Industrial Scale Synthesis of Calcipotriol from Stigmasterol via Fermentation and Wittig Coupling
Industrial Scale Synthesis of Calcipotriol from Stigmasterol via Fermentation and Wittig Coupling
The global demand for effective psoriasis treatments continues to drive innovation in the synthesis of Vitamin D analogs, specifically calcipotriol. A pivotal advancement in this domain is detailed in patent CN105624215B, which discloses a novel, streamlined preparation method starting from stigmasterol rather than the traditional vitamin D2. This technological breakthrough addresses critical bottlenecks in the supply chain of high-purity pharmaceutical intermediates by reducing the synthetic route from twelve steps to just seven. By leveraging microbial fermentation for the critical 1-alpha-hydroxylation and employing a robust Wittig coupling strategy for side-chain reconstruction, this process offers a compelling value proposition for manufacturers seeking a reliable calcipotriol intermediate supplier. The integration of biocatalysis with classical organic synthesis not only enhances regioselectivity but also drastically simplifies the purification protocols required for clinical-grade material.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of calcipotriol has relied heavily on vitamin D2 or D3 as the starting material, a strategy fraught with significant economic and technical challenges. The conventional synthetic pathways, such as those reported by Calverley, typically involve upwards of twelve distinct chemical transformations, each introducing potential yield losses and impurity profiles that complicate downstream processing. The inherent instability of the previtamin D triene system necessitates rigorous temperature control and often results in the formation of difficult-to-remove isomers, thereby inflating the cost of goods sold (COGS). Furthermore, the chemical introduction of the 1-alpha-hydroxyl group in traditional routes often requires harsh oxidizing conditions or multiple protection-deprotection cycles, which generate substantial chemical waste and pose safety risks during commercial scale-up of complex steroid intermediates. These factors collectively contribute to a supply chain that is vulnerable to raw material price volatility and limited production capacity.
The Novel Approach
In stark contrast, the methodology outlined in the patent utilizes stigmasterol, an abundant and inexpensive phytosterol, as the foundational building block, fundamentally altering the cost structure of calcipotriol manufacturing. This innovative route bypasses the instability issues associated with vitamin D2 by constructing the sensitive triene system later in the synthesis, specifically during the photo-isomerization step. The process employs a highly efficient seven-step sequence that integrates biological catalysis for the initial functionalization, followed by oxidative cleavage and carbon-carbon bond formation via Wittig chemistry. This strategic reordering of synthetic logic allows for cost reduction in vitamin D analog manufacturing by minimizing the number of unit operations and maximizing the throughput of each reactor vessel. The use of robust reagents like tert-butyldimethylsilyl chloride for protection and tetrabutylammonium fluoride for deprotection ensures that the process remains compatible with standard stainless steel equipment found in modern multipurpose API plants.
Mechanistic Insights into Microbial Hydroxylation and Wittig Coupling
The cornerstone of this synthetic strategy is the regioselective introduction of the 1-alpha-hydroxyl group via microbial fermentation using strains such as Penicillium oxalicum or Aspergillus terreus. Unlike chemical hydroxylation, which often suffers from poor selectivity and requires extensive chromatographic purification, the enzymatic machinery of these fungi targets the C-1 position with high fidelity under mild physiological conditions (pH 4-9, 26-27°C). This biocatalytic step effectively sets the stereochemistry early in the synthesis, ensuring that the resulting compound 3 possesses the correct configuration for subsequent transformations without the need for chiral resolution. Following protection of the hydroxyl groups, the side chain of the steroid nucleus is cleaved via ozonolysis at low temperatures (-50 to -15°C), generating a ketone intermediate that serves as the electrophile for the critical carbon-carbon bond-forming event. The subsequent Wittig reaction with the phosphonium salt (compound 6) reconstructs the unsaturated side chain characteristic of calcipotriol, a step that is carefully controlled at elevated temperatures (90-120°C) to ensure complete conversion and minimize the formation of Z-isomers.
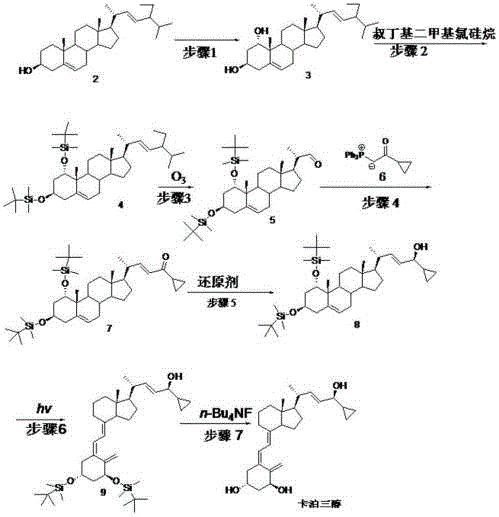
Following the construction of the carbon skeleton, the synthesis proceeds through a stereoselective reduction of the ketone moiety using borohydride reagents in the presence of metal salts like calcium chloride or cerium trichloride. This Luche-type reduction condition is crucial for controlling the diastereoselectivity at the C-20 position, ensuring the formation of the desired alcohol intermediate (compound 8) with high purity. The final activation of the vitamin D pharmacophore is achieved through photo-isomerization using UV light (270-290 nm) in the presence of anthracene as a photosensitizer, which facilitates the electrocyclic ring-opening of the steroid B-ring. This mechanistic sequence demonstrates a sophisticated understanding of steroid chemistry, balancing the reactivity of sensitive functional groups with the need for operational simplicity in a manufacturing environment. The final deprotection step using fluoride sources cleanly removes the silyl protecting groups to yield the active pharmaceutical ingredient, ready for formulation.
How to Synthesize Calcipotriol Efficiently
The execution of this synthesis requires precise control over reaction parameters, particularly during the fermentation and photochemical stages, to ensure consistent quality and yield. The process begins with the inoculation of the selected fungal strain into a nutrient medium containing glucose and corn steep liquor, where the substrate feed concentration is carefully maintained between 1 g/L and 10 g/L to optimize biotransformation efficiency. Subsequent chemical steps involve standard workup procedures such as extraction, washing, and recrystallization, which are well-suited for transfer from laboratory to pilot plant scales. For detailed operational parameters, including specific solvent ratios, stirring speeds, and isolation techniques, operators should refer to the standardized protocols derived from the patent examples.
- Perform microbial fermentation using Penicillium oxalicum or Aspergillus terreus on stigmasterol to introduce the 1-alpha-hydroxyl group.
- Protect the hydroxyl groups with TBDMSCl, followed by ozonolysis to cleave the original side chain.
- Execute a Wittig reaction to attach the new side chain, followed by reduction, UV photo-isomerization, and final deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift from vitamin D2 to stigmasterol as the starting material represents a significant opportunity for cost optimization and supply chain stabilization. Stigmasterol is a commodity chemical derived from soybean oil processing, meaning its availability is tied to the robust agricultural sector rather than the niche market of vitamin extraction, leading to substantially lower raw material costs and reduced price volatility. The reduction in the total number of synthetic steps from twelve to seven directly correlates to a decrease in labor hours, solvent consumption, and waste disposal fees, all of which contribute to a leaner manufacturing cost profile. Furthermore, the elimination of complex chiral separation steps typically required in older routes simplifies the quality control workflow, allowing for faster batch release times and improved inventory turnover rates for supply chain managers.
- Cost Reduction in Manufacturing: The utilization of microbial fermentation for the initial hydroxylation step eliminates the need for expensive chiral catalysts or stoichiometric oxidizing agents that are common in purely chemical routes. This biocatalytic approach operates under ambient pressure and moderate temperatures, significantly reducing energy consumption compared to high-pressure hydrogenation or cryogenic reactions. Additionally, the high yield reported in the patent examples for the Wittig coupling and reduction steps minimizes the loss of valuable intermediates, ensuring that the overall material throughput is maximized. By streamlining the synthesis, manufacturers can achieve a lower cost per kilogram of active ingredient, providing a competitive edge in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Relying on stigmasterol mitigates the risk of supply disruptions associated with the seasonal availability of vitamin D precursors. The fermentation step can be scaled independently in large bioreactors, decoupling the production of the key intermediate from the constraints of chemical synthesis capacity. The use of common organic solvents such as ethyl acetate, methanol, and dichloromethane ensures that reagent sourcing is straightforward and not dependent on specialized chemical suppliers. This flexibility allows production facilities to maintain continuous operation even when specific reagents face temporary market shortages, thereby securing the continuity of supply for downstream formulation partners.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by reducing the E-factor (mass of waste per mass of product) through fewer synthetic steps and higher overall yields. The fermentation broth can be treated using standard biological wastewater treatment methods, and the organic solvents used in the chemical steps are amenable to recovery and recycling via distillation. The avoidance of heavy metal catalysts in the reduction step (using calcium or cerium salts instead) simplifies the removal of trace metals to meet stringent ICH Q3D guidelines, reducing the burden on analytical laboratories. This environmental compatibility facilitates easier regulatory approval and aligns with the sustainability goals of modern pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of the supply source.
Q: What is the primary advantage of using stigmasterol over vitamin D2 for calcipotriol synthesis?
A: Stigmasterol is a readily available, low-cost plant sterol compared to the expensive and unstable vitamin D2. The patented route reduces the step count from 12 to 7, significantly improving overall yield and cost-efficiency.
Q: How does the microbial fermentation step improve regioselectivity?
A: Using specific strains like Penicillium oxalicum allows for the direct introduction of the 1-alpha-hydroxyl group under mild conditions, avoiding complex chemical protection-deprotection sequences required in traditional chemical oxidation methods.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the patent explicitly designs the process for industrialization. Key steps like fermentation and the Wittig coupling are optimized for scalability, using common solvents and reagents that are easily sourced for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Calcipotriol Supplier
The synthesis route described in patent CN105624215B exemplifies the type of process innovation that drives value in the fine chemical industry, offering a pathway to high-quality calcipotriol at a competitive cost. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets rigorous quality standards. Our state-of-the-art facilities are equipped to handle both the fermentation and chemical synthesis components of this route, with stringent purity specifications enforced through our rigorous QC labs to guarantee the absence of genotoxic impurities and residual solvents.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific product portfolio. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this stigmasterol-based route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of our high-purity calcipotriol intermediates into your supply chain.
