Advanced Chiral Copper Complexes for High-Purity Pharmaceutical Intermediate Manufacturing
Advanced Chiral Copper Complexes for High-Purity Pharmaceutical Intermediate Manufacturing
The landscape of asymmetric synthesis in the pharmaceutical industry is constantly evolving, driven by the relentless demand for higher enantiomeric purity and more sustainable catalytic processes. Patent CN103467313A represents a significant technological breakthrough in this domain by disclosing a novel chiral copper complex, specifically Bis[(S)-phenylglycinol] cuprous chloride, which serves as a highly effective catalyst for carbon-carbon bond-forming reactions. This innovation addresses critical bottlenecks in the production of chiral pharmaceutical intermediates, particularly those requiring nitroaldol (Henry) transformations. The disclosed technology leverages the unique coordination chemistry of copper(II) with chiral amino alcohol ligands to create a robust catalytic system that operates under remarkably mild conditions. For R&D directors and process chemists, this patent offers a compelling alternative to traditional noble metal catalysts, providing a pathway to reduce reliance on expensive precious metals while maintaining rigorous stereochemical control. The ability to achieve a 66% conversion rate in the Henry reaction of benzaldehyde at room temperature underscores the practical viability of this complex for large-scale applications.
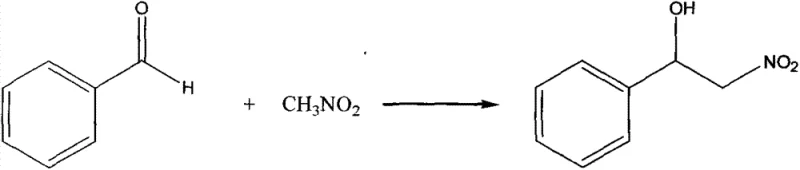
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral nitroaldol products has relied heavily on complex multi-step ligand synthesis or the use of stoichiometric amounts of chiral auxiliaries, which significantly inflate the cost of goods sold (COGS) and generate substantial chemical waste. Conventional Lewis acid catalysts often require stringent anhydrous conditions, cryogenic temperatures, or inert atmospheres to prevent catalyst deactivation, thereby imposing heavy energy burdens on the manufacturing process. Furthermore, many existing copper-based systems suffer from poor solubility in green solvents or lack the necessary structural rigidity to induce high levels of enantioselectivity, leading to difficult downstream purification challenges. The reliance on exotic, non-commercially available ligands also introduces supply chain vulnerabilities, where a single supplier disruption can halt entire production lines. These factors collectively create a high barrier to entry for the cost-effective manufacturing of high-value chiral intermediates, necessitating a shift towards more robust and accessible catalytic technologies.
The Novel Approach
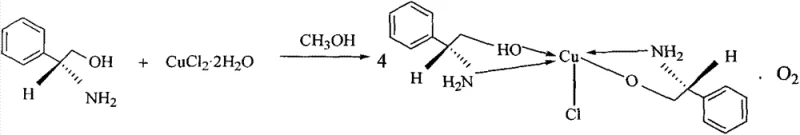
The methodology outlined in patent CN103467313A revolutionizes this landscape by introducing a direct, one-step coordination strategy that utilizes readily available L-phenylglycinol and copper chloride dihydrate. This approach eliminates the need for pre-synthesized, complex ligands, drastically simplifying the catalyst preparation workflow. By reacting the amino alcohol and metal salt in a 3:1 molar ratio in anhydrous methanol under reflux for 48 hours, the process yields stable blue crystals of the active complex with high reproducibility. The use of methanol as a solvent is particularly advantageous from a procurement perspective, as it is a commodity chemical with a stable global supply chain and low toxicity profile compared to chlorinated solvents. This streamlined synthesis not only reduces the operational complexity but also enhances the safety profile of the manufacturing process, making it an ideal candidate for adoption by facilities aiming to improve their environmental, social, and governance (ESG) metrics while achieving cost reduction in fine chemical manufacturing.
Mechanistic Insights into Chiral Copper-Catalyzed Henry Reaction
A deep understanding of the coordination geometry is essential for optimizing the catalytic performance of this novel complex. Crystallographic analysis reveals that the copper center adopts a distorted square pyramidal or trigonal bipyramidal geometry, coordinated by nitrogen and oxygen atoms from the phenylglycinol ligands and a chloride ion. The specific bond lengths, such as Cu-N distances ranging approximately from 1.99 Å to 2.02 Å and Cu-O distances around 1.95 Å to 1.98 Å, indicate strong chelation that locks the ligand into a rigid conformation. This rigidity is crucial for creating a well-defined chiral pocket around the metal center, which effectively discriminates between the prochiral faces of the incoming aldehyde substrate during the nucleophilic attack by the nitronate ion. The presence of the chloride ion as a axial or equatorial ligand further modulates the Lewis acidity of the copper center, fine-tuning its reactivity to balance between activation of the electophile and stabilization of the transition state.
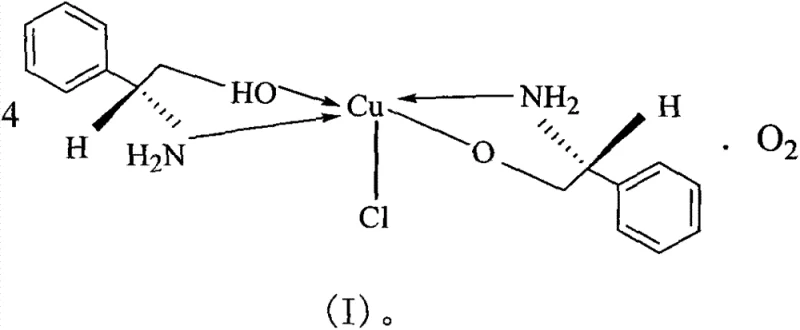
From an impurity control perspective, the stability of this coordination complex plays a pivotal role in ensuring product quality. Unlike labile catalysts that may decompose into free metal ions or racemic ligand species under reaction conditions, this complex maintains its structural integrity throughout the catalytic cycle. This stability minimizes the formation of background racemic products, which are often the most difficult impurities to remove via crystallization or chromatography. The specific spatial arrangement of the phenyl rings in the ligand framework provides steric bulk that shields the active site from non-selective interactions, thereby enhancing the enantiomeric excess of the final product. For quality assurance teams, this means a cleaner reaction profile with fewer by-products, reducing the burden on analytical laboratories and shortening the overall batch release timeline. The mechanistic robustness ensures that the process remains consistent even when scaling from gram-scale laboratory experiments to multi-kilogram pilot runs.
How to Synthesize Bis[(S)-phenylglycinol] Cuprous Chloride Efficiently
The synthesis protocol described in the patent is designed for operational simplicity, requiring standard glassware and heating equipment commonly found in any process development laboratory. The procedure involves dissolving the chiral amino alcohol in methanol followed by the addition of the copper salt, a sequence that ensures homogeneous mixing before the coordination event occurs. Maintaining the precise 3:1 molar ratio is critical to driving the equilibrium towards the formation of the desired tetranuclear or polynuclear cluster structure observed in the crystal data. The extended reflux time of 48 hours allows for the slow rearrangement of the coordination sphere into the thermodynamically most stable configuration, which correlates with the highest catalytic activity. Detailed standardized synthesis steps see the guide below.
- Dissolve L-phenylglycinol (0.03mol) in anhydrous methanol (50ml) within a 100ml round-bottom flask under stirring.
- Add copper chloride dihydrate (0.01mol) to the solution maintaining a strict 3: 1 molar ratio of ligand to metal.
- Heat the mixture to reflux for 48 hours, then filter while hot and allow natural volatilization to precipitate blue crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers tangible strategic benefits beyond mere technical performance. The primary advantage lies in the decoupling of catalyst production from complex organic synthesis supply chains; since the precursors L-phenylglycinol and copper chloride are bulk commodities, their pricing is stable and less susceptible to the volatility seen with specialized chiral ligands. This stability allows for more accurate long-term budgeting and reduces the risk of cost overruns due to raw material price spikes. Furthermore, the simplified synthesis route reduces the number of unit operations required to produce the catalyst itself, translating directly into lower labor costs and reduced equipment occupancy time. The ability to source raw materials from multiple global suppliers enhances supply chain resilience, ensuring continuity of operations even during regional disruptions or logistics bottlenecks.
- Cost Reduction in Manufacturing: The elimination of expensive noble metals like palladium or rhodium in favor of abundant copper significantly lowers the raw material cost base for the catalytic system. Additionally, the use of methanol as a reaction solvent facilitates easier recovery and recycling through standard distillation processes, minimizing solvent purchase costs and waste disposal fees. The high stability of the catalyst potentially allows for reuse or recycling in certain heterogeneous variations, further amortizing the cost of the catalyst over multiple batches. These factors combine to create a substantially lower cost of goods sold, improving the overall margin profile for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: By utilizing a catalyst derived from widely available feedstock chemicals, manufacturers can mitigate the risks associated with single-source dependencies often found in the specialty chemical sector. The robust nature of the synthesis protocol means that production can be easily transferred between different manufacturing sites without the need for highly specialized equipment or proprietary technology transfers. This flexibility supports a distributed manufacturing strategy, allowing companies to regionalize production closer to end markets to reduce lead times and transportation costs. The reliability of the supply chain is further bolstered by the fact that the key chiral building block, L-phenylglycinol, is produced at scale for various other pharmaceutical applications.
- Scalability and Environmental Compliance: The process operates at atmospheric pressure and moderate temperatures, removing the need for high-pressure reactors or extensive cooling infrastructure, which simplifies the engineering requirements for scale-up. From an environmental standpoint, the reduction in heavy metal usage and the potential for solvent recycling align with increasingly stringent global regulations regarding chemical manufacturing emissions and waste. The solid-state isolation of the catalyst via crystallization avoids the generation of aqueous waste streams associated with extraction-based purification methods. This green chemistry profile not only reduces regulatory compliance costs but also enhances the corporate sustainability image, which is becoming a critical factor in vendor selection for major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral copper complex technology. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation to ensure accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this catalyst into existing production workflows. We encourage technical teams to review the specific crystallographic parameters and reaction conditions to fully appreciate the nuances of this system.
Q: What is the catalytic efficiency of this chiral copper complex in Henry reactions?
A: According to patent CN103467313A, the complex demonstrates a conversion rate of 66% in the nitroaldol reaction of benzaldehyde under mild room temperature conditions.
Q: How does the structural geometry contribute to enantioselectivity?
A: The complex forms a rigid chiral pocket through specific Cu-N and Cu-O coordination bonds, creating a steric environment that favors the formation of specific enantiomers during nucleophilic attack.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the one-step reflux method in methanol utilizes common solvents and reagents, eliminating the need for exotic ligands or extreme pressures, which facilitates straightforward commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Copper Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the chiral copper complex described in patent CN103467313A for the next generation of pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including detailed chiral HPLC analysis and trace metal testing to guarantee the quality of every batch. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated process engineering team works closely with clients to optimize reaction parameters for maximum yield and minimal impurity formation.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this copper-based catalytic system for your specific molecule. Please contact us to request specific COA data and route feasibility assessments tailored to your target compound. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner committed to delivering high-purity chiral intermediates with the speed and flexibility required in today's competitive market.
