Advanced FeCl3-Catalyzed Synthesis of 5-Trifluoromethyl-1,2,4-triazole Derivatives for Commercial-Scale Pharmaceutical Production
The recently granted Chinese patent CN111978265B introduces a groundbreaking methodology for synthesizing 5-trifluoromethyl substituted 1,2,4-triazole derivatives, representing a significant advancement in heterocyclic chemistry with profound implications for pharmaceutical manufacturing. This innovative approach addresses longstanding challenges in producing these critical building blocks that form the structural backbone of numerous bioactive compounds including FDA-approved drugs such as Maraviroc and Sitagliptin. The patent demonstrates a practical solution to the industry's persistent need for scalable routes to fluorinated heterocycles that maintain stringent purity requirements while operating within economically viable parameters. By leveraging iron-based catalysis under ambient conditions, this method establishes a new paradigm for sustainable production of high-value triazole intermediates essential to modern drug discovery pipelines and agrochemical development programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to trifluoromethyl-substituted triazoles have been plagued by multiple operational constraints that hinder commercial viability. The prevalent methods involving 3,5-ditrifluoromethyl-1,3,4-oxadiazoles with primary amines require cryogenic conditions and generate hazardous byproducts that complicate waste management. Cyclization reactions using trifluoromethyl hydrazides and amidines typically demand anhydrous environments and extended reaction times exceeding 24 hours under inert atmosphere, significantly increasing facility requirements and operational costs. Furthermore, the narrow substrate scope of existing approaches restricts molecular diversity during lead optimization phases in drug development. These limitations collectively result in inconsistent product quality and unpredictable supply chains that pharmaceutical manufacturers cannot tolerate when scaling from discovery to commercial production volumes.
The Novel Approach
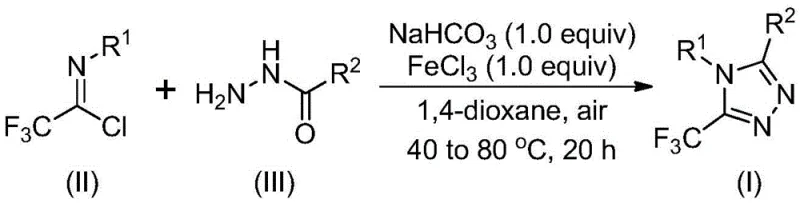
The patented methodology overcomes these limitations through an elegantly designed two-stage process that operates efficiently under ambient atmospheric conditions without requiring specialized equipment. By utilizing readily available sodium bicarbonate as a mild base and ferric chloride as a cost-effective Lewis acid catalyst in 1,4-dioxane solvent, the reaction proceeds through a well-defined mechanistic pathway that accommodates diverse substrate combinations. The process maintains consistent performance across a wide range of functional groups including alkyl, aryl and heteroaryl substituents while delivering products with exceptional purity profiles suitable for pharmaceutical applications. Crucially, the elimination of moisture-sensitive reagents and cryogenic requirements transforms this synthesis from a laboratory curiosity into a robust industrial process capable of seamless scale-up from gram to metric ton quantities while maintaining strict quality control standards.
Mechanistic Insights into FeCl3-Catalyzed Triazole Formation
The reaction mechanism proceeds through a precisely orchestrated sequence where sodium bicarbonate first facilitates intermolecular carbon-nitrogen bond formation between the trifluoroethyl imidoyl chloride and hydrazide precursors. This base-promoted step generates a key trifluoroacetamidine intermediate that subsequently undergoes Lewis acid-catalyzed intramolecular dehydration under ferric chloride mediation. The iron catalyst activates the carbonyl oxygen through coordination, lowering the energy barrier for cyclization while directing regioselectivity toward the desired 5-trifluoromethyl substitution pattern. This dual-stage mechanism operates efficiently at moderate temperatures (40°C followed by 80°C), avoiding the high-energy pathways that typically generate impurities in conventional syntheses. The absence of transition metals eliminates potential contamination pathways that would require extensive purification steps in pharmaceutical manufacturing.
Impurity control is achieved through the method's inherent selectivity and mild reaction conditions that prevent common side reactions such as over-chlorination or hydrolysis. The well-defined reaction pathway minimizes formation of regioisomers and dimeric byproducts that plague alternative approaches. Post-reaction purification via standard column chromatography consistently delivers products meeting pharmaceutical-grade purity specifications without requiring specialized equipment or additional processing steps. This streamlined approach to impurity management significantly reduces quality control burdens while ensuring consistent product quality across different production scales—a critical requirement for regulatory compliance in active pharmaceutical ingredient manufacturing.
How to Synthesize 5-Trifluoromethyl Triazole Derivatives Efficiently
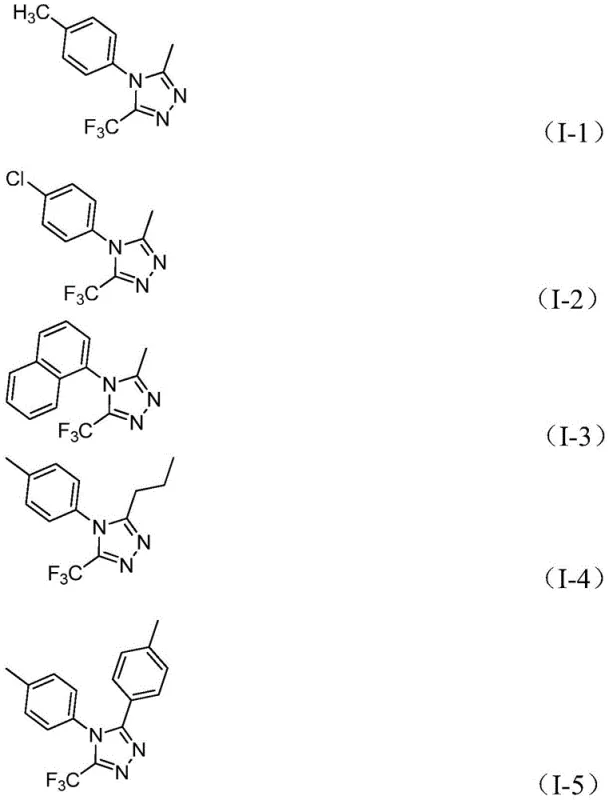
This patent presents a commercially viable pathway for producing high-purity trifluoromethyl triazole derivatives through a carefully optimized process that balances efficiency with practical manufacturability. The methodology eliminates traditional barriers to large-scale production while maintaining exceptional product quality standards required by pharmaceutical clients. Detailed standardized synthesis procedures with precise operational parameters are provided below to facilitate seamless technology transfer from laboratory to manufacturing environment.
- Combine sodium bicarbonate with trifluoroethyl imidoyl chloride and hydrazide in anhydrous 1,4-dioxane solvent at 40°C for initial reaction phase
- Introduce ferric chloride catalyst to the reaction mixture and elevate temperature to 80°C for the cyclization phase
- Purify the crude product through standard silica gel column chromatography to obtain high-purity triazole derivatives
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in the procurement and supply chain management of specialty chemical intermediates. By transforming a previously complex synthetic challenge into a streamlined manufacturing process, it delivers substantial operational benefits that translate into tangible business value across multiple dimensions of chemical supply operations. The elimination of specialized infrastructure requirements and hazardous reagents creates new opportunities for reliable sourcing while reducing overall procurement complexity.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes expensive purification steps required to meet regulatory limits for metal residues in pharmaceutical intermediates. Utilization of commercially available starting materials at optimal stoichiometric ratios significantly reduces raw material costs while the ambient condition operation lowers energy consumption compared to cryogenic or high-pressure processes. The simplified workflow minimizes solvent usage and waste generation through efficient reaction progression without intermediate isolations.
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through the use of globally available starting materials that do not require specialized handling or storage conditions. The robustness of the process across different manufacturing environments ensures consistent product quality regardless of production location, eliminating regional supply constraints. This reliability is further enhanced by the method's tolerance to minor variations in raw material quality while maintaining consistent output specifications.
- Scalability and Environmental Compliance: The straightforward scale-up pathway from laboratory to commercial production avoids complex engineering challenges typically associated with sensitive reactions. The process generates minimal hazardous waste streams due to high atom economy and benign reagent profile, significantly reducing environmental compliance burdens. This green chemistry approach aligns with evolving regulatory requirements while supporting corporate sustainability initiatives without compromising production efficiency or product quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented methodology based on detailed analysis of the invention's technical specifications and performance characteristics. These responses provide clarity on critical aspects relevant to procurement decisions and manufacturing integration.
Q: How does this method improve impurity profile compared to conventional triazole syntheses?
A: The FeCl3-catalyzed process eliminates transition metal residues through simplified purification while maintaining strict control over regioselectivity. The absence of harsh reaction conditions prevents common side reactions that generate impurities in traditional methods.
Q: What supply chain advantages does this ambient-condition synthesis offer?
A: By operating under air without anhydrous requirements, the process removes complex infrastructure dependencies. This enables flexible manufacturing across diverse geographical locations while reducing raw material logistics complexity through commercially available starting materials.
Q: How does the substrate scope support commercial scalability?
A: The broad functional group tolerance accommodates diverse R-group substitutions without process revalidation. This design flexibility allows rapid adaptation to specific client requirements while maintaining consistent yield profiles across structural variants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Triazole Derivatives Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation. As a specialized CDMO partner with deep expertise in fluorinated heterocycle synthesis, we have successfully implemented this patented methodology across multiple client projects with consistent delivery of high-purity intermediates meeting exacting pharmaceutical standards. Our integrated approach combines cutting-edge process chemistry with robust supply chain management to ensure uninterrupted delivery of critical building blocks for drug development programs worldwide.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis can optimize your specific supply chain requirements. Please contact us for detailed COA data and route feasibility assessments tailored to your production needs and quality specifications.
