intact, thereby preserving the structural integrity required for downstream device performance.
How to Synthesize 4-Phenoxazine/Phenothiazine Substituted Aniline Efficiently
The practical implementation of this synthesis route is designed for ease of operation and scalability, making it accessible for both laboratory optimization and pilot plant production. The process involves a straightforward mixing of reagents in a standard reaction vessel, followed by a simple filtration and purification sequence. The robustness of the reaction conditions allows for flexibility in solvent choice, with acetonitrile proving to be the optimal medium, although methanol can also be employed effectively. For detailed procedural specifics regarding stoichiometry, workup protocols, and purification parameters, please refer to the standardized synthesis guide below.
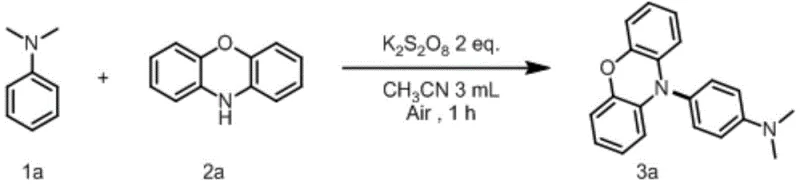
- Mix aniline substrate, phenoxazine/phenothiazine substrate, and potassium persulfate oxidant in acetonitrile solvent within a reaction vessel under air atmosphere.
- Stir the reaction mixture at room temperature (25°C) for approximately 1 hour to allow the dehydrogenative C(sp2)-H/N-H cross-coupling to proceed.
- Filter the reaction mixture, wash the residue with ethyl acetate, and purify the crude product via column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free oxidative coupling technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The elimination of noble metal catalysts and the reduction of synthetic steps directly address the volatility of raw material pricing and the logistical complexities associated with hazardous chemical handling. By shifting to a process that relies on commodity chemicals like persulfates and simple anilines, manufacturers can insulate their supply chains from the geopolitical and market fluctuations that often plague the supply of palladium and specialized ligands. This stability is crucial for maintaining consistent production schedules and meeting the demanding delivery timelines of the consumer electronics sector.
- Cost Reduction in Manufacturing: The economic impact of replacing palladium catalysts with inexpensive inorganic oxidants cannot be overstated, as it removes one of the most significant cost drivers in fine chemical synthesis. Additionally, the one-step nature of the reaction eliminates the need for intermediate isolation and purification stages, which reduces solvent consumption, energy usage, and labor hours significantly. The ability to run the reaction under air atmosphere further cuts costs by removing the requirement for expensive inert gas systems and the associated infrastructure maintenance, leading to substantial overall savings in the cost of goods sold for high-purity OLED material intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available, bulk commodity chemicals such as potassium persulfate and acetonitrile ensures a robust and resilient supply chain that is less susceptible to disruptions. Unlike specialized catalysts that may have limited suppliers and long lead times, the reagents for this process can be sourced from multiple global vendors, providing procurement teams with greater negotiating power and flexibility. The simplified logistics of handling non-hazardous oxidants compared to pyrophoric organometallic reagents also reduces regulatory burdens and transportation costs, facilitating smoother international trade and inventory management.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this process aligns perfectly with modern green chemistry principles, offering a clear path to sustainable manufacturing at scale. The high atom economy and the generation of benign inorganic byproducts minimize the volume of hazardous waste requiring treatment, thereby lowering disposal costs and simplifying compliance with increasingly stringent environmental regulations. The mild reaction conditions reduce the risk of thermal runaway incidents, enhancing plant safety, while the straightforward workup procedure facilitates easier scale-up from kilogram to tonne quantities without the need for complex engineering modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative coupling technology. These insights are derived directly from the experimental data and scope analysis presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations for potential adopters.
Q: Does this method require expensive transition metal catalysts?
A: No, the method described in patent CN112266365A utilizes cheap inorganic oxidants like potassium persulfate instead of noble metal catalysts such as palladium or copper, significantly reducing raw material costs and eliminating heavy metal residue concerns.
Q: Can this process handle sterically hindered triphenylamine substrates?
A: Yes, the technology successfully overcomes substrate limitations found in prior art, enabling the direct oxidative cross-coupling of bulky triphenylamine substrates with phenothiazine derivatives to form complex triarylamine skeletons efficiently.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction operates under mild conditions, typically at room temperature (25°C) in an air atmosphere using acetonitrile as the solvent, completing the transformation in as little as 1 hour without the need for inert gas protection.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenothiazine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the next generation of organic photoelectric materials. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients can transition seamlessly from laboratory discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, essential for the high-performance requirements of OLED and electrochromic applications. We are committed to delivering high-purity phenothiazine derivatives that meet the exacting standards of the global electronics industry.
We invite you to collaborate with us to leverage this innovative technology for your specific product needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing processes, demonstrating how this new route can optimize your expenditure. Please contact our technical procurement team today to request specific COA data for our available intermediates and to discuss route feasibility assessments for your target molecules, ensuring a secure and efficient supply chain for your future projects.
