Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Manufacturing
Scalable Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Manufacturing
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for constructing robust molecular scaffolds found in active pharmaceutical ingredients. Patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in traditional heterocycle synthesis. This technology leverages a non-metallic iodine-promoted cyclization strategy that bypasses the need for harsh conditions or expensive transition metal catalysts. As illustrated in the structural diversity of bioactive molecules below, the 1,2,4-triazole core is ubiquitous in high-value drugs ranging from antifungal agents to enzyme inhibitors, making reliable access to these intermediates vital for the global supply chain.
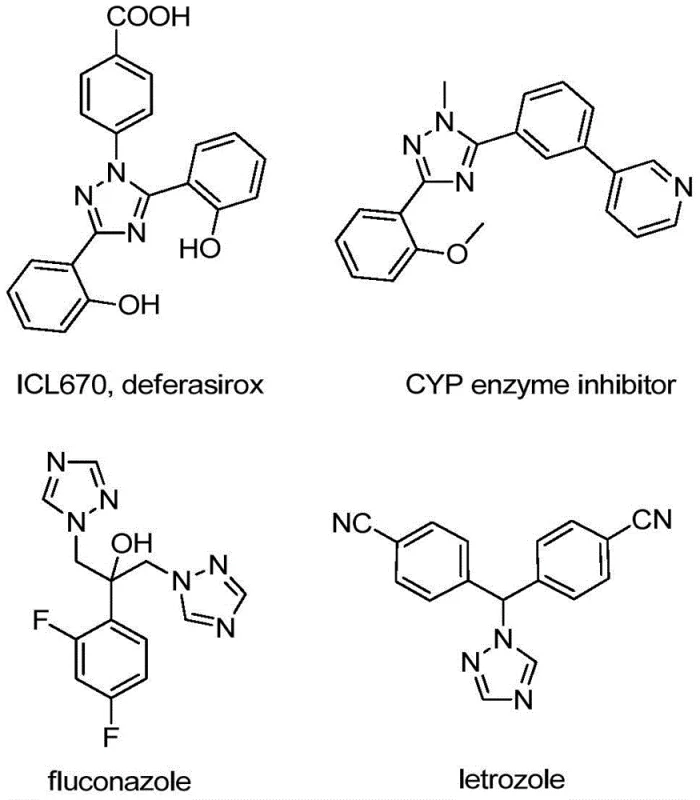
The introduction of a trifluoromethyl group into these heterocyclic systems is strategically significant, as it dramatically enhances the electronegativity, metabolic stability, and lipophilicity of the final drug candidates. For R&D directors seeking to optimize lead compounds, this patent offers a versatile platform where substituents at the 4 and 5 positions can be precisely tuned through substrate design. By utilizing cheap and readily available starting materials like hydrazones and trifluoroethylimidoyl chloride, this method lowers the barrier to entry for producing high-purity pharmaceutical intermediates. The operational simplicity, which eliminates the need for strict anhydrous or oxygen-free environments, further underscores its potential for widespread adoption in both laboratory discovery and commercial manufacturing settings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethylated nitrogen heterocycles has been plagued by significant synthetic challenges that hinder cost reduction in pharmaceutical intermediate manufacturing. Traditional approaches often rely on the post-synthetic trifluoromethylation of pre-formed heterocyclic rings, a process that frequently necessitates the use of specialized and costly trifluoromethylating reagents. Alternatively, methods employing synthons like trifluorodiazoethane pose severe safety hazards due to the explosive nature of diazo compounds, creating substantial risks for scale-up operations. Furthermore, many existing protocols demand rigorous exclusion of moisture and oxygen, requiring specialized equipment and inert gas lines that increase capital expenditure and operational complexity. These factors collectively contribute to longer lead times and higher production costs, limiting the accessibility of these valuable building blocks for broader drug discovery programs.
The Novel Approach
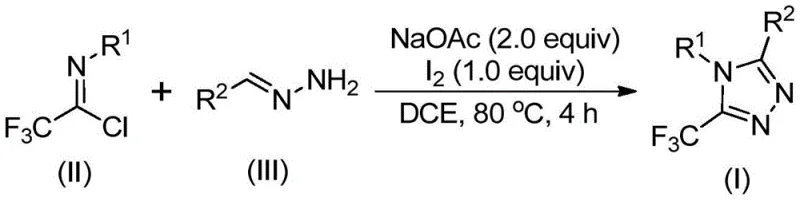
In stark contrast, the methodology outlined in CN110467579B represents a paradigm shift towards safer and more economical synthesis. By reacting trifluoroethylimidoyl chloride with hydrazones in the presence of sodium acetate and elemental iodine, the process achieves direct ring closure with high efficiency. This novel approach circumvents the instability issues associated with diazo reagents and avoids the use of toxic heavy metal catalysts often required in cross-coupling reactions. The reaction proceeds smoothly in common aprotic solvents like 1,2-dichloroethane (DCE) at moderate temperatures around 80°C. This robustness allows for a wider functional group tolerance, enabling the synthesis of diverse derivatives including those with halogen, alkyl, or alkoxy substitutions. For procurement managers, this translates to a supply chain less dependent on exotic reagents and more focused on commodity chemicals, thereby enhancing supply continuity and reducing raw material volatility.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is elegantly simple yet highly effective, relying on a sequence of base-promoted condensation followed by oxidative cyclization. Initially, sodium acetate acts as a mild base to facilitate the intermolecular carbon-nitrogen bond formation between the hydrazone and the trifluoroethylimidoyl chloride, generating a trifluoroacetamidine intermediate. This step is crucial as it sets the stage for the subsequent ring closure without requiring aggressive deprotonation conditions that might degrade sensitive functional groups. Following isomerization, the addition of elemental iodine triggers an oxidative iodination process, creating an electrophilic iodine species on the nitrogen framework. This activation is the key driver that enables the subsequent intramolecular electrophilic substitution.
The final stage involves an intramolecular nucleophilic attack and aromatization to yield the stable 5-trifluoromethyl-1,2,4-triazole core. This mechanism explains the broad substrate scope observed in the patent examples, where various aryl and heteroaryl groups are tolerated at both the N1 and C3 positions. From an impurity control perspective, the absence of transition metals means there is no risk of metal-catalyzed side reactions such as homocoupling or over-reduction, which often complicate purification. The use of iodine as a stoichiometric oxidant rather than a catalytic promoter in a complex cycle ensures that the reaction drives to completion with minimal byproduct formation. For quality control teams, this predictable reaction profile simplifies the identification and removal of impurities, ensuring that the final API intermediates meet stringent purity specifications required for regulatory submission.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for process chemists aiming to rapidly generate libraries of trifluoromethylated analogs. The protocol involves a straightforward one-pot procedure where reagents are mixed in a standard reactor, heated, and then treated with iodine in a second stage. This sequential addition allows for precise control over the reaction kinetics, preventing premature oxidation or decomposition of the intermediate amidine. Detailed standardized synthetic steps, including specific molar ratios and workup procedures, are provided in the technical guide below to ensure reproducibility across different laboratory scales.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane (DCE).
- Heat the reaction mixture to 80°C and stir for 2 to 4 hours to allow initial condensation.
- Add elemental iodine to the system and continue heating for an additional 1 to 2 hours to promote oxidative cyclization.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply chain resilience, this technology offers compelling advantages derived directly from its chemical design. The elimination of precious metal catalysts removes the need for expensive scavenging resins and extensive metal testing, which are significant cost drivers in GMP manufacturing. Additionally, the reliance on stable, shelf-stable starting materials like hydrazones and imidoyl chlorides mitigates the risk of supply disruptions caused by the short shelf-life of reactive reagents. The process operates under ambient atmospheric conditions, removing the energy and infrastructure costs associated with maintaining strict inert atmospheres. These factors combine to create a manufacturing route that is not only chemically efficient but also economically superior for long-term production.
- Cost Reduction in Manufacturing: The most significant economic benefit arises from the complete avoidance of heavy metal catalysts such as palladium or copper, which are notoriously expensive and difficult to remove to ppm levels. By utilizing elemental iodine and sodium acetate, both of which are commodity chemicals, the raw material cost profile is drastically improved. Furthermore, the simplified workup procedure involving filtration and standard column chromatography reduces solvent consumption and labor hours compared to complex extraction protocols required for metal-catalyzed reactions. This streamlined process flow directly contributes to substantial cost savings in the overall manufacturing budget without compromising yield or quality.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically the aldehydes and aromatic amines used to prepare the hydrazones and imidoyl chlorides, are widely available from global chemical suppliers. This abundance ensures that procurement teams can source multiple vendors for key inputs, reducing dependency on single-source suppliers. The stability of the trifluoroethylimidoyl chloride precursor also allows for bulk purchasing and storage, buffering the production schedule against market fluctuations. Consequently, the lead time for high-purity pharmaceutical intermediates can be significantly reduced, enabling faster response to market demands and clinical trial timelines.
- Scalability and Environmental Compliance: The patent explicitly demonstrates that the reaction can be expanded from milligram to gram scales with consistent performance, indicating strong potential for kilogram and ton-scale production. The use of dichloroethane (DCE) as a solvent, while requiring proper handling, is a well-established industrial solvent with established recovery and recycling protocols. Moreover, the absence of toxic heavy metals in the waste stream simplifies environmental compliance and wastewater treatment, aligning with increasingly stringent green chemistry regulations. This environmental compatibility facilitates smoother regulatory approvals and reduces the ecological footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived from the specific experimental data and beneficial effects reported in the patent documentation, providing clarity on safety, scalability, and application scope. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline projects.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the method described in patent CN110467579B utilizes elemental iodine as a promoter instead of toxic or expensive heavy metal catalysts, significantly simplifying downstream purification and reducing heavy metal residue risks.
Q: What are the advantages of using trifluoroethylimidoyl chloride over other synthons?
A: Trifluoroethylimidoyl chloride is relatively inexpensive and readily available compared to unstable reagents like trifluorodiazoethane. It allows for direct construction of the triazole ring under mild conditions without strict anhydrous or anaerobic requirements.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method can be easily expanded to the gram level and potentially larger scales. The use of common solvents like DCE and simple workup procedures supports commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of robust synthetic methodologies like the one described in CN110467579B for advancing drug discovery pipelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole intermediate meets the highest industry standards. We are committed to leveraging our technical expertise to optimize this iodine-promoted route for your specific needs, delivering high-quality intermediates that accelerate your time to market.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free protocol. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to unlock the full potential of trifluoromethylated heterocycles in your next generation of therapeutic agents.
