Advanced Iridium-Catalyzed C-H Amination for Scalable Lactam Manufacturing
Advanced Iridium-Catalyzed C-H Amination for Scalable Lactam Manufacturing
The pharmaceutical industry continuously seeks robust methodologies for constructing nitrogen-containing heterocycles, particularly gamma-lactams and delta-lactams, which serve as critical scaffolds in numerous bioactive molecules and clinical drug candidates. Patent CN111170918B introduces a transformative approach to synthesizing these valuable structures through intramolecular C-H amination, addressing long-standing challenges in selectivity and operational complexity. This technology leverages a specialized iridium catalyst system to directly construct carbon-nitrogen bonds from dioxazolone precursors, bypassing the need for pre-functionalized substrates that often plague traditional synthetic routes. For R&D directors and procurement specialists alike, this innovation represents a significant leap forward in process efficiency, offering a pathway to high-purity intermediates with reduced material costs and simplified downstream processing requirements.
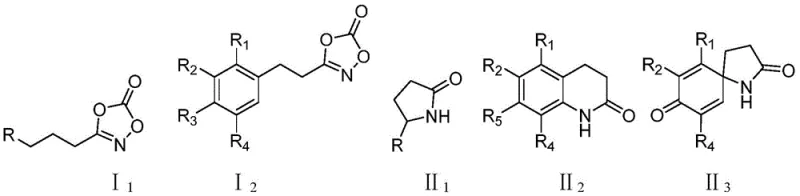
The strategic value of this patent lies in its ability to access diverse lactam architectures, including spiro-lactams, from readily available carboxylic acid derivatives. By utilizing a catalytic cycle that tolerates a wide range of functional groups, the method enables the rapid generation of chemical libraries essential for drug discovery programs. Furthermore, the mild reaction conditions described in the patent, typically ranging from 40°C to 60°C, ensure compatibility with sensitive functional moieties that might degrade under harsher thermal regimes. This capability is paramount for manufacturers aiming to produce complex API intermediates where maintaining structural integrity throughout the synthesis is non-negotiable for regulatory compliance and final product efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of lactam rings has relied heavily on classical cyclization strategies involving the reaction of amino acids or their derivatives, which often necessitate multi-step protection and deprotection sequences. These traditional pathways are frequently hampered by the requirement for stoichiometric activating agents, harsh reaction conditions, and the generation of substantial chemical waste, all of which drive up the cost of goods sold (COGS) for the final active pharmaceutical ingredient. Moreover, alternative C-H amination strategies have struggled with a fundamental chemoselectivity issue: the tendency of nitrene intermediates to undergo Curtius rearrangement rather than the desired C-H insertion. As illustrated in the background art, this side reaction leads to the formation of isocyanates instead of the target lactam, drastically reducing yield and complicating purification efforts.
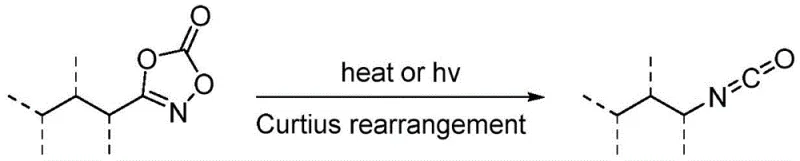
The Novel Approach
The methodology disclosed in CN111170918B elegantly circumvents these obstacles by employing a highly selective iridium catalyst that favors C-H amination over rearrangement pathways. This novel approach utilizes dioxazolones as stable nitrene precursors, which, under the influence of the specific iridium complex, undergo efficient intramolecular cyclization to form the desired lactam ring. The process eliminates the need for expensive and toxic heavy metal oxidants often required in other C-H functionalization reactions, thereby aligning with green chemistry principles. For supply chain managers, this translates to a more reliable sourcing strategy, as the raw materials—simple carboxylic acids and hydroxylamine—are commodity chemicals with stable global supply chains, reducing the risk of production bottlenecks associated with exotic reagents.
Mechanistic Insights into Iridium-Catalyzed C-H Amination
At the heart of this technological breakthrough is a uniquely designed iridium catalyst formed by the coordination of pentamethylcyclopentadienyl iridium dichloride with a 2-(4,5-dihydro-1H-imidazol-2-yl)pyridine ligand. This specific ligand architecture is crucial; its simple structure and low cost stand in stark contrast to the elaborate chiral ligands often employed in asymmetric catalysis, yet it delivers exceptional activity and selectivity. The catalyst facilitates the activation of the dioxazolone ring to generate a metal-nitrenoid species, which then selectively inserts into the proximal sp3 C-H bond. This mechanistic pathway is kinetically favored over the thermodynamic sink of the Curtius rearrangement, ensuring that the reaction flux is directed almost exclusively toward lactam formation.
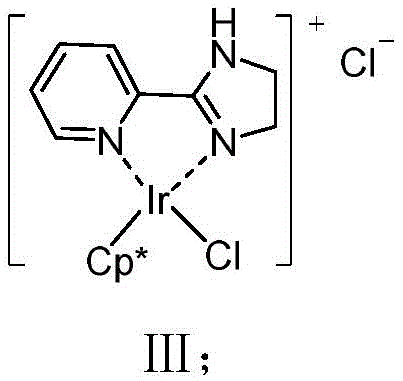
From an impurity control perspective, the high selectivity of this catalytic system is a major advantage for process chemistry teams. By minimizing the formation of isocyanate byproducts and other rearrangement derivatives, the crude reaction profile is significantly cleaner, which simplifies the workup and purification stages. The patent details the use of sodium tetrakis[3,5-bis(trifluoromethyl)phenyl]borate as an additive, which likely plays a role in stabilizing the cationic iridium species or facilitating the departure of the leaving group during nitrene formation. Understanding these mechanistic nuances allows process engineers to fine-tune reaction parameters, such as solvent polarity and temperature, to maximize throughput while maintaining the stringent purity specifications required for pharmaceutical grade intermediates.
How to Synthesize Gamma-Lactams Efficiently
The practical implementation of this synthesis route is straightforward and amenable to standard laboratory and pilot plant equipment. The protocol begins with the in situ or ex situ preparation of the dioxazolone substrate from the corresponding carboxylic acid using carbonyldiimidazole (CDI) and hydroxylamine hydrochloride, a transformation that proceeds efficiently at room temperature. Once the precursor is ready, it is subjected to the cyclization conditions in the presence of the iridium catalyst and the borate additive. The reaction typically reaches completion within 10 to 14 hours at moderate temperatures, after which the solvent is removed, and the product is isolated via standard column chromatography. Detailed standardized synthesis steps are provided in the guide below.
- Prepare the iridium catalyst by reacting pentamethylcyclopentadienyl iridium dichloride dimer with 2-(4,5-dihydro-1H-imidazol-2-yl)pyridine in dichloromethane at room temperature.
- Mix the dioxazolone substrate with the prepared iridium catalyst and sodium tetrakis[3,5-bis(trifluoromethyl)phenyl]borate additive in an organic solvent such as dichloromethane.
- Heat the reaction mixture to 40-60°C for 10-14 hours, then purify the resulting lactam product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this C-H amination technology offers tangible benefits that extend beyond mere chemical elegance. The primary driver for cost reduction lies in the catalyst design; by utilizing a ligand that is structurally simple and commercially inexpensive, the overall cost of the catalytic system is drastically lowered compared to systems relying on proprietary or complex chiral ligands. Furthermore, the elimination of stoichiometric oxidants and the use of mild reaction conditions reduce energy consumption and the burden on waste treatment facilities. These factors collectively contribute to a more sustainable and economically viable manufacturing process, allowing for competitive pricing in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of commodity starting materials and a low-cost catalyst system. Unlike traditional methods that may require multiple steps with low overall yields, this direct C-H functionalization approach consolidates the synthesis into fewer operational units. The avoidance of expensive protecting group manipulations and the high atom economy of the cyclization step mean that less raw material is wasted. Additionally, the simplified purification process resulting from high reaction selectivity reduces the consumption of silica gel and solvents during chromatography, further driving down the variable costs associated with large-scale production runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on widely available reagents. The key components, including the iridium source, the imidazole-pyridine ligand, and the dioxazolone precursors, are sourced from established chemical supply chains with minimal geopolitical risk. The robustness of the reaction conditions, which do not require cryogenic cooling or high-pressure reactors, means that the process can be executed in a broader range of manufacturing facilities, including those in emerging markets. This flexibility allows for diversified sourcing strategies, ensuring continuity of supply even in the face of regional disruptions or logistical challenges.
- Scalability and Environmental Compliance: Scaling this technology from gram to kilogram quantities is facilitated by the inherent safety of the reaction parameters. Operating at temperatures between 40°C and 60°C eliminates the thermal hazards associated with exothermic runaway reactions common in high-energy processes. From an environmental standpoint, the method aligns with modern regulatory expectations by minimizing the use of hazardous reagents and generating less toxic waste. The ability to recycle the organic solvent, such as dichloromethane or ethyl acetate, further enhances the environmental profile, making it easier for manufacturers to meet increasingly stringent emissions and waste disposal regulations without incurring prohibitive compliance costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iridium-catalyzed lactam synthesis. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for evaluating the technology's fit for your specific application. Whether you are assessing the feasibility of scaling this route or evaluating the purity profile of the resulting intermediates, these insights are designed to clarify the operational realities of the process.
Q: How does this method prevent Curtius rearrangement during lactam synthesis?
A: The patented iridium catalyst exhibits high selectivity for intramolecular C-H amination, effectively suppressing the competing Curtius rearrangement pathway that typically leads to isocyanate byproducts under conventional conditions.
Q: What are the cost advantages of the ligand used in this catalyst system?
A: The process utilizes 2-(4,5-dihydro-1H-imidazol-2-yl)pyridine, a ligand characterized by a simple structure and low market price compared to complex chiral ligands, significantly reducing the overall catalyst cost.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the reaction operates under mild temperatures (40-60°C) and uses commercially available solvents like dichloromethane, making it highly suitable for scale-up without requiring extreme pressure or cryogenic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Lactam Supplier
As the demand for complex nitrogenous heterocycles continues to grow in the pharmaceutical sector, partnering with a CDMO that possesses deep expertise in advanced catalytic methodologies is essential. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging cutting-edge technologies like the iridium-catalyzed C-H amination described in CN111170918B to deliver superior results. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of gamma-lactam or delta-lactam intermediate meets the exacting standards required for downstream API synthesis.
We invite you to discuss how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact us to request specific COA data for our lactam portfolio and to receive comprehensive route feasibility assessments that demonstrate the commercial potential of integrating this technology into your production pipeline.
