Advanced Nickel-Catalyzed Thioester Synthesis Technology for High-Purity Pharmaceutical Intermediates at Commercial Scale
Patent CN112239384B presents a groundbreaking methodology for synthesizing thioester compounds through a nickel-catalyzed carbonylation reaction utilizing sulfonyl chloride as an innovative sulfur source. This novel approach addresses critical limitations in conventional thioester synthesis by eliminating the need for malodorous and catalyst-poisoning mercaptans while maintaining exceptional functional group tolerance. The process demonstrates remarkable versatility across diverse substrate classes, enabling the production of complex thioester structures essential for advanced pharmaceutical applications. With reaction conditions optimized at 120°C for 16 hours in N-methylpyrrolidone solvent, this method achieves high yields without requiring additional oxidants or reductants. The strategic use of nickel triflate with dtbpy ligand and molybdenum carbonyl creates a highly efficient catalytic system that significantly enhances the practicality of thioester synthesis for industrial implementation. This patent represents a substantial advancement in synthetic methodology with direct implications for pharmaceutical intermediate manufacturing.
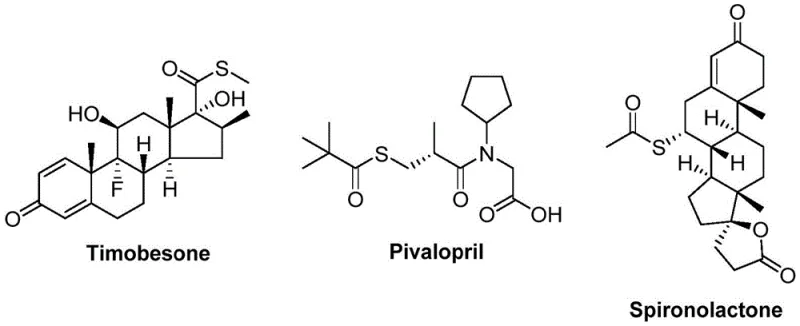
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to thioester synthesis predominantly rely on acylation reactions between mercaptans and carboxylic acid derivatives, presenting significant operational challenges for industrial-scale production. Mercaptans are notoriously difficult to handle due to their extremely unpleasant odor and high volatility, creating hazardous working conditions that require specialized containment systems. Furthermore, these sulfur-containing compounds readily poison transition metal catalysts, severely limiting their compatibility with modern catalytic methodologies. The narrow functional group tolerance of conventional methods restricts substrate diversity, making it challenging to synthesize complex thioester structures required for advanced pharmaceutical applications. Additional complications arise from the need for stoichiometric oxidants or reductants in many protocols, generating substantial waste streams that complicate purification and increase environmental impact. These cumulative limitations have constrained the broader adoption of thioester chemistry in commercial manufacturing despite its demonstrated value in drug development pipelines.
The Novel Approach
The patented methodology overcomes these longstanding challenges through an innovative nickel-catalyzed carbonylation strategy that employs sulfonyl chloride as a superior sulfur source. This approach eliminates the problematic characteristics of mercaptans while maintaining excellent reactivity across a wide range of substrates. The catalytic system comprising nickel triflate, dtbpy ligand, and molybdenum carbonyl operates under mild conditions (120°C) without requiring additional redox agents, significantly simplifying process design. The exceptional functional group tolerance enables the synthesis of structurally diverse thioesters that were previously inaccessible through conventional methods. Crucially, the use of commercially available and cost-effective starting materials—arylboronic acids and sulfonyl chlorides—provides substantial economic advantages while ensuring supply chain reliability. This method represents a paradigm shift in thioester synthesis by combining operational simplicity with industrial scalability.
Mechanistic Insights into Nickel-Catalyzed Thioester Synthesis
The catalytic cycle begins with oxidative addition of the sulfonyl chloride to the nickel(0) species generated in situ from nickel(II) precatalyst reduction. This key step forms a nickel-sulfonate complex that undergoes transmetalation with the arylboronic acid to create an aryl-nickel intermediate. Subsequent carbon monoxide insertion into the Ni-Ar bond generates an acyl-nickel species, which then undergoes reductive elimination to yield the final thioester product while regenerating the active nickel catalyst. The dtbpy ligand plays a critical role in stabilizing the various nickel oxidation states throughout this cycle, while molybdenum carbonyl serves as both a CO source and reducing agent. This mechanistic pathway explains the high efficiency observed across diverse substrates, as the reaction avoids common side reactions through precise control of intermediate species. The absence of strong oxidants or reductants prevents undesired functional group transformations, contributing to exceptional product purity.
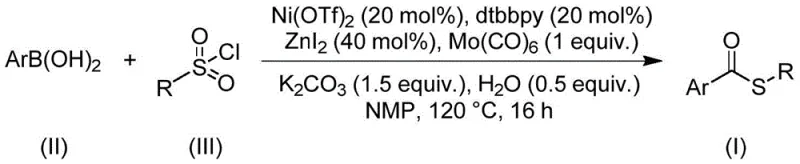
Impurity control is achieved through multiple synergistic factors inherent to this catalytic system. The mild reaction temperature (120°C) prevents thermal decomposition pathways that commonly generate byproducts in alternative methods. The carefully balanced stoichiometry of potassium carbonate and zinc iodide maintains optimal pH conditions that suppress hydrolysis side reactions. The high chemoselectivity of the nickel catalyst ensures preferential formation of the desired C-S bond over competing reaction pathways. Additionally, the use of NMP as solvent provides excellent solubility for all reaction components while minimizing unwanted solvolysis reactions. These combined features result in consistently high purity profiles that meet stringent pharmaceutical requirements without requiring extensive purification beyond standard column chromatography.
How to Synthesize Thioester Compounds Efficiently
This patented methodology provides a robust framework for synthesizing diverse thioester compounds through a streamlined nickel-catalyzed carbonylation process. The reaction sequence begins with careful preparation of the catalytic system using commercially available nickel triflate and dtbpy ligand, followed by controlled addition of all components under inert atmosphere to prevent oxidation. The optimized temperature profile at 120°C ensures complete conversion while maintaining selectivity across various functional groups. Detailed standardized synthesis procedures have been developed to ensure consistent results from laboratory scale through commercial production volumes. The following step-by-step guide outlines the precise methodology for achieving optimal yields and purity in thioester synthesis.
- Prepare reaction mixture by combining arylboronic acid, sulfonyl chloride, nickel triflate catalyst, dtbpy ligand, molybdenum carbonyl, potassium carbonate, zinc iodide, water, and NMP solvent in Schlenk tube.
- Heat mixture to 120°C under inert atmosphere maintaining consistent temperature for precisely 16 hours to ensure complete conversion without side reactions.
- Perform post-treatment via filtration followed by silica gel mixing and column chromatography purification to isolate high-purity thioester product meeting pharmaceutical specifications.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points in traditional manufacturing approaches. The strategic selection of readily available starting materials significantly enhances supply chain resilience while reducing dependency on specialized or hazardous reagents. The simplified process design minimizes equipment requirements and operational complexity, translating to more efficient resource allocation throughout production cycles. These advantages collectively contribute to improved manufacturing economics without compromising product quality or regulatory compliance.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts typically required in alternative methods provides significant cost savings through reduced raw material expenses and simplified catalyst recovery processes. The use of inexpensive sulfonyl chloride instead of problematic mercaptans removes costly containment requirements while avoiding catalyst poisoning that would necessitate frequent catalyst replacement. The streamlined purification process reduces solvent consumption and processing time, contributing to substantial operational cost reductions without requiring specific percentage claims.
- Enhanced Supply Chain Reliability: All starting materials—arylboronic acids, sulfonyl chlorides, and catalyst components—are commercially available from multiple global suppliers with established distribution networks. This broad sourcing capability ensures consistent material availability regardless of regional supply disruptions. The stability of sulfonyl chloride compared to volatile mercaptans enables safer transportation and longer shelf life, reducing logistics complications and inventory management challenges while maintaining reliable production schedules.
- Scalability and Environmental Compliance: The reaction demonstrates excellent scalability from laboratory to commercial production volumes due to its robustness under consistent operating conditions. The absence of hazardous reagents simplifies waste stream management and reduces environmental impact compared to conventional methods requiring strong oxidants or reductants. The simplified process design minimizes energy consumption through moderate temperature requirements and eliminates specialized equipment needs, enhancing overall sustainability while meeting increasingly stringent regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding the implementation of this patented thioester synthesis methodology. These answers are derived directly from the patent documentation and reflect the specific advantages demonstrated through experimental validation.
Q: What are key advantages of sulfonyl chloride over mercaptan as sulfur source?
A: Sulfonyl chloride eliminates mercaptan's foul odor and catalyst poisoning issues while providing stable handling characteristics. This enables broader substrate scope with exceptional functional group tolerance without requiring additional oxidants or reductants.
Q: How does this method ensure high purity for pharmaceutical applications?
A: The mild reaction conditions (120°C) prevent thermal decomposition while precise stoichiometry control minimizes hydrolysis side reactions. High chemoselectivity combined with standard column chromatography yields products meeting stringent pharmaceutical purity requirements.
Q: What makes this process suitable for commercial scale-up?
A: The method uses cost-effective commercially available materials with robust reaction conditions that scale seamlessly from laboratory to industrial volumes. Simplified waste management from eliminating hazardous reagents enhances environmental compliance during scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
Our patented methodology represents a significant advancement in thioester synthesis technology with profound implications for pharmaceutical intermediate manufacturing. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs. Our specialized expertise in complex molecule synthesis ensures seamless transition from laboratory development to full-scale manufacturing with consistent quality outcomes that meet global regulatory standards.
We invite you to initiate a technical discussion with our team to explore how this innovative approach can enhance your specific manufacturing requirements. Request a Customized Cost-Saving Analysis from our technical procurement team to evaluate potential benefits for your production pipeline, including specific COA data and route feasibility assessments tailored to your unique needs.
