Advanced FeCl3-Catalyzed Synthesis of 5-Trifluoromethyl-1,2,4-Triazole Derivatives for Commercial Scale
Advanced FeCl3-Catalyzed Synthesis of 5-Trifluoromethyl-1,2,4-Triazole Derivatives for Commercial Scale
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which enhance metabolic stability and lipophilicity. Patent CN111978265B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole derivatives that addresses long-standing synthetic challenges. This technology leverages a ferric chloride-promoted cyclization strategy that operates under mild conditions, avoiding the stringent anhydrous and oxygen-free environments typically demanded by traditional heterocyclic chemistry. The significance of this advancement cannot be overstated, as 1,2,4-triazole scaffolds are ubiquitous in high-value bioactive molecules.
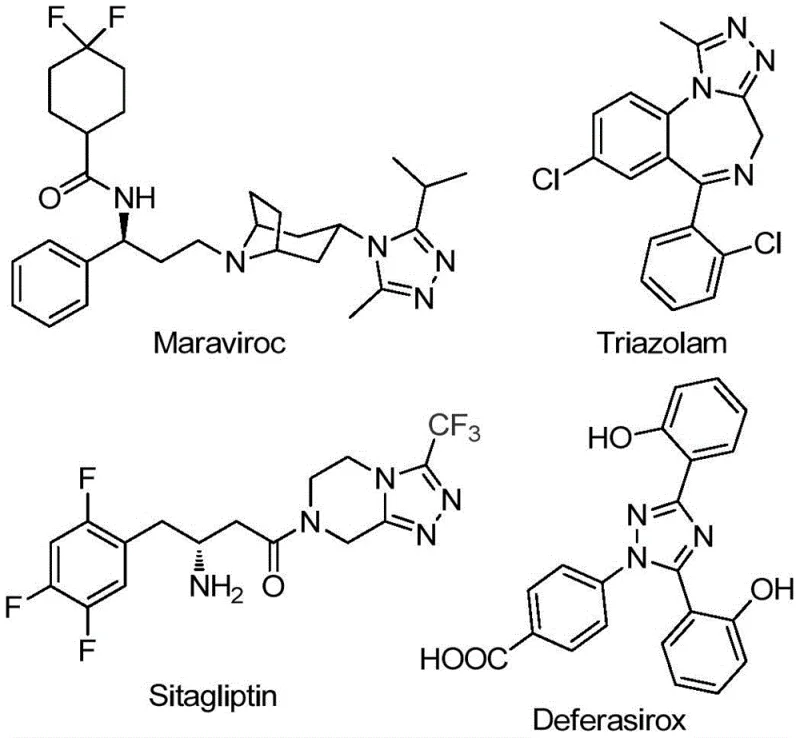
As illustrated in the structural diversity of marketed drugs like Maraviroc, Triazolam, Sitagliptin, and Deferasirox, the 1,2,4-triazole core is a critical pharmacophore. The introduction of the trifluoromethyl group further amplifies the electronic and steric properties of these molecules, making them indispensable in modern drug design. However, accessing these specific substituted derivatives has historically been fraught with difficulties regarding yield, substrate scope, and operational complexity. The disclosed invention provides a streamlined pathway that not only simplifies the synthetic workflow but also significantly expands the chemical space accessible to medicinal chemists and process engineers alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the literature described several traditional routes for synthesizing trifluoromethyl-substituted 1,2,4-triazoles, each plagued by significant drawbacks that hindered efficient manufacturing. Common strategies included the condensation of 3,5-ditrifluoromethyl-1,3,4-oxadiazoles with primary amines or the cyclization of trifluoromethyl hydrazides with amidines. These legacy methods often suffered from extremely harsh reaction conditions, requiring specialized equipment and rigorous exclusion of moisture and air. Furthermore, they were characterized by lengthy multi-step sequences that inevitably led to cumulative yield losses and increased waste generation.
A critical failure point in existing technologies was the narrow substrate scope, particularly regarding alkyl hydrazones. Previous tandem cyclization reactions developed by the inventors themselves, while efficient for certain substrates, completely failed to react with alkyl hydrazones. This limitation effectively blocked the synthesis of 3-alkyl fluoro-1,2,4-triazoles, a vital subclass of compounds for optimizing the physicochemical properties of drug candidates. The inability to access these structures forced researchers to rely on convoluted alternative pathways or abandon promising molecular designs entirely, creating a bottleneck in the development of new active pharmaceutical ingredients.
The Novel Approach
The novel approach detailed in the patent fundamentally shifts the paradigm by utilizing cheap and readily available acyl hydrazides and trifluoroethylimidoyl chlorides as starting materials. By employing ferric chloride as a promoter in conjunction with sodium bicarbonate, the reaction proceeds through a simplified mechanism that tolerates a wide array of functional groups. This method successfully overcomes the historical inability to utilize alkyl hydrazones, thereby unlocking the synthesis of 3-alkyl substituted derivatives that were previously inaccessible. The operational simplicity is a major advantage, as the reaction does not require inert atmosphere techniques, making it far more amenable to standard manufacturing facilities.
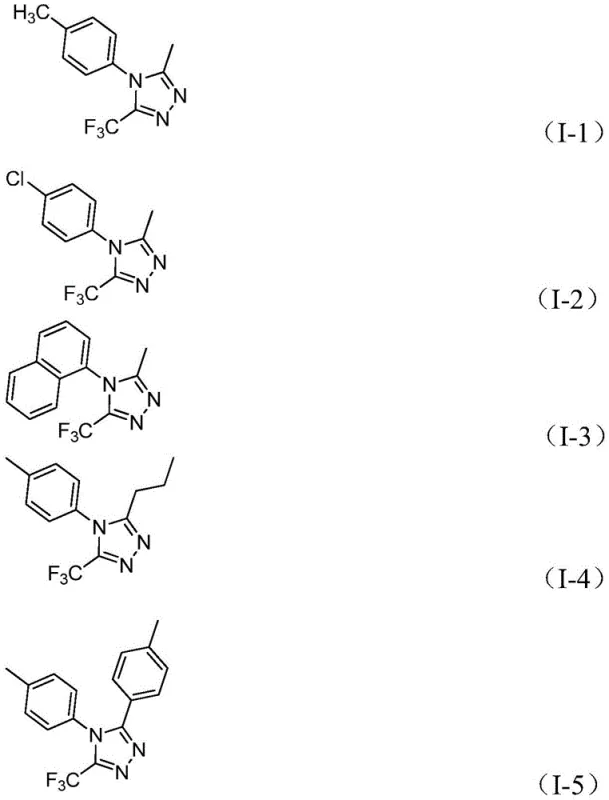
The versatility of this new methodology is evidenced by the successful synthesis of various derivatives, including those with methyl, methoxy, and halogen substituents on the aromatic rings. As shown in the specific examples provided, the system accommodates both electron-rich and electron-deficient substrates with high efficiency. This broad tolerance allows for the rapid generation of diverse libraries of triazole derivatives, accelerating the lead optimization phase in drug discovery. The ability to easily scale this process to the gram level and beyond provides a clear path from benchtop discovery to pilot plant production, ensuring continuity in the supply chain for critical intermediates.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation involves a sophisticated interplay between base-promoted nucleophilic attack and Lewis acid-catalyzed cyclization. Initially, the reaction likely undergoes a base-promoted intermolecular carbon-nitrogen bond formation between the hydrazide and the imidoyl chloride. This step generates a trifluoroacetamidine derivative intermediate, which serves as the precursor for the subsequent ring closure. The use of sodium bicarbonate is crucial here, acting as a mild base to neutralize the hydrochloric acid byproduct without degrading sensitive functional groups on the substrate.
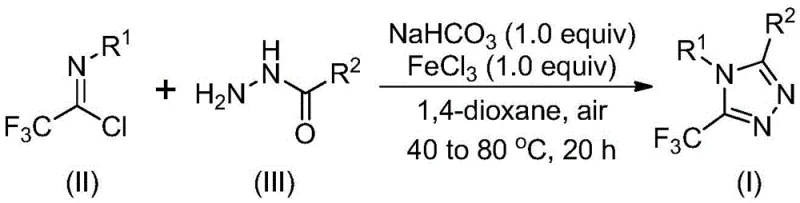
Following the initial bond formation, the addition of the metal Lewis acid, specifically ferric chloride, triggers the intramolecular dehydration condensation. The iron center coordinates with the nitrogen atoms, activating the intermediate towards cyclization and facilitating the elimination of water to form the stable 1,2,4-triazole ring. This two-stage temperature protocol, starting at a lower temperature for the coupling and increasing for the cyclization, optimizes the reaction kinetics and minimizes side reactions. From an impurity control perspective, the mildness of the reagents and the specificity of the Lewis acid promotion result in a cleaner crude reaction profile, reducing the burden on downstream purification processes.
Furthermore, the choice of solvent plays a pivotal role in the reaction efficiency. While various organic solvents can dissolve the reactants, aprotic solvents like 1,4-dioxane are preferred as they effectively promote the reaction progress without interfering with the Lewis acid catalyst. The solubility of all components in 1,4-dioxane ensures a homogeneous reaction mixture, which is essential for consistent heat transfer and reaction rates during scale-up. This mechanistic understanding allows process chemists to fine-tune parameters such as stoichiometry and temperature ramps to maximize yield and purity for specific target molecules.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Derivatives Efficiently
The synthesis protocol outlined in the patent offers a straightforward procedure that balances reaction efficiency with operational safety. The process begins with the careful weighing and mixing of sodium bicarbonate, trifluoroethylimidoyl chloride, and the chosen hydrazide in an appropriate volume of organic solvent. Maintaining the correct molar ratios is essential; typically, an excess of hydrazide is used relative to the imidoyl chloride to drive the reaction to completion. The mixture is stirred at a controlled temperature to allow the initial coupling to proceed before the catalyst is introduced for the final cyclization step.
- Mix sodium bicarbonate, trifluoroethylimidoyl chloride, and hydrazide in an aprotic organic solvent such as 1,4-dioxane.
- Stir the mixture at a moderate temperature range of 30 to 50 degrees Celsius for 8 to 16 hours to facilitate initial bond formation.
- Add ferric chloride catalyst and increase temperature to 70 to 90 degrees Celsius for 6 to 10 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route presents compelling economic and logistical benefits. The primary driver for cost reduction lies in the selection of raw materials; both the acyl chlorides used to prepare hydrazides and the ferric chloride catalyst are commodity chemicals available in bulk quantities at low prices. Unlike methods requiring precious metal catalysts or exotic reagents, this process relies on earth-abundant iron, which drastically lowers the material cost per kilogram of the final product. Additionally, the elimination of expensive drying agents and inert gas protection systems reduces utility costs and capital expenditure on specialized reactor infrastructure.
Supply chain reliability is significantly enhanced by the robustness of the reaction conditions. Since the process does not require strictly anhydrous or oxygen-free environments, it is less susceptible to disruptions caused by variations in raw material moisture content or minor equipment leaks. This tolerance translates to higher batch success rates and reduced downtime for reactor cleaning and preparation. The ability to source starting materials from multiple global suppliers without stringent specification requirements further mitigates the risk of supply shortages, ensuring a steady flow of intermediates for downstream API manufacturing.
Scalability and environmental compliance are also key advantages of this technology. The reaction can be easily scaled from laboratory glassware to industrial reactors without encountering the heat transfer or mixing issues often associated with exothermic cyclizations. The use of ferric chloride, a relatively benign metal salt, simplifies waste treatment compared to processes generating heavy metal contamination. This aligns with increasingly strict environmental regulations and corporate sustainability goals, potentially reducing the costs associated with hazardous waste disposal and environmental permitting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this method for industrial production.
Q: What are the primary advantages of the FeCl3-catalyzed method over traditional triazole synthesis?
A: The FeCl3-catalyzed method described in patent CN111978265B eliminates the need for harsh anhydrous or oxygen-free conditions required by conventional routes. It utilizes cheap, commercially available starting materials and offers a broader substrate scope, successfully synthesizing 3-alkyl substituted derivatives that previous methods could not access.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is highly scalable. The use of inexpensive reagents like sodium bicarbonate and ferric chloride, combined with simple post-processing steps like filtration and column chromatography, makes it ideal for commercial scale-up from gram to multi-kilogram levels without complex equipment.
Q: What types of substituents are tolerated in this triazole synthesis?
A: The method demonstrates excellent functional group tolerance. It accommodates various substituents on the aryl rings, including methyl, methoxy, halogens, and trifluoromethyl groups at ortho, meta, or para positions, allowing for the design of diverse pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the timely delivery of high-quality pharmaceutical intermediates. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to full-scale manufacturing is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 5-trifluoromethyl-1,2,4-triazole derivatives meets the exacting standards required by global regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how this advanced FeCl3-catalyzed route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your production volumes. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most reliable and cost-effective chemical solution available.
