Advanced NHC-Catalyzed Synthesis of Trifluoromethyl Homoisoflavones for Commercial Scale-Up
Advanced NHC-Catalyzed Synthesis of Trifluoromethyl Homoisoflavones for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust and scalable synthetic routes for bioactive scaffolds, particularly those exhibiting potent antitumor properties such as homoisoflavonoids. A groundbreaking approach detailed in patent CN108727323B introduces a highly efficient method for synthesizing trifluoromethyl-substituted homoisoflavone compounds using nitrogen heterocyclic carbene (NHC) organocatalysis. This innovation represents a significant paradigm shift from traditional transition-metal-dependent methodologies, offering a greener and more economically viable pathway for producing these critical pharmaceutical intermediates. The process leverages readily available trifluoromethyl-substituted olefine aldehyde compounds as starting materials, subjecting them to a mild NHC-catalyzed cyclization followed by a strategic oxidative aromatization using elemental iodine. This dual-step strategy not only ensures high reaction efficiency and excellent selectivity but also aligns perfectly with modern green chemistry principles by minimizing hazardous waste and energy consumption.

For procurement specialists and supply chain managers, the implications of this technology are profound. The reliance on stable, non-precious metal catalysts and common oxidants like iodine drastically reduces the raw material cost volatility often associated with palladium or rhodium-based systems. Furthermore, the broad substrate scope demonstrated in the patent suggests that this platform technology can be adapted to generate a diverse library of analogues, providing a reliable homoisoflavone supplier with the flexibility to meet varying drug discovery needs without retooling entire production lines. The ability to operate the initial cyclization at room temperature further underscores the energy efficiency of the process, contributing to substantial cost reduction in pharmaceutical intermediate manufacturing while maintaining a safety profile suitable for large-scale operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of homoisoflavonoid compounds has been plagued by significant technical and economic hurdles that limit their widespread application in mass production. Traditional representative schemes often rely on bio-enzyme catalyzed coupling reactions between chromones and halogenated hydrocarbons, or Lewis acid-catalyzed dehydration couplings involving chromone-derived alcohols and terminal alkynes. These legacy methods suffer from severe limitations, including the absolute necessity for extensive pre-functionalization of starting materials, which adds multiple synthetic steps and increases overall material costs. Moreover, the requirement for expensive and often toxic Lewis acid catalysts creates substantial challenges in downstream processing, particularly regarding the removal of trace metal residues to meet stringent regulatory standards for active pharmaceutical ingredients. The harsh reaction conditions frequently employed in these conventional routes often lead to poor atom economy and limited substrate latitude, restricting the chemical diversity accessible to medicinal chemists and complicating the scale-up process due to safety concerns associated with exothermic or high-pressure reactions.
The Novel Approach
In stark contrast, the novel NHC-catalyzed methodology presented in the patent data offers a streamlined and elegant solution to these longstanding synthetic challenges. By utilizing nitrogen heterocyclic carbenes as organocatalysts, the new route bypasses the need for transition metals entirely, thereby eliminating the risk of heavy metal contamination and the costly purification steps required to remove them. The reaction proceeds under remarkably mild conditions, with the initial cyclization occurring at room temperature, which significantly lowers the energy footprint of the manufacturing process. The subsequent oxidative step utilizes elemental iodine and concentrated sulfuric acid at moderate temperatures ranging from 50 to 100 degrees Celsius, conditions that are easily manageable in standard glass-lined reactors found in most fine chemical facilities. This approach not only simplifies the operational workflow but also enhances the overall yield and selectivity, making it an ideal candidate for the cost reduction in electronic chemical manufacturing and pharmaceutical sectors alike where purity and efficiency are paramount.
Mechanistic Insights into NHC-Catalyzed Cyclization and Oxidation
The core of this innovative synthesis lies in the unique reactivity of the N-heterocyclic carbene (NHC) catalyst, which facilitates the formation of the chromone backbone through a sophisticated organocatalytic cycle. The mechanism likely involves the nucleophilic attack of the NHC on the aldehyde moiety of the trifluoromethyl-substituted olefine aldehyde substrate, generating a Breslow intermediate or a related zwitterionic species. This activated intermediate then undergoes an intramolecular conjugate addition or cyclization with the proximal ether oxygen or alkene functionality, effectively constructing the heterocyclic ring system under mild basic conditions. The use of bases such as potassium carbonate, sodium carbonate, or triethylamine is critical in this stage to deprotonate the catalyst precursor and maintain the catalytic cycle without promoting unwanted side reactions. This organocatalytic step is highly advantageous for R&D directors focused on impurity control, as the mild nature of the reaction prevents the thermal decomposition of sensitive functional groups often present in complex drug candidates.
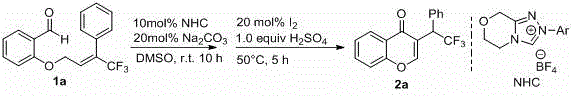
Following the formation of the intermediate flavonoid structure, the process employs a distinct oxidative aromatization strategy using elemental iodine as the oxidant. In the presence of concentrated sulfuric acid, the iodine facilitates the removal of hydrogen atoms to establish the fully aromatic pyrone ring characteristic of the homoisoflavone skeleton. This oxidative step is conducted at elevated temperatures between 50 and 100 degrees Celsius, ensuring complete conversion while maintaining control over the reaction kinetics. The choice of iodine is particularly strategic from a commercial perspective; it is inexpensive, readily available, and generates byproducts that are relatively easy to separate from the organic product phase during workup. The combination of NHC catalysis and iodine oxidation results in a highly atom-economical process that maximizes the incorporation of starting materials into the final product, thereby reducing waste generation and enhancing the overall sustainability profile of the manufacturing campaign.
How to Synthesize Trifluoromethyl Homoisoflavones Efficiently
The practical execution of this synthesis involves a straightforward two-stage protocol that can be readily implemented in standard laboratory or pilot plant settings. The process begins with the preparation of the key trifluoromethyl-substituted olefine aldehyde substrate, which is synthesized via the alkylation of salicylaldehyde derivatives with appropriate bromo-trifluoromethyl-butene precursors in the presence of a base like potassium carbonate in DMF. Once the substrate is secured, the NHC-catalyzed cyclization is performed by mixing the aldehyde with a catalytic amount of NHC salt and a stoichiometric base in a polar aprotic solvent such as DMSO or DMF at room temperature for several hours. The detailed standardized synthesis steps, including precise molar ratios, solvent volumes, and workup procedures, are outlined in the guide below to ensure reproducibility and high yield.
- Prepare the trifluoromethyl-substituted olefine aldehyde substrate by reacting salicylaldehyde derivatives with trifluoromethyl-containing alkyl halides in the presence of a base like potassium carbonate.
- Perform the NHC-catalyzed cyclization at room temperature using an organic solvent and a base to form the intermediate flavonoid structure.
- Execute the oxidative aromatization step by adding elemental iodine and concentrated sulfuric acid at elevated temperatures (50-100°C) to yield the final trifluoromethyl homoisoflavone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NHC-catalyzed route offers compelling economic and logistical benefits that directly impact the bottom line. The elimination of precious metal catalysts removes a major source of cost volatility and supply risk, as the prices of metals like palladium can fluctuate wildly based on geopolitical factors. Instead, the process relies on abundant and cheap reagents such as iodine and simple organic bases, which ensures a stable and predictable cost structure for long-term production contracts. Furthermore, the simplified purification process, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and shortens the overall production cycle time, leading to significant operational efficiencies.
- Cost Reduction in Manufacturing: The transition from transition-metal catalysis to organocatalysis fundamentally alters the cost equation by removing the need for expensive ligands and metal salts. This shift not only lowers the direct material costs but also reduces the indirect costs associated with waste disposal and environmental compliance, as the process generates less hazardous heavy metal waste. The high atom economy of the reaction means that a greater proportion of the raw materials end up in the final product, minimizing waste and maximizing resource utilization, which translates to substantial cost savings in high-volume manufacturing scenarios.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like iodine, sulfuric acid, and common solvents ensures that the supply chain is robust and resilient to disruptions. Unlike specialized catalysts that may have single-source suppliers and long lead times, the reagents for this process are widely available from multiple global vendors. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for downstream pharmaceutical customers who depend on timely access to these critical intermediates.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of standard equipment make this process highly scalable from gram to multi-ton quantities without the need for specialized high-pressure or cryogenic infrastructure. The green chemistry credentials of the method, characterized by lower energy consumption and reduced toxic waste generation, align perfectly with increasingly stringent environmental regulations. This compliance advantage reduces the regulatory burden on manufacturing sites and facilitates faster approval for new production lines, enabling rapid response to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic methodology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering a transparent view of the process capabilities and limitations for potential partners and stakeholders.
Q: What are the primary advantages of using NHC catalysis over traditional Lewis acid methods for homoisoflavone synthesis?
A: The NHC-catalyzed method described in patent CN108727323B operates under significantly milder conditions compared to traditional Lewis acid or enzymatic methods. It eliminates the need for expensive transition metal catalysts and harsh pre-functionalization steps, resulting in higher atom economy and simplified downstream purification processes.
Q: How does this synthetic route impact the purity profile of the final API intermediate?
A: By utilizing a highly selective organocatalytic cycle followed by a controlled iodine oxidation, the process minimizes the formation of complex metal-based impurities. The mild reaction temperatures reduce thermal degradation byproducts, allowing for the achievement of stringent purity specifications through standard recrystallization or chromatography.
Q: Is this methodology suitable for large-scale industrial production?
A: Yes, the protocol is designed for scalability. The use of inexpensive reagents like elemental iodine and common organic solvents, combined with the avoidance of sensitive air-free techniques typically required for some organometallic catalysts, makes this route highly viable for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Homoisoflavone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this NHC-catalyzed technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory protocol into a robust commercial manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity and potency at every stage of production.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free approach can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply chain for your trifluoromethyl homoisoflavone requirements.
