Revolutionizing Pharmaceutical Intermediate Production: Scalable and Cost-Efficient Synthesis of High-Purity Trifluoromethyl Triazole Derivatives
The recently granted Chinese patent CN111978265B represents a significant advancement in the synthesis of biologically critical heterocyclic compounds, specifically introducing an innovative route for producing high-purity 5-trifluoromethyl substituted 1,2,4-triazole derivatives. This breakthrough methodology addresses longstanding challenges in pharmaceutical intermediate manufacturing by leveraging iron(III) chloride catalysis under ambient conditions—a stark contrast to conventional approaches requiring specialized equipment and stringent environmental controls. The patent demonstrates how strategic catalyst selection enables efficient cyclization while maintaining exceptional substrate flexibility across diverse molecular architectures. Crucially, this development emerges at a pivotal moment when global pharmaceutical manufacturers increasingly demand reliable sources of complex heterocyclic building blocks with stringent purity requirements. By eliminating traditional barriers to scalable production, this technology positions itself as a transformative solution for modern drug development pipelines where molecular complexity must be balanced with commercial viability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for trifluoromethyl-substituted triazoles have historically been plagued by multiple critical constraints that severely limit their industrial applicability. The first four established methods—namely condensation reactions involving dioxadiazoles with primary amines, cyclization of trifluoromethyl hydrazides with amidines, cyclization of substituted hydrazones with nitriles, and hydrazinolysis of oxadiazoles—all require harsh reaction conditions including elevated temperatures under inert atmospheres or specialized pressure equipment. These processes typically involve lengthy multi-step sequences that generate significant impurities requiring complex purification protocols. Furthermore, their narrow substrate scope restricts molecular diversity while consistently delivering suboptimal yields that fail to meet commercial production demands. The fifth previously developed method using trifluoroethylimidoyl chloride showed promise but exhibited critical limitations by excluding alkyl hydrazones from participating in the reaction pathway—thereby preventing access to valuable 3-alkyl fluoro triazole derivatives essential for next-generation pharmaceutical applications.
The Novel Approach
This patent introduces a fundamentally different paradigm by utilizing inexpensive sodium bicarbonate and iron(III) chloride as dual promoters in a simplified two-stage reaction sequence that operates effectively under ambient air conditions. The methodology begins with base-promoted intermolecular carbon-nitrogen bond formation between readily available trifluoroethyl imidoyl chloride and hydrazide precursors at moderate temperatures between 30–50°C over an extended period. This is followed by Lewis acid-mediated intramolecular dehydration cyclization at elevated temperatures using iron(III) chloride—a catalyst chosen for its exceptional cost-effectiveness and environmental profile compared to precious metal alternatives. The process demonstrates remarkable functional group tolerance across diverse aryl and alkyl substituents while maintaining consistent high yields without requiring specialized equipment or hazardous reagents. This strategic combination of accessible starting materials and operationally simple conditions creates an unprecedented pathway for producing structurally complex triazole derivatives at commercial scale.
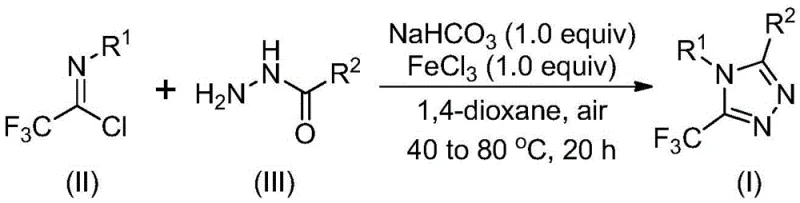
Mechanistic Insights into FeCl3-Catalyzed Triazole Formation
The reaction mechanism proceeds through a carefully orchestrated sequence where sodium bicarbonate first facilitates deprotonation and nucleophilic attack by the hydrazide nitrogen on the imidoyl chloride carbon center. This initial step forms a key trifluoroacetamidine intermediate through intermolecular carbon-nitrogen bond formation under mild thermal conditions that prevent undesired side reactions. The subsequent addition of iron(III) chloride activates the carbonyl oxygen through Lewis acid coordination, significantly lowering the energy barrier for intramolecular cyclization while simultaneously promoting dehydration through chloride-assisted proton transfer. This dual activation pathway creates a highly selective route to the triazole ring system where the iron catalyst not only accelerates the rate-determining step but also directs regioselectivity toward the desired substitution pattern at the C5 position. The mechanism's inherent efficiency stems from synergistic interactions between the base promoter and Lewis acid catalyst that minimize competing decomposition pathways.
Impurity control is achieved through multiple built-in molecular safeguards within this catalytic cycle. The mild reaction conditions prevent thermal degradation of sensitive functional groups commonly found in pharmaceutical intermediates while the selective activation pathway suppresses common side reactions such as over-chlorination or hydrolysis that plague alternative methods. The use of non-coordinating solvents like dioxane further enhances selectivity by avoiding catalyst poisoning while maintaining optimal solubility profiles across diverse substrate combinations. Crucially, the absence of transition metals eliminates concerns about heavy metal contamination—a critical requirement for pharmaceutical applications where even trace impurities can compromise final product quality. This inherent selectivity profile enables consistent production of high-purity derivatives meeting stringent regulatory standards without requiring additional purification steps beyond standard chromatographic techniques.
How to Synthesize High-Purity Trifluoromethyl Triazoles Efficiently
This patent provides a robust framework for synthesizing complex triazole derivatives through a meticulously optimized process that balances scientific precision with practical manufacturing considerations. The methodology represents a significant departure from conventional approaches by eliminating specialized equipment requirements while maintaining exceptional product quality—making it particularly valuable for pharmaceutical manufacturers seeking reliable sources of high-value intermediates. The following standardized procedure details the precise implementation steps required to achieve consistent results across various production scales while maintaining strict adherence to quality control parameters essential for regulatory compliance.
- Combine sodium bicarbonate (1.0 equiv), trifluoroethyl imidoyl chloride (II), and hydrazide (III) in anhydrous dioxane solvent under ambient air conditions at controlled temperatures between 40–80°C for the initial reaction phase.
- Introduce iron(III) chloride catalyst at stoichiometric equivalence and elevate reaction temperature to optimize cyclization kinetics while maintaining atmospheric pressure conditions.
- Execute straightforward purification through silica gel column chromatography following simple filtration to achieve stringent purity specifications required for pharmaceutical applications.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial strategic value across procurement and supply chain operations by addressing fundamental pain points inherent in traditional intermediate sourcing strategies. The elimination of specialized environmental controls significantly reduces capital expenditure requirements while enhancing operational flexibility across diverse manufacturing sites worldwide. By leveraging commercially available starting materials with established global supply chains, this approach minimizes vulnerability to single-source dependencies that frequently disrupt pharmaceutical production timelines. The process design inherently supports just-in-time manufacturing models through its rapid setup capabilities and minimal equipment requirements—enabling manufacturers to respond dynamically to fluctuating market demands without costly reconfiguration.
- Cost Reduction in Manufacturing: The strategic elimination of transition metal catalysts removes both direct material costs associated with precious metals and substantial downstream processing expenses required for metal residue removal; this dual benefit creates significant cost savings while simplifying quality control protocols through reduced analytical testing requirements for heavy metals.
- Enhanced Supply Chain Reliability: Utilization of widely available starting materials with established global distribution networks ensures consistent raw material availability while minimizing exposure to geopolitical supply risks; the process's tolerance for ambient air conditions further enhances reliability by eliminating dependencies on specialized gas handling systems that frequently cause production delays.
- Scalability and Environmental Compliance: The straightforward reaction sequence demonstrates exceptional scalability from laboratory development through commercial production without requiring process re-engineering; its inherent simplicity reduces waste generation through fewer processing steps while eliminating hazardous reagents—thereby supporting sustainability initiatives without compromising economic viability.
Frequently Asked Questions (FAQ)
The following technical questions address critical considerations raised by procurement specialists and R&D teams during initial evaluations of this patented methodology; each response is grounded in experimental data from the patent's detailed implementation examples while highlighting practical implications for commercial adoption.
Q: How does this method overcome limitations of conventional triazole synthesis routes?
A: The novel approach eliminates stringent anhydrous and oxygen-free requirements while utilizing cost-effective starting materials through a streamlined two-stage reaction sequence; this significantly reduces operational complexity compared to traditional methods that suffer from narrow substrate scope and harsh reaction conditions.
Q: What scalability advantages does this iron-catalyzed process offer?
A: The reaction demonstrates exceptional scalability from laboratory to commercial production due to its tolerance of ambient air conditions and simplified purification protocol; this enables seamless transition from gram-scale to multi-kilogram batches without specialized equipment or complex process modifications.
Q: How does this synthesis ensure high purity for pharmaceutical applications?
A: The mechanism inherently minimizes impurity formation through selective cyclization pathways while leveraging readily available starting materials with broad functional group tolerance; this provides precise control over product quality without requiring additional purification steps beyond standard column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable High-Purity Trifluoromethyl Triazole Derivative Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities; this proven capability ensures seamless transition from development to full-scale manufacturing without compromising quality or delivery timelines. As a specialized CDMO partner with dedicated expertise in complex heterocyclic chemistry, we combine deep technical knowledge with state-of-the-art facilities featuring rigorous QC labs that implement comprehensive analytical protocols for every batch produced. Our commitment to quality extends beyond mere compliance—we actively collaborate with clients to optimize processes for maximum efficiency while ensuring complete regulatory documentation support throughout the product lifecycle.
Leverage our technical procurement team's expertise through a Customized Cost-Saving Analysis that identifies specific opportunities within your current supply chain; we welcome inquiries requesting detailed COA data and route feasibility assessments tailored to your unique manufacturing requirements—enabling informed decision-making based on actual technical capabilities rather than theoretical projections.
