Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Commercial API Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to incorporate fluorinated motifs into bioactive scaffolds, driven by the profound impact of the trifluoromethyl group on metabolic stability and lipophilicity. Patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current synthetic strategies. This technology leverages a non-metallic iodine-promoted cyclization strategy that transforms inexpensive hydrazones and trifluoroethylimidoyl chlorides into high-value heterocyclic intermediates. For R&D directors and procurement specialists, this represents a significant shift away from hazardous diazo compounds and expensive transition metal catalysts, offering a pathway to high-purity 1,2,4-triazole derivatives that are essential for next-generation drug discovery. The simplicity of the operation, combined with the avoidance of stringent anhydrous conditions, positions this chemistry as a prime candidate for commercial scale-up of complex heterocycles in a GMP environment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethylated nitrogen-containing heterocycles has been plagued by significant operational hazards and economic inefficiencies. Traditional literature methods often rely on the direct trifluoromethylation of pre-synthesized heterocyclic cores, which necessitates the use of specialized and often unstable trifluoromethylating reagents that drive up raw material costs dramatically. Alternatively, the use of trifluorodiazoethane as a synthon, while effective, introduces severe safety concerns due to the explosive nature of diazo compounds, requiring specialized equipment and rigorous safety protocols that are difficult to maintain in large-scale production. Furthermore, many existing protocols depend on precious metal catalysts like palladium or copper, which not only increase the bill of materials but also create substantial downstream purification burdens to meet strict residual metal specifications required for API intermediate manufacturing. These factors collectively result in prolonged lead times and inflated production costs, limiting the accessibility of these valuable scaffolds for widespread medicinal chemistry applications.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a metal-free, iodine-promoted cascade reaction that operates under remarkably mild and practical conditions. By employing trifluoroethylimidoyl chloride and hydrazones as readily available building blocks, the process eliminates the need for dangerous diazo reagents and expensive transition metals entirely. The reaction proceeds efficiently in common organic solvents like dichloroethane at moderate temperatures around 80°C, without the necessity for inert atmosphere techniques or rigorous drying of solvents. This operational simplicity translates directly into enhanced process safety and reduced capital expenditure for manufacturing facilities. As illustrated in the general reaction scheme below, the transformation is highly convergent, allowing for the rapid assembly of the triazole core with excellent atom economy.
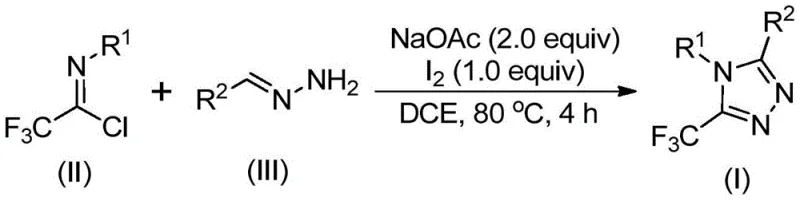
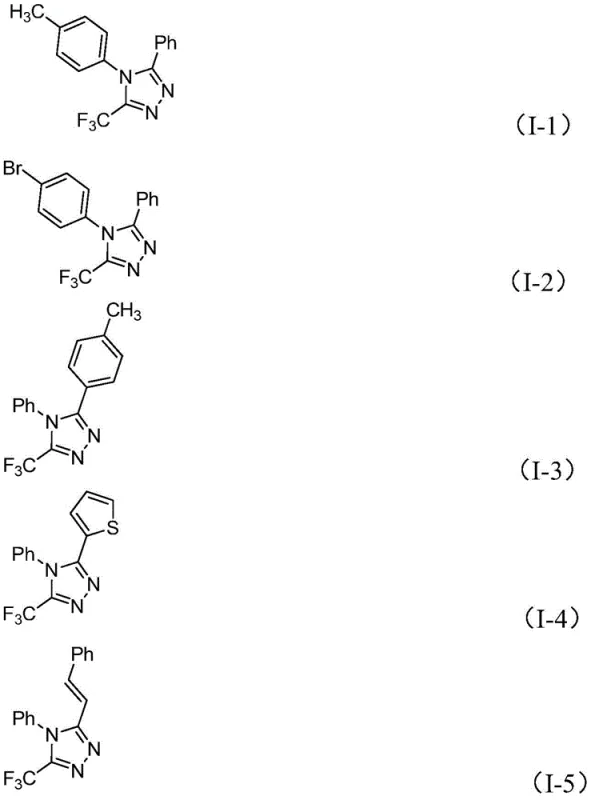
The versatility of this new methodology is further evidenced by its broad substrate scope, accommodating a wide array of electronic and steric environments on both coupling partners. Whether utilizing electron-rich or electron-deficient aryl hydrazones, the system maintains high conversion rates, yielding diverse 5-trifluoromethyl-1,2,4-triazole structures such as those depicted in the examples below. This flexibility is crucial for medicinal chemists who require rapid access to analog libraries for structure-activity relationship (SAR) studies. The ability to tolerate functional groups like halogens, ethers, and nitro groups without protection-deprotection sequences streamlines the synthetic route, significantly reducing the number of unit operations required to reach the final target molecule.
Mechanistic Insights into Iodine-Promoted Oxidative Cyclization
The mechanistic pathway of this transformation offers fascinating insights into how simple reagents can orchestrate complex bond formations without metallic assistance. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond formation between the hydrazone and the trifluoroethylimidoyl chloride, generating a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization to align the reactive centers for cyclization. The critical role of elemental iodine then comes into play during the oxidative phase, where it facilitates the formation of an iodo-intermediate through base-promoted oxidative iodination. This activated species is primed for an intramolecular electrophilic substitution reaction, which closes the five-membered ring. The final step involves aromatization, driven by the elimination of HI, to yield the stable 5-trifluoromethyl-1,2,4-triazole core. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as the stoichiometry of sodium acetate and iodine, to maximize yield and minimize side products.
From an impurity control perspective, the mildness of the reaction conditions is a distinct advantage for maintaining product quality. Harsh acidic or basic conditions often found in traditional heterocycle synthesis can lead to hydrolysis of sensitive functional groups or polymerization of reactive intermediates. However, the buffered environment provided by sodium acetate in this protocol ensures that the reaction proceeds selectively towards the desired triazole product. The absence of transition metals also removes a major source of colored impurities and metal-complexed byproducts that are notoriously difficult to remove via crystallization. Consequently, the crude reaction mixture is often cleaner, reducing the load on downstream purification units like column chromatography or recrystallization, which is a key consideration for achieving cost reduction in API manufacturing where purification often accounts for the majority of processing time and cost.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to ensure optimal reproducibility and safety. The process begins with the precise weighing of sodium acetate, trifluoroethylimidoyl chloride, and the chosen hydrazone substrate, which are then suspended in a suitable organic solvent such as 1,2-dichloroethane (DCE). The mixture is heated to a controlled temperature of 80°C to 100°C, typically maintained for a duration of 2 to 4 hours to ensure complete consumption of the starting materials and formation of the amidine intermediate. Following this initial phase, elemental iodine is introduced to the reaction vessel to trigger the oxidative cyclization step, with the reaction continuing for an additional 1 to 2 hours. Detailed standardized synthesis steps follow below.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane (DCE) under stirring.
- Heat the reaction mixture to 80°C and maintain for 2 to 4 hours to allow initial condensation and isomerization.
- Add elemental iodine to the system and continue heating for 1 to 2 hours to promote oxidative cyclization and aromatization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted synthesis route offers compelling strategic advantages that extend beyond mere chemical elegance. The primary driver for value creation lies in the drastic simplification of the supply chain for raw materials. By utilizing commodity chemicals like hydrazones and imidoyl chlorides instead of proprietary fluorinating agents or unstable diazo compounds, manufacturers can secure a more resilient and cost-effective supply base. This shift reduces dependency on single-source suppliers for exotic reagents, thereby mitigating the risk of production stoppages due to raw material shortages. Furthermore, the elimination of heavy metal catalysts removes the need for expensive scavenging resins and extensive analytical testing for residual metals, which are significant cost centers in pharmaceutical production. These factors combine to create a leaner, more agile manufacturing process capable of responding quickly to market demands.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the replacement of high-cost catalytic systems with inexpensive iodine and sodium acetate. Transition metal catalysts, particularly those based on palladium, represent a volatile cost component subject to global market fluctuations; removing them stabilizes the cost of goods sold (COGS). Additionally, the operational simplicity means that the reaction does not require specialized reactors capable of handling high pressures or extreme temperatures, allowing production to occur in standard glass-lined or stainless steel vessels. The reduced complexity of the workup procedure, facilitated by the absence of metal sludge, lowers labor costs and solvent consumption during purification. Collectively, these efficiencies contribute to a substantially lower production cost per kilogram, enhancing the competitiveness of the final API in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount in the pharmaceutical industry, and this methodology strengthens reliability by relying on widely available starting materials. Hydrazones and trifluoroethylimidoyl chlorides are commercially accessible from multiple vendors, ensuring that procurement teams can negotiate better terms and avoid bottlenecks. The robustness of the reaction conditions, which do not demand strict anhydrous or anaerobic environments, further reduces the risk of batch failures due to environmental excursions. This tolerance makes the process ideal for transfer to contract manufacturing organizations (CMOs) that may have varying levels of infrastructure sophistication. By minimizing the technical barriers to entry for production, companies can diversify their manufacturing network, ensuring a steady flow of pharmaceutical intermediates even during periods of global disruption.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the tonne scale often reveals hidden challenges, but this protocol is inherently designed for scalability. The use of dichloroethane as a solvent is well-established in industrial settings, with mature recovery and recycling protocols already in place. Moreover, the avoidance of toxic heavy metals aligns perfectly with increasingly stringent environmental regulations regarding waste disposal and effluent treatment. Processes that generate heavy metal waste require costly treatment procedures to meet regulatory limits, whereas the waste stream from this iodine-promoted reaction is significantly easier to manage. This environmental compatibility not only reduces compliance costs but also enhances the corporate sustainability profile, a factor that is becoming increasingly important for partnerships with major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 5-trifluoromethyl-1,2,4-triazole synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details is crucial for project managers evaluating the integration of this route into existing production pipelines. The responses cover aspects ranging from catalyst recovery to substrate compatibility, ensuring a comprehensive overview of the technology's capabilities and limitations.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the process utilizes elemental iodine and sodium acetate as promoters, completely avoiding the need for costly palladium or copper catalysts, which simplifies purification and reduces heavy metal residues.
Q: What are the reaction conditions regarding moisture and oxygen?
A: Unlike many sensitive organometallic reactions, this method does not require strict anhydrous or anaerobic conditions, making it highly robust and easier to operate in standard manufacturing environments.
Q: Is the substrate scope limited to specific aryl groups?
A: The method demonstrates broad functional group tolerance, successfully accommodating various substituted aryl groups including methyl, methoxy, bromo, and nitro substituents on both the imidoyl chloride and hydrazone components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iodine-promoted synthesis for the production of high-value fluorinated heterocycles. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented methodology into a robust commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial supply is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch of 5-trifluoromethyl-1,2,4-triazole intermediate adheres to the highest quality standards required by the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce overall manufacturing costs. By leveraging our capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing solutions can support your long-term strategic goals in the development of fluorinated therapeutic agents.
