Advanced Synthesis Of Telmisartan Intermediates For Commercial Scale Manufacturing
Advanced Synthesis Of Telmisartan Intermediates For Commercial Scale Manufacturing
The global demand for angiotensin II receptor blockers (ARBs) continues to drive innovation in the synthesis of antihypertensive agents, with Telmisartan remaining a cornerstone therapy. A pivotal advancement in this domain is detailed in patent CN102050791B, which discloses a novel key intermediate and a robust synthetic pathway that addresses long-standing inefficiencies in traditional manufacturing. This patent introduces a streamlined method for producing 4'-[(4'-methyl-2'propyl[2,6'-bi-1H-benzimidazole]-1'-yl)methyl]-1,1'-biphenyl-2-carbonitrile, a critical precursor that enables the subsequent formation of Telmisartan with superior purity and yield. By shifting away from hazardous and costly reagents, this technology offers a compelling value proposition for pharmaceutical manufacturers seeking to optimize their supply chains and reduce environmental impact while maintaining stringent quality standards.
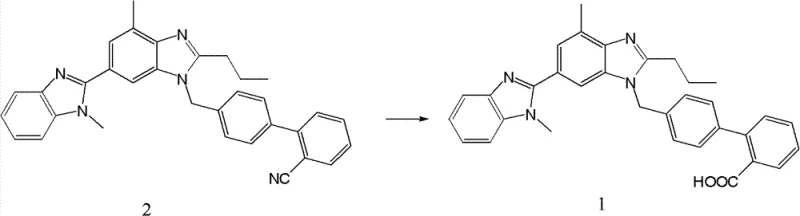
The strategic importance of this intermediate lies in its structural integrity and the efficiency of its assembly. Unlike previous iterations that struggled with isomeric contamination and difficult purification steps, this new route leverages a precise cyclization strategy that ensures the correct positioning of the benzimidazole rings. For R&D directors and process chemists, the ability to access this high-purity intermediate reliably translates directly into more consistent final API quality. Furthermore, the synthetic flexibility described in the patent allows for various activation methods and solvent systems, providing process engineers with the adaptability needed to integrate this chemistry into existing production facilities without extensive retrofitting.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Telmisartan has relied heavily on the condensation of bisimidazole intermediates, a process fraught with significant technical and economic challenges. Conventional routes, such as those disclosed in earlier patents, often necessitate the use of N-methyl o-phenylenediamine, a raw material that is not only expensive but also difficult to source in high purity on a large scale. The synthesis of the requisite bisimidazole scaffold typically demands extremely severe reaction conditions, including high temperatures and the use of strong acids or bases, which can lead to degradation of sensitive functional groups. Moreover, these traditional pathways are prone to generating isomeric byproducts, specifically the 4' isomer, which complicates downstream purification and drastically reduces overall yield. The reliance on such harsh conditions and problematic starting materials creates a bottleneck for manufacturers, inflating production costs and introducing variability that is unacceptable in modern GMP environments.
The Novel Approach
In stark contrast, the methodology outlined in CN102050791B presents a paradigm shift by utilizing o-phenylenediamine, a commodity chemical that is readily available and cost-effective. This novel approach circumvents the need for pre-formed N-methylated diamines by introducing the methyl group at a later stage or through a different mechanistic pathway that avoids early-stage alkylation issues. The core innovation involves the formation of an amide intermediate followed by an acid-catalyzed cyclization, a sequence that proceeds under significantly milder conditions compared to direct condensation methods. This strategic redesign of the synthetic tree not only simplifies the operational workflow but also inherently suppresses the formation of unwanted isomers. By decoupling the complexity of the biphenyl system from the harsh conditions of imidazole ring closure, this method achieves a level of process robustness that is ideal for continuous manufacturing and large-batch production.
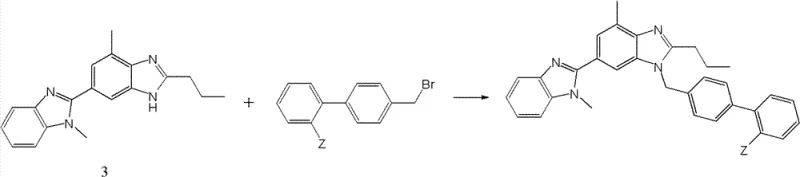
Mechanistic Insights into Acid-Catalyzed Benzimidazole Cyclization
The heart of this synthetic breakthrough is the efficient cyclization of the N-(2-aminophenyl) amide precursor into the fused bi-benzimidazole system. Mechanistically, this transformation is driven by the activation of the amide carbonyl or the adjacent nitrile group under acidic conditions, facilitating a nucleophilic attack by the ortho-amino group of the phenylenediamine moiety. The patent specifies the use of organic acids like glacial acetic acid or strong organic sulfonic acids such as p-toluenesulfonic acid to catalyze this ring closure. This acid-mediated pathway promotes the elimination of water and the subsequent aromatization of the imidazole ring, locking the molecular architecture into the desired thermodynamic product. The choice of solvent plays a critical role here; polar aprotic solvents or inert aromatic solvents are employed to maintain solubility at elevated temperatures (100°C to 150°C), ensuring that the reaction kinetics favor the formation of the target bi-cycle over potential oligomerization or decomposition pathways.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the generation of regioisomers. In traditional syntheses, the lack of directional control during ring closure often leads to a mixture of 2-substituted and 1-substituted benzimidazoles, requiring tedious chromatographic separations. However, the intramolecular nature of this cyclization, guided by the pre-organized amide linkage, imposes a geometric constraint that favors the formation of the specific 2,6'-bi-1H-benzimidazole configuration. This inherent selectivity reduces the burden on downstream purification units, allowing for simpler crystallization or extraction protocols to achieve the required purity levels. For quality assurance teams, this means a more predictable impurity profile and a reduced risk of genotoxic impurities often associated with harsh alkylation reagents used in alternative routes.
How to Synthesize Telmisartan Key Intermediate Efficiently
The practical execution of this synthesis involves a logical three-step sequence that balances reactivity with operational safety. Initially, the carboxylic acid precursor is activated using standard coupling reagents such as thionyl chloride or carbodiimides to form a reactive acyl species. This activated intermediate is then immediately reacted with o-phenylenediamine in a controlled temperature environment to generate the stable amide linkage. The final and most critical step involves heating this amide in the presence of an acid catalyst to induce cyclization, yielding the nitrile intermediate in high purity. Detailed standardized operating procedures for each stage, including specific molar ratios and work-up protocols, are essential for replicating the high yields reported in the patent examples.
- Activate 1-((2'-cyanobiphenyl-4-yl)methyl)-4-methyl-2-propyl-1H-benzimidazole-6-carboxylic acid using thionyl chloride or similar reagents.
- React the activated intermediate with o-phenylenediamine in a solvent like dichloromethane to form the amide precursor.
- Cyclize the amide precursor under acidic conditions (e.g., glacial acetic acid reflux) to form the final bi-benzimidazole nitrile intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The substitution of specialized, high-cost raw materials with commodity chemicals like o-phenylenediamine directly impacts the bill of materials, leading to substantial cost savings per kilogram of produced intermediate. Furthermore, the mild reaction conditions reduce the energy consumption associated with heating and cooling cycles, contributing to a lower carbon footprint and reduced utility costs. The robustness of the process also implies fewer batch failures and less waste generation, which translates into higher overall equipment effectiveness (OEE) and more reliable delivery schedules for downstream API manufacturers.
- Cost Reduction in Manufacturing: The elimination of expensive N-methyl o-phenylenediamine and the avoidance of complex purification steps for isomer removal significantly lower the direct production costs. By utilizing widely available reagents and solvents, manufacturers can leverage bulk purchasing power and reduce dependency on niche suppliers, thereby stabilizing the cost base against market volatility.
- Enhanced Supply Chain Reliability: The use of common chemical feedstocks ensures a steady supply of raw materials, mitigating the risk of shortages that often plague specialized intermediates. The simplified process flow, characterized by fewer unit operations and higher yields, shortens the overall production lead time, enabling faster response to market demand fluctuations and ensuring continuity of supply for critical hypertension medications.
- Scalability and Environmental Compliance: The process operates under relatively mild thermal conditions and avoids the use of heavy metal catalysts or highly toxic reagents, simplifying waste treatment and regulatory compliance. This environmental friendliness facilitates easier permitting for plant expansion and aligns with global sustainability goals, making the technology attractive for long-term investment and large-scale commercial deployment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this telmisartan intermediate synthesis. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential licensees and manufacturing partners.
Q: What are the primary advantages of this synthesis route over conventional methods?
A: This method avoids the use of expensive and difficult-to-produce N-methyl o-phenylenediamine, utilizing readily available o-phenylenediamine instead. It also eliminates the formation of isomeric impurities common in older routes, significantly simplifying purification.
Q: What are the typical reaction conditions for the cyclization step?
A: The cyclization is typically performed using organic acids like glacial acetic acid or p-toluenesulfonic acid at temperatures between 100°C and 150°C, ensuring high conversion rates without harsh conditions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents and reagents with mild reaction conditions and high yields (up to 98%), making it highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telmisartan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify every batch against the highest industry standards. Our facility is equipped to handle the specific solvent systems and thermal requirements of this advanced synthesis, guaranteeing a consistent supply of this key telmisartan intermediate.
We invite global pharmaceutical partners to collaborate with us to leverage this cost-effective and robust synthetic technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this innovative process can enhance your manufacturing efficiency and competitive advantage in the antihypertensive market.
