Scalable NHC-Catalyzed Synthesis of Trifluoromethyl Homoisoflavones for Pharmaceutical Applications
Scalable NHC-Catalyzed Synthesis of Trifluoromethyl Homoisoflavones for Pharmaceutical Applications
The rapidly evolving landscape of oncology drug discovery demands robust and scalable synthetic routes for complex heterocyclic scaffolds, particularly those incorporating fluorine motifs which often enhance metabolic stability and bioavailability. In this context, the Chinese patent CN108727323B presents a groundbreaking methodology for the synthesis of trifluoromethyl-substituted homoisoflavone compounds, a class of molecules renowned for their potent antitumor activities. This proprietary technology leverages N-Heterocyclic Carbene (NHC) organocatalysis to achieve a green, efficient, and highly selective transformation that addresses the critical bottlenecks of traditional synthesis. By utilizing readily available trifluoromethyl-substituted alkenal compounds as starting materials, the process circumvents the need for pre-functionalized, expensive precursors, thereby establishing a new benchmark for cost-effective manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the homoisoflavonoid core has relied heavily on methodologies that impose significant operational and economic burdens on large-scale production. Representative conventional schemes often involve bio-enzyme catalyzed coupling reactions between chromones and halogenated hydrocarbons, or Lewis acid-catalyzed dehydration couplings of chromone-derived alcohols with terminal alkynes. These legacy approaches are frequently plagued by the necessity for harsh reaction conditions, such as elevated temperatures and strongly acidic or basic environments, which can degrade sensitive functional groups. Furthermore, the reliance on stoichiometric amounts of expensive Lewis acids or specialized enzymes drastically inflates the raw material costs, while the limited substrate scope restricts the chemical diversity accessible to medicinal chemists. The low atom economy and complicated workup procedures associated with these methods often result in substantial waste generation, conflicting with modern green chemistry principles and increasing the environmental compliance costs for manufacturers.
The Novel Approach
In stark contrast, the innovative route disclosed in patent CN108727323B utilizes a dual-catalytic strategy that merges NHC organocatalysis with iodine-mediated oxidation to streamline the synthesis. This method initiates with a mild, room-temperature cyclization catalyzed by nitrogen heterocyclic carbenes, which activates the aldehyde functionality without requiring toxic heavy metals. The subsequent oxidative aromatization step employs elemental iodine and concentrated sulfuric acid at moderate temperatures (50-100°C), ensuring high conversion rates and excellent selectivity. This approach not only broadens the substrate applicability to include various electron-donating and electron-withdrawing groups but also simplifies the operational workflow. The ability to perform the initial cyclization at ambient temperature significantly reduces energy consumption, while the use of inexpensive, commodity-grade oxidants like iodine replaces costly transition metal catalysts, fundamentally altering the cost structure of homoisoflavone manufacturing.

Mechanistic Insights into NHC-Catalyzed Cyclization and Oxidation
The core of this technological advancement lies in the unique reactivity of the N-Heterocyclic Carbene (NHC) catalyst, which acts as a nucleophilic activator for the aldehyde group in the trifluoromethyl-substituted substrate. Upon deprotonation of the NHC precursor salt by a mild base such as sodium carbonate or triethylamine, the free carbene species attacks the carbonyl carbon, forming a zwitterionic tetrahedral intermediate known as the Breslow intermediate. This key species undergoes an intramolecular conjugate addition (Michael-type addition) to the electron-deficient alkene moiety bearing the trifluoromethyl group, effectively closing the pyran ring to form the dihydroflavone skeleton. The presence of the strong electron-withdrawing trifluoromethyl group enhances the electrophilicity of the alkene, facilitating this cyclization step under remarkably mild conditions. This mechanistic pathway avoids the high-energy transition states typical of thermal cyclizations, thereby minimizing the formation of polymeric byproducts and ensuring a clean reaction profile that is crucial for downstream purification.
Following the NHC-mediated ring closure, the intermediate flavonoid requires oxidative aromatization to yield the final homoisoflavone product. This is achieved through the introduction of elemental iodine and concentrated sulfuric acid, which function synergistically to remove the extra hydrogen atoms and establish the fully conjugated ketone system. The iodine acts as a mild yet effective oxidant, while the sulfuric acid likely protonates the oxygen species to facilitate dehydration and rearomatization. This second step is conducted at elevated temperatures ranging from 50°C to 100°C, which provides the necessary thermal energy to drive the oxidation to completion without decomposing the sensitive trifluoromethyl motif. The combination of these two distinct catalytic cycles ensures high atom economy and minimizes the generation of hazardous waste, aligning perfectly with the sustainability goals of modern pharmaceutical supply chains.
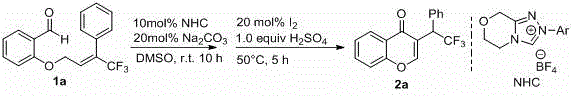
How to Synthesize Trifluoromethyl Homoisoflavones Efficiently
To implement this synthesis effectively, the process begins with the preparation of the critical trifluoromethyl-substituted olefine aldehyde substrate, which serves as the foundational building block for the entire sequence. As illustrated in the patent examples, this precursor is typically synthesized via an etherification reaction between a substituted salicylaldehyde and a trifluoroalkenyl bromide in the presence of a base like potassium carbonate in DMF. Once the substrate is secured, the NHC catalytic cycle is initiated by mixing the aldehyde with a catalytic amount of NHC precursor (10-20 mol%) and a stoichiometric base in a polar aprotic solvent such as DMSO or DMF. The detailed standardized synthesis steps for the complete transformation, including precise stoichiometric ratios and workup procedures, are outlined below.
- Prepare the trifluoromethyl-substituted olefine aldehyde substrate by reacting salicylaldehyde derivatives with trifluoroalkenyl bromides in the presence of a base like potassium carbonate.
- Perform the NHC-catalyzed cyclization of the aldehyde substrate in an organic solvent at room temperature for 3-10 hours to form the intermediate flavonoid.
- Execute the oxidative aromatization by adding elemental iodine and concentrated sulfuric acid, heating the mixture to 50-100°C for 2-5 hours to yield the final homoisoflavone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this NHC-catalyzed protocol offers transformative benefits that extend far beyond simple yield improvements. The elimination of precious metal catalysts such as palladium or rhodium, which are subject to volatile market pricing and supply constraints, represents a significant strategic advantage. By replacing these scarce resources with abundant organic catalysts and commodity chemicals like iodine, manufacturers can decouple their production costs from the fluctuations of the precious metals market. Furthermore, the mild reaction conditions reduce the energy load on manufacturing facilities, as the initial cyclization proceeds at room temperature, eliminating the need for extensive heating or cooling infrastructure during the most critical bond-forming step. This energy efficiency translates directly into lower utility costs and a reduced carbon footprint, enhancing the overall sustainability profile of the supply chain.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substitution of expensive catalytic systems with cost-effective organocatalysts. Traditional methods often require stoichiometric amounts of Lewis acids or specialized enzymes that are difficult to recover and recycle, leading to high consumable costs per kilogram of product. In contrast, the NHC catalyst is used in sub-stoichiometric quantities (10-20 mol%), and the oxidant (iodine) is inexpensive and readily available globally. Additionally, the simplified workup procedure, which often allows for purification via simple recrystallization rather than complex chromatographic separations, drastically reduces solvent consumption and labor hours. This streamlined downstream processing significantly lowers the Cost of Goods Sold (COGS), making the final API intermediate more competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials and reagents. The substrates, derived from salicylaldehydes and trifluoroalkenyl bromides, are commodity chemicals with established global supply networks, minimizing the risk of raw material shortages. Unlike bio-enzymatic processes that may require cold-chain logistics or have limited shelf-lives, the reagents in this chemical synthesis are stable and easy to store. The robustness of the reaction conditions also means that the process is less susceptible to batch failures due to minor variations in temperature or pressure, ensuring consistent output and reliable delivery schedules for downstream pharmaceutical customers. This reliability is critical for maintaining continuous production lines for life-saving antitumor medications.
- Scalability and Environmental Compliance: From a scale-up perspective, the process is inherently safer and easier to manage than exothermic Lewis acid reactions. The room-temperature initiation allows for better thermal control in large reactors, reducing the risk of runaway reactions. Furthermore, the avoidance of heavy metals simplifies the regulatory burden associated with residual metal limits in pharmaceutical products (ICH Q3D guidelines). The waste stream is predominantly organic and lacks toxic heavy metal contaminants, making wastewater treatment more straightforward and less costly. This alignment with green chemistry principles not only facilitates regulatory approval but also future-proofs the manufacturing site against increasingly stringent environmental regulations, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NHC-catalyzed synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and advantages of the technology for potential partners and licensees.
Q: What are the primary advantages of this NHC method over traditional Lewis acid catalysis?
A: The NHC-catalyzed method described in CN108727323B operates under significantly milder conditions (room temperature initiation) compared to harsh Lewis acid protocols. It eliminates the need for expensive transition metal catalysts, thereby reducing heavy metal contamination risks and simplifying downstream purification processes for pharmaceutical grades.
Q: Is the substrate scope limited for this trifluoromethyl homoisoflavone synthesis?
A: No, the patent demonstrates excellent substrate latitude. The method accommodates various substituents (R1) on the benzene ring, including alkyl, halogen, alkoxy, and nitro groups, as well as different aryl or alkenyl groups (R2) on the side chain, ensuring versatility for diverse drug discovery programs.
Q: How is the final product purified to meet high-purity standards?
A: The process allows for flexible purification strategies suitable for scale-up. The crude product can be effectively purified via recrystallization using mixed solvent systems (e.g., ethyl acetate/petroleum ether) or through column chromatography, ensuring the removal of unreacted starting materials and side products to achieve stringent purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Homoisoflavone Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of trifluoromethyl-substituted homoisoflavones in the development of next-generation antitumor therapies. As a premier CDMO partner, we possess the technical expertise to translate the innovative NHC-catalyzed protocols described in CN108727323B from bench-scale discovery to industrial reality. Our facility is equipped with state-of-the-art reactors capable of handling sensitive organocatalytic reactions under strictly controlled inert atmospheres, ensuring the highest levels of safety and reproducibility. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to seamlessly support your clinical trial material needs through to full commercial launch. Our rigorous QC labs enforce stringent purity specifications, guaranteeing that every batch meets the exacting standards required for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development pipeline. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this green synthesis route can optimize your budget. We encourage you to contact our technical procurement team today to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. Let us help you secure a sustainable, cost-effective, and reliable supply of these critical building blocks for your oncology portfolio.
