Advanced Manufacturing of 5-Trifluoromethyl-1,2,4-Triazole Derivatives for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles, particularly 1,2,4-triazole derivatives, which serve as critical scaffolds in numerous bioactive molecules. As highlighted in patent CN111978265B, a novel preparation method for 5-trifluoromethyl substituted 1,2,4-triazole derivatives has emerged, addressing long-standing challenges in efficiency and scalability. This technology is particularly relevant given the prevalence of the triazole motif in blockbuster drugs such as Maraviroc, Triazolam, Sitagliptin, and Deferasirox, where the introduction of a trifluoromethyl group can drastically enhance metabolic stability and lipophilicity. The disclosed method utilizes a simple yet powerful combination of sodium bicarbonate and ferric chloride to drive the cyclization, offering a streamlined alternative to legacy processes that often suffer from low atom economy.
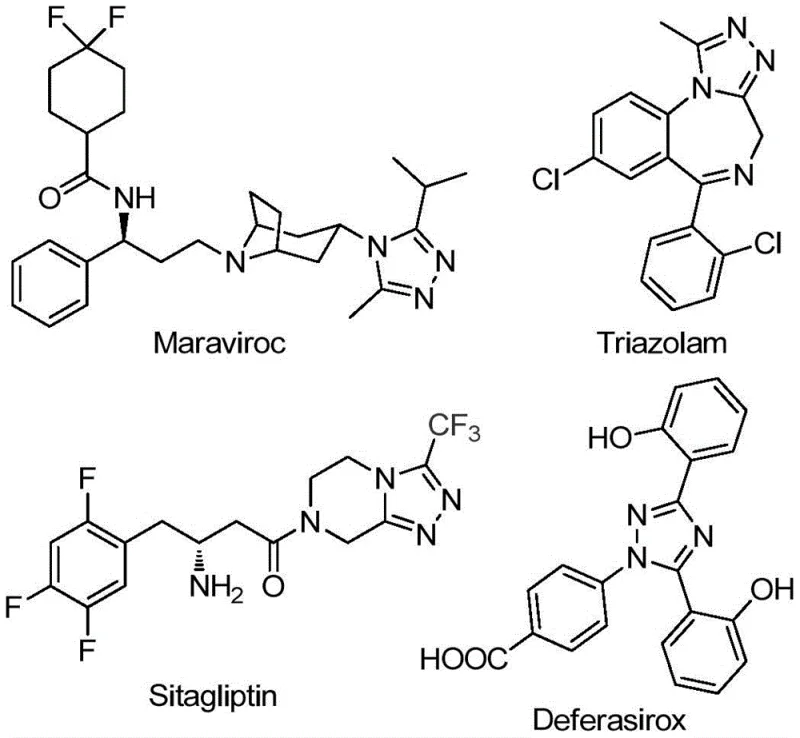
For R&D directors evaluating new pathways, the structural versatility demonstrated in this patent is compelling. The ability to tolerate various substituents on both the aryl and alkyl positions allows for the rapid generation of diverse libraries for structure-activity relationship (SAR) studies. Furthermore, the reliance on readily available starting materials like acyl chlorides and hydrazines ensures that the supply chain for these precursors remains stable and cost-effective. This innovation represents a significant step forward in the manufacturing of high-purity pharmaceutical intermediates, providing a reliable foundation for the development of next-generation therapeutics targeting a wide array of diseases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been plagued by significant operational hurdles that hinder industrial adoption. Traditional literature methods often rely on the condensation of 3,5-ditrifluoromethyl-1,3,4-oxadiazoles with primary amines or the cyclization of trifluoromethyl hydrazides with amidines. These pathways frequently demand严苛 reaction conditions, including extreme temperatures or the use of hazardous reagents that complicate waste management. Moreover, many of these classical approaches exhibit a narrow substrate scope, failing to accommodate alkyl hydrazones effectively, which limits the chemical space accessible to medicinal chemists. The cumulative effect of lengthy reaction steps, coupled with consistently low reaction yields, results in prohibitive costs and extended lead times for critical API intermediates.
The Novel Approach
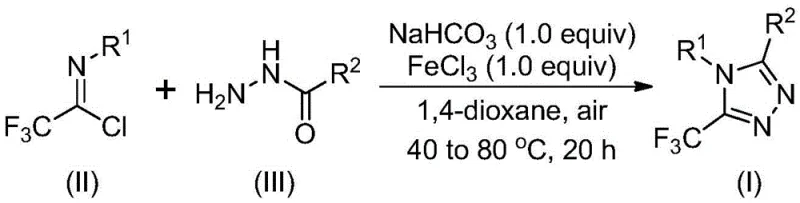
In stark contrast, the methodology described in CN111978265B introduces a paradigm shift by employing a ferric chloride-promoted tandem cyclization that is both operationally simple and highly efficient. This novel approach utilizes trifluoroethylimidoyl chloride and hydrazides as starting materials, reacting them in the presence of sodium bicarbonate and a catalytic amount of iron(III) chloride. The process eliminates the need for stringent anhydrous or oxygen-free environments, which are typically expensive to maintain on a large scale. By operating under mild thermal conditions and utilizing a common Lewis acid, this method achieves superior conversion rates and simplifies the downstream purification process. The reaction scheme below illustrates the elegant transformation from simple precursors to the complex triazole core.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing process parameters and ensuring consistent product quality. The reaction is believed to proceed through a sequential pathway initiated by base-promoted intermolecular carbon-nitrogen bond formation. In the first stage, sodium bicarbonate facilitates the nucleophilic attack of the hydrazide nitrogen onto the imidoyl chloride, generating a trifluoroacetamidine intermediate. This step is critical as it establishes the necessary connectivity for the subsequent ring closure without requiring aggressive bases that might degrade sensitive functional groups. The mild basicity of sodium bicarbonate ensures that the reaction environment remains controlled, minimizing side reactions such as hydrolysis of the acid chloride moiety.
Following the initial bond formation, the addition of ferric chloride triggers the second, decisive phase of the mechanism: a Lewis acid-promoted intramolecular dehydration condensation. The iron center coordinates with the nitrogen atoms, activating the intermediate towards cyclization and facilitating the elimination of water to form the aromatic 1,2,4-triazole ring. This dual-catalyst system (base followed by Lewis acid) is key to the method's success, as it decouples the bond formation and cyclization events, allowing each to proceed under its optimal conditions. This mechanistic clarity provides confidence in the reproducibility of the process, assuring procurement teams that the synthesis is robust against minor fluctuations in reaction parameters while maintaining high purity specifications.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Derivatives Efficiently
The practical execution of this synthesis is designed for ease of handling, making it accessible for both laboratory-scale optimization and pilot plant operations. The protocol involves a straightforward two-stage heating profile where reagents are first mixed at a lower temperature to form the intermediate, followed by the addition of the catalyst and elevation to a higher temperature to drive the cyclization to completion. This segmented approach maximizes yield while minimizing the formation of polymeric byproducts. For detailed operational parameters and specific stoichiometric ratios tailored to your specific derivative, please refer to the standardized synthesis guide below.
- Combine sodium bicarbonate, trifluoroethylimidoyl chloride, and hydrazide in an aprotic organic solvent such as 1,4-dioxane.
- Stir the mixture at a moderate temperature range of 30 to 50 degrees Celsius for 8 to 16 hours to facilitate initial bond formation.
- Introduce ferric chloride to the system and elevate the temperature to 70 to 90 degrees Celsius for 6 to 10 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial benefits that directly impact the bottom line and supply chain resilience. The shift away from exotic catalysts and harsh conditions translates into immediate cost savings and reduced operational complexity. For procurement managers, the availability of cheap, commodity-grade reagents like ferric chloride and sodium bicarbonate mitigates the risk of supply disruptions associated with specialized chemicals. Furthermore, the simplified workup procedure, which typically involves filtration and standard chromatography, reduces the consumption of solvents and silica gel, contributing to a more sustainable and cost-efficient manufacturing process.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the avoidance of cryogenic or ultra-high temperature requirements significantly lowers the overall cost of goods sold. By utilizing iron, one of the most abundant and affordable metals on earth, the process removes the financial burden associated with precious metal recovery and removal, which is a major cost driver in traditional pharmaceutical manufacturing. Additionally, the high yields reported across a broad range of substrates mean less raw material is wasted, further enhancing the economic viability of producing these complex heterocycles at scale.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures a stable and secure supply chain, reducing the risk of production delays due to raw material shortages. Since the reaction does not require strictly anhydrous conditions, the logistical challenges and costs associated with storing and transporting moisture-sensitive reagents are effectively eliminated. This robustness allows for greater flexibility in sourcing and inventory management, enabling manufacturers to respond more agilely to market demands and ensuring continuous availability of critical pharmaceutical intermediates for downstream API production.
- Scalability and Environmental Compliance: The simplicity of the reaction setup and the use of benign reagents make this process highly amenable to scale-up from gram to multi-ton quantities without significant re-engineering. The reduced generation of hazardous waste and the potential for solvent recycling align with increasingly stringent environmental regulations, lowering the compliance burden on manufacturing facilities. This green chemistry profile not only improves the corporate sustainability footprint but also future-proofs the supply chain against evolving regulatory landscapes regarding chemical safety and waste disposal in the fine chemical industry.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the process capabilities and limitations. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for industrial implementation. Understanding these nuances is essential for integrating this synthetic route into existing manufacturing workflows and maximizing its potential value.
Q: What are the primary advantages of this FeCl3-catalyzed method over conventional triazole synthesis?
A: Unlike traditional methods requiring harsh conditions or narrow substrate scopes, this protocol operates under mild, non-anhydrous conditions with broad functional group tolerance and high yields.
Q: Is the ferric chloride catalyst cost-effective for large-scale production?
A: Yes, ferric chloride is an inexpensive, commercially abundant Lewis acid that eliminates the need for precious metal catalysts, significantly reducing raw material costs.
Q: Can this synthesis route be scaled for industrial API intermediate manufacturing?
A: Absolutely. The process has been demonstrated to scale easily from gram-level laboratory synthesis to multi-kilogram production without loss of efficiency or purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global pharmaceutical landscape. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to adapt and optimize complex synthetic routes like the FeCl3-catalyzed triazole formation allows us to provide customized solutions that address the unique challenges of your drug development pipeline.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can accelerate your timeline and reduce your overall production costs. Let us be your partner in bringing innovative therapies to market faster and more efficiently.
