Advanced C-H Amination Technology for Scalable Gamma-Lactam and Delta-Lactam Production
Introduction to Next-Generation Lactam Synthesis
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct nitrogen-containing heterocycles, which serve as critical scaffolds in countless bioactive molecules. A significant technological breakthrough in this domain is detailed in Chinese Patent CN111170918A, which discloses a robust method for synthesizing γ-lactam and δ-lactam derivatives via intramolecular C-H amination. This innovation addresses long-standing challenges in organic synthesis by utilizing a highly active and selective iridium catalyst system that operates under mild conditions. For R&D directors and process chemists, this patent represents a pivotal shift away from traditional, cumbersome synthetic routes towards a more direct and atom-economical approach. The core of this technology lies in its ability to directly construct C-N bonds from dioxazolone precursors, bypassing the need for pre-functionalized substrates that often generate excessive waste. By leveraging this proprietary methodology, manufacturers can achieve superior control over reaction selectivity, ensuring high purity profiles essential for regulatory compliance in API manufacturing.
Furthermore, the versatility of this catalytic system extends beyond simple lactams to include complex spiro-lactam architectures, which are increasingly valuable in modern drug discovery programs targeting central nervous system disorders and oncology. The patent explicitly highlights the use of a simplified ligand framework that drastically reduces the economic barrier associated with precious metal catalysis. This is not merely an academic exercise but a commercially viable strategy that aligns with the industry's push for greener, more sustainable chemistry. As we delve deeper into the technical specifics, it becomes evident that this process offers a compelling value proposition for supply chain leaders looking to secure reliable sources of high-value intermediates while mitigating the risks associated with volatile raw material markets.
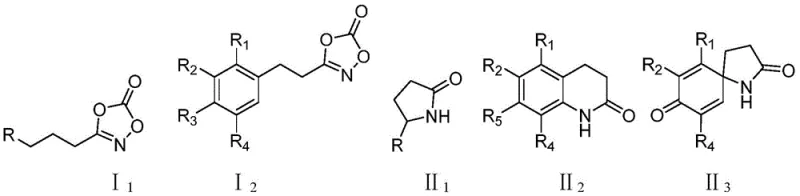
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct synthesis of lactams via C-H amination has been plagued by significant mechanistic hurdles that limited its industrial applicability. The primary obstacle has been the inherent instability of nitrene intermediates generated during the reaction. Under conventional reaction conditions, these reactive species are highly susceptible to undergoing Curtius rearrangement, a side reaction that leads to the formation of isocyanates rather than the desired cyclic amine products. This competing pathway not only consumes valuable starting materials but also introduces difficult-to-remove impurities that complicate downstream purification processes. Additionally, prior art solutions, such as those reported by the Chang project group, often relied on catalysts that were prohibitively expensive and synthetically challenging to prepare. These legacy systems frequently required distinct catalyst formulations for different types of lactams, forcing manufacturers to maintain complex inventory lists and incur high licensing or procurement costs. The reliance on sophisticated chiral ligands further exacerbated the cost structure, making these routes economically unfeasible for large-scale production of commodity intermediates.
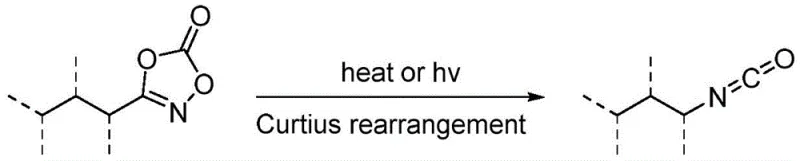
The Novel Approach
In stark contrast to these legacy limitations, the methodology described in CN111170918A introduces a paradigm shift through the deployment of a novel iridium catalyst complexed with 2-(4,5-dihydro-1H-imidazol-2-yl)pyridine. This specific ligand architecture is characterized by its structural simplicity and low cost, yet it imparts exceptional activity and selectivity to the metal center. The new approach successfully inhibits the detrimental Curtius rearrangement, channeling the reaction energy exclusively towards the desired intramolecular C-H insertion. This results in the clean formation of γ-lactams, δ-lactams, and even spiro-lactams from a single, versatile catalyst system. The operational simplicity is another major advantage; reactions proceed efficiently in common organic solvents like dichloromethane or hexafluoroisopropanol at moderate temperatures ranging from 40°C to 60°C. This eliminates the need for cryogenic conditions or high-pressure equipment, thereby reducing capital expenditure and energy consumption. For procurement managers, this universality means a single catalyst source can support the production of a diverse library of lactam intermediates, streamlining supply chain logistics and reducing overall operational complexity.
Mechanistic Insights into Iridium-Catalyzed C-H Amination
At the heart of this technological advancement is the unique electronic and steric environment created by the iridium-ligand complex. The catalyst, designated as Formula III in the patent, features a pentamethylcyclopentadienyl (Cp*) moiety coordinated with the bidentate imidazole-pyridine ligand. This configuration stabilizes the high-valent iridium-nitrenoid species formed upon activation of the dioxazolone substrate. Unlike less selective catalysts that allow the nitrene to rearrange, this specific coordination geometry favors a concerted insertion mechanism into the proximal C-H bond. The presence of the additive, sodium tetrakis(3,5-bis(trifluoromethyl)phenyl)borate (NaBArF4), plays a crucial role in generating the cationic active species by abstracting a chloride ligand, thereby increasing the electrophilicity of the metal center. This enhanced electrophilicity is key to activating the relatively inert C-H bonds found in alkyl and aryl chains, enabling the cyclization to occur under mild thermal conditions. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters for specific substrates, as it highlights the delicate balance between catalyst activation and intermediate stability.
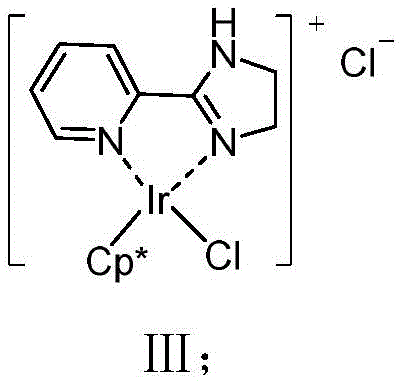
From an impurity control perspective, the suppression of the Curtius rearrangement is the most critical aspect of this mechanism. In traditional nitrene chemistry, the migration of the R-group to the nitrogen atom is often kinetically favored over C-H insertion. However, the steric bulk and electronic properties of the Cp*-Ir-ligand framework appear to lower the activation energy barrier for C-H insertion relative to rearrangement. This kinetic preference ensures that the reaction trajectory remains fixed on the formation of the lactam ring. Furthermore, the mild reaction temperatures (40-60°C) prevent thermal degradation of the sensitive dioxazolone precursors, which can otherwise decompose into various oligomeric byproducts. The result is a reaction profile with a significantly cleaner crude mixture, reducing the burden on purification units such as column chromatography or crystallization. For quality assurance teams, this translates to a more consistent impurity profile and higher overall yields, directly impacting the cost of goods sold (COGS) and the speed of batch release.
How to Synthesize Gamma-Lactams Efficiently
The practical implementation of this synthesis route is designed for ease of adoption in both laboratory and pilot plant settings. The process begins with the preparation of the dioxazolone precursor from readily available carboxylic acids using carbonyldiimidazole (CDI) and hydroxylamine hydrochloride, a standard and scalable transformation. Once the precursor is secured, the key C-H amination step requires precise stoichiometry of the iridium catalyst and the borate additive to ensure maximum turnover. The reaction is typically conducted in dichloromethane, though alternative solvents like ethyl acetate or hexafluoroisopropanol can be employed depending on substrate solubility. Following the reaction, standard workup procedures involving solvent removal and column chromatography yield the pure lactam product. The detailed standardized synthesis steps see the guide below.
- Preparation of the Iridium Catalyst: Complex pentamethylcyclopentadienyl iridium dichloride dimer with 2-(4,5-dihydro-1H-imidazol-2-yl)pyridine ligand in dichloromethane at room temperature.
- Substrate Activation: React the corresponding carboxylic acid with carbonyldiimidazole (CDI) and hydroxylamine hydrochloride to generate the dioxazolone precursor.
- C-H Amination Reaction: Treat the dioxazolone with the iridium catalyst and NaBArF4 additive in an organic solvent (e.g., DCM or HFIP) at 40-60°C to yield the target lactam.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the adoption of this C-H amination technology offers transformative benefits. The primary driver of value is the drastic simplification of the catalyst system. By replacing complex, custom-synthesized ligands with a simple, commercially available imidazole-pyridine derivative, the raw material costs associated with catalysis are significantly reduced. This cost efficiency is compounded by the high turnover numbers and the ability of a single catalyst to handle a broad spectrum of substrates, eliminating the need for multiple specialized reagents. Moreover, the use of inexpensive starting materials like carboxylic acids, which are abundant in the global chemical market, ensures a stable and resilient supply chain. This reduces exposure to price volatility often seen with exotic reagents, providing procurement managers with greater predictability in budgeting and sourcing strategies.
- Cost Reduction in Manufacturing: The elimination of expensive chiral ligands and the use of a universal catalyst system lead to substantial savings in reagent costs. The mild reaction conditions also reduce energy consumption and equipment wear, contributing to a lower overall manufacturing footprint. Additionally, the high selectivity minimizes waste generation, lowering disposal costs and improving the environmental profile of the process.
- Enhanced Supply Chain Reliability: Since the key ligand and iridium source are standard industrial chemicals, the risk of supply disruption is minimized. The robustness of the reaction across various substrates means that production lines can be quickly adapted to manufacture different lactam variants without extensive retooling or requalification, ensuring continuous supply to downstream customers.
- Scalability and Environmental Compliance: The process operates at near-ambient temperatures and pressures, making it inherently safer and easier to scale from kilogram to multi-ton quantities. The reduction in hazardous byproducts and the use of common solvents facilitate easier waste treatment and compliance with increasingly stringent environmental regulations, positioning manufacturers as leaders in sustainable chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this C-H amination technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing production workflows.
Q: How does this method prevent the formation of isocyanate byproducts?
A: The novel iridium catalyst utilizing the 2-(4,5-dihydro-1H-imidazol-2-yl)pyridine ligand exhibits high selectivity for intramolecular C-H insertion, effectively suppressing the competing Curtius rearrangement that typically leads to isocyanate formation.
Q: What are the cost advantages of this catalyst system compared to prior art?
A: Unlike previous methods requiring expensive and complex chiral ligands or multiple specific catalysts, this process utilizes a simple, commercially available ligand structure, significantly reducing raw material costs and simplifying catalyst preparation.
Q: Is this process suitable for large-scale manufacturing of spiro-lactams?
A: Yes, the patent demonstrates successful synthesis of spiro-lactam compounds under mild conditions (40-60°C) with high yields, indicating strong potential for commercial scale-up without requiring extreme temperatures or pressures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Lactam Supplier
As the demand for complex nitrogen heterocycles continues to grow in the pharmaceutical sector, partnering with a technically proficient manufacturer is essential. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced catalytic technologies like the one described in CN111170918A to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of gamma-lactam or delta-lactam intermediate meets the highest international standards. Our commitment to quality ensures that your downstream synthesis proceeds without interruption due to impurity-related failures.
We invite you to explore how our optimized manufacturing processes can enhance your project's economics and timeline. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of modern chemical synthesis with confidence, securing a competitive edge in the global marketplace through innovation and reliability.
