Advanced Chiral Copper Catalysis for High-Purity Pharmaceutical Intermediate Manufacturing
Introduction to Breakthrough Chiral Copper Catalysis
The landscape of asymmetric synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign catalytic systems. A significant advancement in this field is detailed in Chinese Patent CN111423351A, which introduces a novel class of chiral copper complexes designed specifically for the asymmetric Mukaiyama Aldol reaction. This technology addresses critical bottlenecks in the synthesis of chiral 3-hydroxy-2-oxoindole compounds, which are privileged scaffolds in medicinal chemistry. The core innovation lies in the structural design of the copper complex, represented by general formula (I), which utilizes specific chiral ligands to create a highly defined stereochemical environment around the copper center. Unlike traditional methods that often rely on expensive rare-earth metals or suffer from strict moisture sensitivity, this copper-based system offers a robust alternative that maintains high enantioselectivity and diastereoselectivity even under less stringent conditions.
For R&D directors and process chemists, the implications of this patent are profound. The ability to utilize a copper catalyst that tolerates water additives not only simplifies the reaction setup but also opens new avenues for process optimization that were previously inaccessible with moisture-sensitive Lewis acids. The patent explicitly demonstrates that the stereoselectivity of the product is preserved even when the reaction is scaled up to the gram level, a crucial metric for assessing the viability of a synthetic route for commercial manufacturing. By leveraging this technology, pharmaceutical manufacturers can achieve reliable access to high-purity pharmaceutical intermediates with reduced dependency on exotic reagents, thereby stabilizing the supply chain for critical drug substances.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral 3-hydroxy-2-oxoindole cores via aldol-type reactions has faced significant hurdles regarding catalyst cost and operational sensitivity. Conventional approaches often employed scandium(III)-pyridine-bisoxazoline complexes or bifunctional thiourea organocatalysts. While effective in academic settings, scandium-based systems present a substantial economic burden due to the high cost of the metal and the specialized ligands required. Furthermore, many traditional Lewis acid-catalyzed Mukaiyama Aldol reactions are exquisitely sensitive to moisture, necessitating the use of rigorously dried solvents and inert atmosphere techniques that increase both capital expenditure and operational time. This sensitivity often leads to batch-to-batch variability when scaling up, as trace amounts of water can deactivate the catalyst or promote background non-selective reactions, resulting in lower yields and compromised optical purity.
The Novel Approach
The methodology disclosed in CN111423351A represents a paradigm shift by utilizing a chiral divalent copper complex that overcomes these traditional limitations. The novel approach employs readily available copper salts, such as copper hexafluoroantimonate or copper trifluoromethanesulfonate, coordinated with specially designed chiral ligands (LA or LB) containing pyrrolidine and phenolic moieties. This combination creates a catalytic species that is not only highly active but also remarkably tolerant to protic additives. The patent data reveals that the addition of water, typically a poison for Lewis acid catalysis, actually enhances the reaction rate and selectivity in this specific system. This counter-intuitive finding allows for cost reduction in pharmaceutical intermediate manufacturing by relaxing the specifications for solvent dryness. Moreover, the system demonstrates exceptional substrate scope, accommodating isatins with various electronic and steric properties, ensuring that the process is versatile enough for the synthesis of diverse API precursors without requiring extensive re-optimization for each new analog.
Mechanistic Insights into Copper-Catalyzed Asymmetric Mukaiyama Aldol Reaction
The high level of stereocontrol observed in this transformation is attributed to the precise three-dimensional arrangement of the chiral ligand around the copper center. The ligand, featuring a pyrrolidine ring linked to a phenolic group with specific aryl substitutions (Ar1 and Ar2), coordinates to the copper atom to form a rigid chiral pocket. When the isatin substrate binds to this activated copper complex, it is held in a specific orientation that favors nucleophilic attack from one specific face of the carbonyl group. The silyl enol ether, acting as the nucleophile, approaches the activated electrophile through a transition state that is energetically favored due to minimization of steric clashes and maximization of electronic interactions within the chiral environment. This mechanistic pathway ensures that the newly formed carbon-carbon bond is established with high diastereoselectivity, typically favoring the syn- or anti-isomer depending on the specific ligand geometry employed.
Furthermore, the impurity profile of the reaction is tightly controlled by the catalyst's specificity. In conventional non-catalyzed or poorly catalyzed aldol reactions, competing pathways such as self-condensation of the enol silane or polymerization of the isatin can lead to complex mixtures that are difficult to purify. However, the chiral copper complex described in this patent accelerates the desired cross-aldol pathway significantly faster than these side reactions. The presence of the nitrogen-containing organic base, such as N-ethylmorpholine, likely plays a dual role in activating the silyl enol ether and stabilizing the copper species, further suppressing the formation of undesired byproducts. This high chemoselectivity translates directly to simplified downstream processing, as the crude reaction mixture contains fewer impurities, reducing the load on purification columns and increasing the overall recovery of the target high-purity pharmaceutical intermediate.
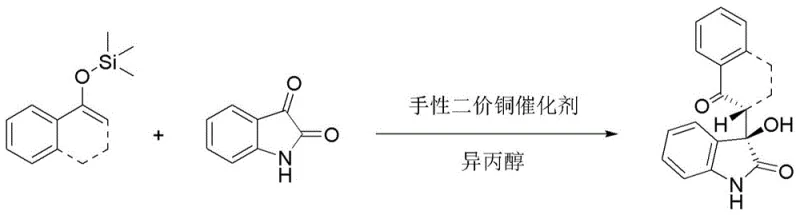
How to Synthesize Chiral 3-Hydroxy-2-oxoindoles Efficiently
The practical implementation of this catalytic system is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins with the in-situ generation of the active catalyst species by mixing the copper salt, the chiral ligand, and the base in a suitable solvent like isopropanol. This mixture is stirred at low temperatures to ensure proper complexation before the substrates are introduced. The reaction is then initiated by the addition of the isatin and the silyl enol ether, optionally with a controlled amount of water to boost performance. The entire process operates under mild conditions, typically around -10°C, which balances reaction rate with stereocontrol. For a detailed breakdown of the specific molar ratios, solvent choices, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Prepare the chiral copper catalyst by mixing a divalent copper salt (e.g., Cu(SbF6)2), a chiral ligand (LA or LB), and a nitrogen-containing organic base in a solvent like isopropanol at temperatures between -20°C and 10°C.
- Add the silyl enol ether substrate and the isatin derivative to the pre-formed catalyst mixture. Optionally, add a small amount of water or acidic additive to enhance reaction rate and selectivity.
- Maintain the reaction at approximately -10°C until completion, then perform standard workup procedures including extraction, drying, and purification via column chromatography to isolate the high-purity chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this chiral copper catalysis technology offers compelling strategic advantages that extend beyond mere chemical efficiency. The shift from precious or rare-earth metals to abundant copper significantly alters the raw material cost structure, providing a buffer against market volatility associated with scarce metal prices. Additionally, the operational simplicity afforded by the system's water tolerance reduces the logistical burden of sourcing and storing ultra-dry solvents, which are often more expensive and hazardous to handle. This relaxation of process constraints allows for more flexible manufacturing scheduling and reduces the risk of production delays caused by reagent quality issues, thereby enhancing overall supply chain reliability for critical drug intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive scandium or specialized organocatalysts with commodity copper salts results in a drastic reduction in catalyst loading costs. Since copper is orders of magnitude cheaper than rare-earth alternatives, the direct material cost for the catalytic system is substantially lowered. Furthermore, the elimination of stringent drying protocols for solvents and reagents reduces energy consumption and equipment wear, contributing to significant operational expenditure savings. The high selectivity of the reaction also minimizes waste generation, lowering the costs associated with waste disposal and solvent recovery, which are major cost drivers in fine chemical production.
- Enhanced Supply Chain Reliability: Copper salts and the requisite organic ligands are commercially available from multiple global suppliers, mitigating the risk of single-source dependency that often plagues proprietary catalyst systems. The robustness of the reaction against moisture means that the process is less susceptible to environmental fluctuations, ensuring consistent batch quality regardless of seasonal humidity changes. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream API manufacturers, effectively reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The patent data confirms that the stereoselectivity is maintained upon scaling to gram quantities, indicating a smooth path to kilogram and ton-scale production without the need for extensive re-engineering. The use of greener solvents like isopropanol and the reduction of hazardous waste due to high atom economy align with modern environmental, social, and governance (ESG) goals. This compliance facilitates easier regulatory approval and supports the company's commitment to sustainable manufacturing practices, making the supply of these complex pharmaceutical intermediates more resilient and future-proof.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral copper catalysis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing processes. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: How does the water tolerance of this copper catalyst impact manufacturing costs?
A: The catalyst's unique tolerance to water eliminates the need for rigorous solvent drying and strictly anhydrous conditions, significantly reducing operational complexity and energy consumption associated with solvent purification in large-scale API intermediate production.
Q: What is the substrate scope for this asymmetric Mukaiyama Aldol reaction?
A: The method exhibits broad substrate compatibility, successfully accommodating isatins with electron-withdrawing or electron-donating groups at the 4, 5, and 6 positions, as well as various N-substituted isatins, ensuring versatility for diverse pharmaceutical pipelines.
Q: Can this catalytic process be scaled up for commercial production?
A: Yes, the patent data confirms successful gram-scale synthesis (10 mmol scale) where high enantioselectivity (up to 99% ee) and diastereoselectivity were maintained, demonstrating strong potential for commercial scale-up without loss of stereochemical integrity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Copper Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the chiral copper complex described in CN111423351A for the synthesis of vital pharmaceutical building blocks. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results observed in the laboratory can be seamlessly translated into industrial reality. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of handling sensitive chiral separations and analyses, guaranteeing that every batch of chiral 3-hydroxy-2-oxoindole intermediates meets the highest standards of optical purity and chemical quality required by global regulatory agencies.
We invite you to collaborate with our technical team to explore how this cost-effective and robust copper-catalyzed route can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for similar chiral intermediates and to discuss route feasibility assessments that could accelerate your project timelines and reduce your overall cost of goods sold.
