Revolutionizing Nitrile Production Through Cobalt-Catalyzed Dehydration for Scalable Pharmaceutical Intermediate Manufacturing
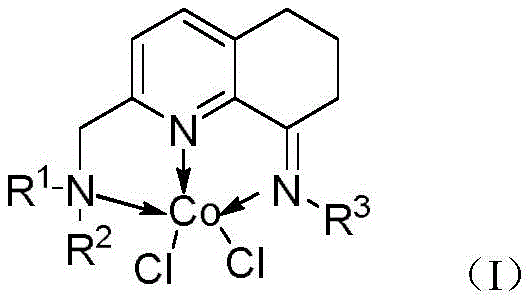
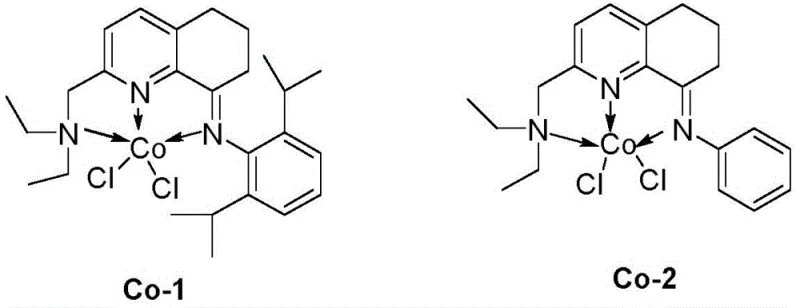
The groundbreaking Chinese patent CN112961079A introduces an innovative cobalt-catalyzed methodology for converting primary amides into valuable nitrile compounds through dehydration chemistry under remarkably mild conditions This significant advancement addresses long-standing challenges in synthetic chemistry where traditional approaches required harsh dehydrating agents like phosphorus pentoxide or thionyl chloride which often led to decomposition of sensitive functional groups The novel process utilizes an amine pyridine imine tridentate nitrogen ligand cobalt complex as catalyst combined with silane-based dehydrating agents enabling efficient transformation at temperatures as low as 60°C while maintaining exceptional substrate compatibility across diverse molecular architectures This represents a paradigm shift in nitrile synthesis methodology particularly for pharmaceutical intermediates where structural complexity demands gentle yet effective transformation protocols The technology demonstrates particular promise for manufacturing high-value intermediates requiring stringent purity specifications due to its inherent selectivity and minimal byproduct formation characteristics
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional dehydration methods employing strong acids such as P₂O₅ POCl₃ SOCl₂ or TiCl₄ operate under extremely harsh conditions that frequently cause decomposition of thermally sensitive substrates commonly encountered in pharmaceutical synthesis These aggressive reagents also generate substantial amounts of hazardous waste requiring complex disposal procedures that significantly increase environmental compliance costs while creating operational bottlenecks in manufacturing facilities More recent approaches using ethyl dichlorophosphate with DBU or pivaloyl chloride-pyridine mixtures offer milder conditions but introduce new challenges including difficult reagent storage requirements complicated handling procedures and generation of large volumes of organic waste streams that necessitate expensive treatment protocols Transition metal catalyzed systems previously reported often require high catalyst loadings up to ten mol percent particularly with palladium systems which substantially increases raw material costs while creating additional purification challenges due to residual metal contamination concerns Furthermore many existing transition metal approaches exhibit poor compatibility with heterocyclic substrates such as pyridine rings which are prevalent in modern pharmaceutical compounds thereby limiting their applicability across diverse molecular scaffolds
The Novel Approach
The patented cobalt-catalyzed methodology overcomes these limitations through its innovative use of an amine pyridine imine tridentate nitrogen ligand cobalt complex which operates effectively at low catalyst loadings while maintaining exceptional stability throughout the reaction process This system utilizes readily available silane dehydrating agents such as polymethylhydrosiloxane which offer superior handling characteristics compared to traditional corrosive reagents while generating minimal waste byproducts The reaction proceeds under mild thermal conditions between sixty and one hundred degrees Celsius with optimal performance observed at sixty degrees Celsius for six hours thereby eliminating thermal degradation risks associated with conventional methods Crucially this approach demonstrates remarkable substrate tolerance including successful conversion of heterocyclic compounds like pyridine derivatives which previously failed under other transition metal catalysis systems The simplified workup procedure involving ethyl acetate quenching followed by straightforward column chromatography significantly reduces processing time while enhancing overall operational efficiency compared to multi-step purification protocols required by alternative methodologies
Mechanistic Insights into Cobalt-Catalyzed Dehydration
The catalytic cycle begins with coordination of the primary amide substrate to the cobalt center through nitrogen lone pair interaction followed by activation through hydride transfer from sodium triethylborohydride which generates a key cobalt-hydride intermediate This intermediate facilitates deprotonation of the amide nitrogen creating a nucleophilic species that attacks silicon from polymethylhydrosiloxane forming an O-silylated intermediate The subsequent elimination step releases water equivalent while regenerating the active cobalt species through reductive elimination pathways The tridentate nitrogen ligand architecture provides exceptional geometric control around the cobalt center ensuring precise orientation of substrates while preventing unwanted side reactions through steric protection mechanisms This ligand framework also stabilizes multiple oxidation states of cobalt during catalytic turnover enabling efficient redox cycling without catalyst decomposition The rigid molecular framework maintains structural integrity throughout repeated catalytic cycles contributing significantly to the observed high turnover numbers

Impurity control mechanisms are inherently built into this catalytic system through precise steric and electronic regulation by the tridentate ligand architecture which prevents over-reduction pathways commonly observed with alternative methods The ligand design creates a protected pocket around the active site that selectively accommodates primary amides while excluding potential side-reaction pathways involving other functional groups present in complex molecules This spatial control minimizes formation of aldehyde or carboxylic acid byproducts that typically arise from hydrolysis or over-reduction side reactions The mild reaction conditions further suppress thermal decomposition pathways that could generate colored impurities or other quality-defecting species particularly critical for pharmaceutical intermediates requiring high purity specifications The consistent performance across diverse substrates including electron-rich heterocycles demonstrates exceptional selectivity control that directly translates to reduced impurity profiles compared to conventional dehydration techniques
How to Synthesize Nitriles Efficiently
This patented methodology represents a significant advancement in nitrile synthesis technology offering pharmaceutical manufacturers an efficient pathway to produce high-value intermediates through cobalt-catalyzed dehydration chemistry Detailed standardized synthesis procedures including precise reagent ratios temperature profiles and purification protocols have been developed based on extensive optimization studies documented in CN112961079A The following section provides comprehensive step-by-step guidance for implementing this innovative process in manufacturing environments ensuring consistent product quality while maximizing operational efficiency across various production scales
- Combine primary amide substrate with polymethylhydrosiloxane dehydrating agent, sodium triethylborohydride activator, and cobalt complex catalyst under inert atmosphere using toluene solvent
- Heat reaction mixture at precisely controlled temperature between 60°C and 100°C for duration ranging from six to twenty-four hours
- Quench with ethyl acetate followed by concentration and purification through silica gel column chromatography using petroleum ether/ethyl acetate eluent
Commercial Advantages for Procurement and Supply Chain Teams
This innovative cobalt-catalyzed dehydration process delivers substantial commercial benefits specifically addressing critical pain points faced by procurement and supply chain professionals in pharmaceutical manufacturing environments By eliminating reliance on hazardous traditional dehydrating agents while utilizing cost-effective cobalt-based catalysis this methodology creates significant opportunities for operational improvement across multiple dimensions including raw material sourcing processing efficiency and environmental compliance The following analysis details how this technology directly supports strategic business objectives related to cost management supply chain resilience and sustainable manufacturing practices
- Cost Reduction in Manufacturing: The elimination of expensive transition metals requiring high loadings combined with simplified waste management protocols delivers substantial cost savings through reduced raw material expenditures and lower environmental compliance expenses The use of stable cobalt complexes minimizes catalyst replacement frequency while avoiding costly metal removal steps required by alternative methodologies This approach significantly reduces overall production costs without compromising product quality or yield consistency
- Enhanced Supply Chain Reliability: Utilization of readily available silane dehydrating agents and stable cobalt catalyst precursors ensures consistent raw material sourcing while minimizing supply chain vulnerabilities associated with specialized reagents requiring special handling or storage conditions The robust nature of this process maintains consistent performance across different production scales reducing batch failure risks that could disrupt supply continuity for critical pharmaceutical intermediates
- Scalability and Environmental Compliance: The mild reaction conditions enable seamless scale-up from laboratory development through commercial production without significant process re-engineering requirements while generating minimal hazardous waste streams This approach substantially reduces environmental impact compared to traditional methods requiring strong acids or generating large volumes of organic waste thereby simplifying regulatory compliance across global manufacturing sites
Frequently Asked Questions (FAQ)
The following questions address common technical concerns regarding implementation of this patented cobalt-catalyzed dehydration methodology based on detailed analysis of CN112961079A specifications These answers provide essential insights for technical evaluation teams assessing process viability for specific manufacturing applications requiring high-purity nitrile intermediates
Q: How does this cobalt-catalyzed method overcome harsh reaction conditions of traditional dehydration techniques?
A: The novel cobalt complex enables dehydration under mild conditions (60°C) without requiring strong acids like P₂O₅ or POCl₃ that cause decomposition issues in sensitive pharmaceutical intermediates
Q: What advantages does this method offer over existing transition metal-catalyzed approaches?
A: The tridentate nitrogen ligand cobalt complex provides exceptional stability with low catalyst loading while maintaining high selectivity across diverse substrates including heterocyclic compounds
Q: How does this process ensure consistent purity for pharmaceutical applications?
A: The mild reaction profile minimizes side reactions while precise control over silane stoichiometry prevents over-reduction byproducts commonly observed in alternative dehydration methods
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrile Supplier
Our company leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global pharmaceutical clients Our rigorous QC labs implement advanced analytical protocols ensuring consistent product quality across all production scales We specialize in transforming complex synthetic routes into reliable manufacturing processes through deep technical expertise in transition metal catalysis and specialized intermediate production This patented cobalt-catalyzed dehydration technology represents exactly the type of innovation our team excels at implementing for client-specific manufacturing needs
Contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your requirements They will provide a Customized Cost-Saving Analysis demonstrating how this technology can optimize your supply chain while meeting your exact quality specifications
