Scalable Synthesis of Aggregation-Induced Emission Iridium Complexes for Next-Gen OLEDs
Scalable Synthesis of Aggregation-Induced Emission Iridium Complexes for Next-Gen OLEDs
The recent technological disclosure in patent CN111187301B introduces a significant advancement in the field of phosphorescent materials, specifically focusing on a class of cyclometalated iridium complexes exhibiting remarkable Aggregation-Induced Emission (AIE) properties. This innovation addresses a critical bottleneck in organic light-emitting diode (OLED) technology, where traditional luminescent materials often suffer from efficiency loss at high concentrations. The patent details the successful preparation of six distinct iridium derivatives, designated as Ir1 through Ir6, which utilize 2-phenylpyridine derivatives as cyclometalated ligands and 2,2'-bipyridine as auxiliary ligands. These complexes are engineered to overcome the notorious Aggregation-Caused Quenching (ACQ) effect, thereby offering superior luminous efficiency in solid-state applications. For R&D directors and procurement specialists in the electronic chemicals sector, this represents a pivotal shift towards more robust and efficient emissive materials that can be synthesized through reliable transition metal catalysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of high-efficiency OLED emitters has been plagued by the phenomenon of Aggregation-Caused Quenching, where conventional fluorescent molecules exhibit strong emission in dilute solutions but lose their luminosity when concentrated or in the solid state. This quenching occurs due to detrimental intermolecular interactions, such as pi-pi stacking, which facilitate non-radiative decay pathways and drastically reduce device efficiency. In the context of industrial manufacturing, this limitation necessitates complex doping strategies or the use of excessive host materials to isolate the emitter molecules, thereby increasing material costs and complicating the device architecture. Furthermore, many traditional phosphorescent materials rely on expensive and scarce ligands that are difficult to synthesize on a large scale, leading to supply chain vulnerabilities and inconsistent batch-to-batch quality. The inability to maintain high quantum yields in the aggregated state remains a persistent barrier to the commercialization of next-generation display technologies.
The Novel Approach
The methodology outlined in the patent presents a transformative solution by leveraging the Aggregation-Induced Emission (AIE) mechanism within a transition metal complex framework. By strategically modifying the cyclometalated ligands with bulky substituents such as biphenyl, fluoro, and triphenylamine groups, the inventors have successfully restricted the intramolecular motion that typically leads to energy loss in the aggregated state. This structural design ensures that the iridium complexes not only maintain but actually enhance their phosphorescence intensity when transitioning from solution to solid film. The synthetic route employs a modular Suzuki cross-coupling strategy, allowing for the precise installation of diverse functional groups without compromising the integrity of the core iridium coordination sphere. This approach not only solves the quenching problem but also provides a versatile platform for tuning emission colors and energy levels, making it highly attractive for the fabrication of full-color OLED displays with improved stability and longevity.
Mechanistic Insights into Suzuki-Coupled Cyclometalation
The core of this synthetic innovation lies in the efficient construction of the cyclometalated ligands via palladium-catalyzed Suzuki cross-coupling reactions. The process begins with the reaction of 2-bromopyridine derivatives and aryl boronic acids in the presence of palladium acetate and potassium carbonate within an ethanol-water solvent system at 80°C. This mild yet effective catalytic cycle facilitates the formation of carbon-carbon bonds between the pyridine ring and various aryl groups, creating the necessary steric bulk to induce the AIE effect. Following ligand synthesis, the coordination step involves reacting the ligand with IrCl3·3H2O in a mixture of ethylene glycol monoethyl ether and water under nitrogen protection at 120°C. This forms a stable dichloro-bridged dimer intermediate, which is subsequently cleaved by 2,2'-bipyridine to yield the final cationic complex. The final anion exchange with KPF6 ensures the isolation of a stable, non-coordinating hexafluorophosphate salt, which is crucial for the electrochemical stability required in OLED devices.
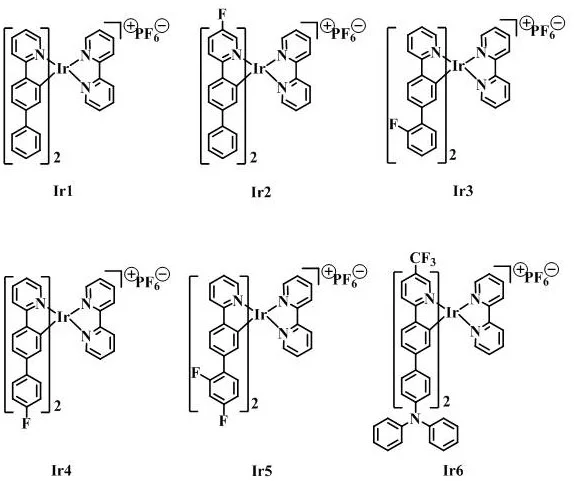
The structural diversity achieved through this mechanism is evident in the specific derivatives Ir1 through Ir6, each tailored with unique electronic and steric properties. As illustrated in the structural diagrams, variations such as the introduction of fluorine atoms in Ir2, Ir3, Ir4, and Ir5 serve to lower the HOMO energy levels, potentially blue-shifting the emission and improving electron transport properties. Meanwhile, the incorporation of a triphenylamine moiety in Ir6 introduces hole-transporting capabilities directly into the emitter molecule, simplifying device layering. The rigorous control over these substituent patterns allows for fine-tuning of the photophysical properties, ensuring that the resulting materials exhibit maximum emission intensity at specific water contents in mixed solvents, a key indicator of their AIE behavior. This level of molecular precision is essential for meeting the stringent purity and performance specifications demanded by high-end display manufacturers.
How to Synthesize Aggregation-Induced Emission Iridium Complexes Efficiently
The synthesis of these high-value phosphorescent materials follows a streamlined three-stage protocol that balances high yield with operational simplicity, making it suitable for both laboratory optimization and pilot-scale production. The initial phase focuses on the modular assembly of the cyclometalated ligands using robust Suzuki coupling conditions that tolerate a wide range of functional groups. Subsequent coordination with the iridium precursor requires careful control of temperature and atmosphere to prevent oxidation and ensure the formation of the correct stereoisomer. Finally, the purification process utilizes standard column chromatography techniques with dichloromethane and petroleum ether, ensuring the removal of residual palladium and iridium salts to meet electronic-grade purity standards. Detailed standardized synthetic steps for each specific derivative are provided in the technical guide below to assist process engineers in replicating these results.
- Synthesize the cyclometalated ligand intermediate via Suzuki cross-coupling of 2-bromopyridine derivatives and aryl boronic acids using palladium acetate catalyst at 80°C.
- Coordinate the synthesized ligand with IrCl3·3H2O in ethylene glycol monoethyl ether/water at 120°C under nitrogen to form the dichloro-bridged dimer intermediate.
- React the dimer with 2,2'-bipyridine and perform anion exchange with KPF6 to isolate the final cationic iridium complex with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the synthetic route described in this patent offers substantial advantages over traditional methods for producing phosphorescent OLED materials. The reliance on Suzuki cross-coupling utilizes widely available and cost-effective starting materials such as bromopyridines and aryl boronic acids, which are commodity chemicals with stable global supply chains. This reduces the risk of raw material shortages and price volatility that often plagues the sourcing of exotic heterocyclic compounds. Furthermore, the use of common solvents like ethanol, water, and ethylene glycol monoethyl ether simplifies waste management and solvent recovery processes, contributing to a more sustainable and cost-efficient manufacturing operation. The elimination of complex multi-step ligand syntheses in favor of a convergent approach significantly shortens the overall production timeline, enhancing the responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The implementation of palladium-catalyzed coupling reactions allows for the use of lower-cost precursors while maintaining high conversion rates, effectively driving down the cost of goods sold. By avoiding the need for specialized cryogenic conditions or ultra-high vacuum equipment, the capital expenditure required for setting up production lines is significantly minimized. The high yields reported in the examples, particularly for the ligand synthesis steps, indicate a material-efficient process that reduces waste generation and maximizes the output per batch. Additionally, the modular nature of the synthesis means that multiple derivatives can be produced on the same equipment train with minimal changeover time, further optimizing asset utilization and lowering overhead costs associated with dedicated production lines.
- Enhanced Supply Chain Reliability: The precursors required for this synthesis, including iridium chloride and various boronic acids, are established industrial chemicals with multiple qualified suppliers globally, ensuring a resilient supply chain. The robustness of the reaction conditions, which proceed efficiently in air for the ligand synthesis steps, reduces the dependency on specialized inert atmosphere infrastructure, thereby simplifying logistics and storage requirements. This accessibility of raw materials mitigates the risk of single-source dependency, allowing procurement managers to negotiate better terms and secure long-term supply agreements. The consistency of the synthetic method also ensures that quality parameters remain stable across different production batches, reducing the need for extensive incoming quality control testing and accelerating the release of finished goods to customers.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of homogeneous catalysis in standard solvent systems that are well-understood in chemical engineering. The ability to conduct key steps in aqueous-organic mixtures reduces the overall volume of volatile organic compounds (VOCs) emitted, aligning with increasingly stringent environmental regulations in the fine chemical industry. The straightforward workup procedures involving extraction and column chromatography can be readily adapted to continuous flow chemistry or large-scale batch reactors, facilitating the transition from gram-scale laboratory synthesis to kilogram or ton-scale commercial production. This scalability ensures that the technology can meet the growing demand for high-performance OLED materials without compromising on environmental safety or regulatory compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this AIE iridium complex technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the material's capabilities and limitations. Understanding these aspects is crucial for R&D teams evaluating the material for specific device architectures and for supply chain planners assessing the feasibility of large-scale adoption. The clarity provided here aims to accelerate the decision-making process for integrating these advanced phosphorescent materials into next-generation display and lighting products.
Q: What is the primary advantage of these iridium complexes over traditional fluorescent materials?
A: Unlike traditional fluorophores that suffer from Aggregation-Caused Quenching (ACQ) at high concentrations, these complexes exhibit Aggregation-Induced Emission (AIE), meaning their luminescence intensity significantly increases in the aggregated or solid state, making them ideal for OLED applications.
Q: What catalytic system is used for the ligand synthesis?
A: The synthesis utilizes a palladium-catalyzed Suzuki cross-coupling reaction. Specifically, palladium acetate is used with potassium carbonate as a base in an ethanol-water mixed solvent system at 80°C, ensuring high yields and environmental compatibility.
Q: Can these complexes be scaled for industrial OLED production?
A: Yes, the synthetic route relies on robust and widely available reagents such as aryl boronic acids and standard iridium salts. The use of common solvents like ethylene glycol monoethyl ether and dichloromethane facilitates straightforward scale-up from laboratory to commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iridium Complex Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercially viable fine chemical solutions, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely equipped to handle the complexities of organometallic synthesis, ensuring that the stringent purity specifications required for electronic grade materials are consistently met through our rigorous QC labs. We understand that the transition from patent to production involves nuanced challenges in catalyst removal and isomer separation, and our process chemists are dedicated to optimizing these parameters to deliver a product that exceeds industry standards. By partnering with us, clients gain access to a supply chain that is not only robust but also deeply knowledgeable about the specific physicochemical properties of aggregation-induced emission materials.
We invite procurement leaders and CTOs to engage with our Customized Cost-Saving Analysis service, where we evaluate your current material sourcing strategies against the efficiencies offered by this new synthetic route. Our technical procurement team is ready to provide specific COA data and route feasibility assessments tailored to your unique production requirements. Whether you are developing high-resolution smartphone displays or large-area lighting panels, our commitment to quality and delivery reliability ensures that your project timelines are met without compromise. Contact us today to discuss how we can support your transition to high-efficiency AIE phosphorescent materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
